| Issue |
Parasite
Volume 33, 2026
|
|
|---|---|---|
| Article Number | 14 | |
| Number of page(s) | 18 | |
| DOI | https://doi.org/10.1051/parasite/2026013 | |
| Published online | 23 March 2026 | |
Research Article
Host genetic background influences the severity of disease in Schistosoma haematobium infections
Le fond génétique de l’hôte influence la gravité de la maladie dans les infections à Schistosoma haematobium
1
IHPE, Université de Perpignan Via Domitia, CNRS, IFREMER, Université de Montpellier, Perpignan, France
2
Centre de Recherche pour la Lutte contre les Maladies Infectieuses Tropicales (CReMIT/TIDRC), Université d’Abomey-Calavi, Bénin
3
Institut de Recherche Clinique du Bénin (IRCB), Abomey-Calavi, Bénin
4
Institut de Recherche Pour le Développement (IRD), UMR Intertryp IRD-CIRAD-UM, 34398 Montpellier Cedex 5, France
5
Service de Radiologie et Imagerie Médicale, Centre National Hospitalier et Universitaire Hubert Koutoukou Maga (CNHU-HKM), Cotonou, Bénin
* Corresponding authors: This email address is being protected from spambots. You need JavaScript enabled to view it.
; This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
29
October
2025
Accepted:
5
February
2026
Abstract
Host genetic factors influence the severity of infectious diseases, including schistosomiasis, which are major public-health burdens in Africa. While the role of host genetic background in Schistosoma mansoni infection has been clearly established, this link remains poorly explored for S. haematobium infections (Sh). Therefore, this study aims to investigate the relationship between genetic background and morbidity associated with urogenital schistosomiasis using a candidate gene approach. We analyzed urine samples from 334 Beninese men, measuring urinary eosinophil cationic protein (ECP) by ELISA as a marker for bladder inflammation. Abdominopelvic ultrasonography was performed in a subgroup of 146 participants (69 Sh-positive and 77 Sh-negative) to assess morbidities associated with Schistosoma infection. Blood samples were analyzed for TNF-α levels by ELISA and for TNF-α promoter polymorphisms by sequencing to assess associations between genetic variation and morbidity. Results showed that 25.4% of Sh+ had significantly higher mean TNF-α (U = 5888; p = 0.0098) and ECP (U = 912.5; p < 0.0001) levels than Sh−. Positive correlations were observed between egg count and both ECP (Tau = 0.4016; p < 0.0001) and TNF-α levels (Tau = 0.2238; p = 0.014). Morbidity mainly included bladder irregularities (6%), thickening (29%), and kidney dilation (6%). The G mutant allele on the rs3093660 marker was significantly associated with morbidity (χ2 = 4.47; p = 0.034; OR = 5.09 [95% CI: 1.04–24.9]). Our results suggest, for the first time, that carriers of the G mutant allele at rs3093660 marker have a five-fold increased risk of developing severe urogenital schistosomiasis.
Résumé
Les facteurs génétiques de l’hôte influencent la gravité des maladies infectieuses, notamment la schistosomiase, qui représentent un problème majeur de santé publique en Afrique. Si le rôle du patrimoine génétique de l’hôte dans l’infection à Schistosoma mansoni est bien établi, ce lien reste peu exploré pour les infections à S. haematobium (Sh). Cette étude vise donc à examiner la relation entre le patrimoine génétique et la morbidité associée à la schistosomiase urogénitale par une approche de gènes candidats. Nous avons analysé des échantillons d’urine de 334 hommes béninois, en mesurant la protéine cationique éosinophile urinaire (ECP) par ELISA comme marqueur de l’inflammation vésicale. Une échographie abdominopelvienne a été réalisée chez un sous-groupe de 146 participants (69 Sh-positifs et 77 Sh-négatifs) afin d’évaluer les comorbidités associées à l’infection à Schistosoma. Des échantillons sanguins ont été analysés pour déterminer les taux de TNF-α par ELISA et les polymorphismes du promoteur du TNF-α par séquençage afin d’évaluer les associations entre la variation génétique et la morbidité. Les résultats ont montré que 25,4% des sujets Sh+ présentaient des taux moyens de TNF-α (U = 5888; p = 0,0098) et d’ECP (U = 912,5; p < 0,0001) significativement plus élevés que les sujets Sh−. Des corrélations positives ont été observées entre le nombre d’œufs et les taux d’ECP (Tau = 0,4016; p < 0,0001) ainsi que de TNF-α (Tau = 0,2238; p = 0,014). La morbidité comprenait principalement des irrégularités vésicales (6 %), un épaississement de la paroi vésicale (29 %) et une dilatation rénale (6 %). L’allèle mutant G du marqueur rs3093660 était significativement associé à la morbidité (χ2 = 4,47; p = 0,034; OR = 5,09 [IC à 95 % : 1,04–24,9]). Nos résultats suggèrent pour la première fois que les porteurs de l’allèle mutant G du marqueur rs3093660 présentent un risque cinq fois plus élevé de développer une schistosomiase urogénitale sévère.
Key words: Urogenital schistosomiasis / Morbidity / Genetic factors / TNF-α promoter / Ultrasound / Benin
Edited by Jean-Lou Justine
© B.S.A.E. Savassi et al., published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Schistosomiasis is a major parasitic disease that mainly affects developing countries. In 2020, the World Health Organization (WHO) estimated that about 236 million people were at risk of this infection, with 90% in Africa [98]. In 2021, more than 151 million people were affected by schistosomiasis, with around 12,858 deaths reported worldwide [88]. Classified as a neglected tropical disease, schistosomiasis is the second most prevalent parasitic disease after malaria [19]. Schistosoma haematobium (Sh) and Schistosoma mansoni (Sm), responsible for the urogenital and mesenteric forms of schistosomiasis, respectively, cause the greatest burdens in Africa [34]. Since 2003, schistosomiasis control has focused on mass drug administration programs using praziquantel treatment for children aged 5 to 14 [95]. This treatment reduces prevalence by 91% and intensity of infection by 93% [95]. Despite this apparent effectiveness, schistosomiasis persists as a major health concern in several sub-Saharan African countries [11, 14, 57, 69]. The difficulty in controlling this parasitosis is partly due to the complexity of the parasite’s life-cycle which includes an initial environmental phase, as the disease is contracted through contact with fresh water contaminated by parasite larvae (cercariae) released by a snail vector [81]; and also a second phase that occurs in humans, where susceptibility to infection is influenced by environmental factors (such as density of infected snails and water temperature), socio-behavioral factors (such as frequency and modes of exposure to contaminated water, age, sex, and occupation), as well as factors specific to the human host, including immune response and genetic predispositions that affect resistance or susceptibility to severe forms of the disease [6, 20, 59, 90].
The influence of age, sex, exposure level, and immunity has been extensively studied in Sm infections. Studies have shown considerable differences in schistosome infection levels between individuals, even when infected in the same environment and exposed to similar frequencies [20, 32]. However, these factors account for only 20–25% of the variations between individuals [2, 65, 74]. Numerous studies have demonstrated that human genetic background significantly influences susceptibility to infection and the resulting morbidity. Mathematical models indicate that parasite distribution is highly aggregated, with approximately 80% of eggs released into the environment originating from only 20% of infected patients [102]. Genetic epidemiology studies have shown that 23% to 31% of the risk for Sm infection is due to human genetic factors [21]. The chromosomal region 5q31–q33, which includes several immune genes (IL-4, IL-5, IL-9, and IL-13), has repeatedly been associated with the intensity of infection, particularly with the number of eggs excreted in stools [21, 51, 64]. Other studies on the association between human host genetics and immune responses to helminths have demonstrated a strong correlation between single nucleotide polymorphisms (SNPs) in certain genes (IFN-γ, IL-4, IL-10…etc.) and immune responses to these parasites [29]. Thus, the analysis of single-nucleotide polymorphisms of immune genes appears to be a relevant approach to better understand the interactions between human host genetics and the severity of schistosomiasis. It has recently been reported that 32 candidate SNPs identified in 10 genes could help predict an individual’s susceptibility to developing severe mesenteric schistosomiasis with 63% sensitivity and 90% specificity [59]. Most of these genetic variants are located in genes involved in immunoregulatory functions, such as the TNF-α gene (encoding tumor necrosis factor-alpha, TNF-α) and the RNASE3 gene (encoding eosinophil cationic protein, ECP). TNF-α is a potent pro-inflammatory cytokine that together with interferon-γ promotes granuloma formation, causing immunological pathologies in schistosomiasis [14]. TNF-α overexpression and functional variants located in the TNF-α promoter are implicated in numerous diseases and clinical parameters. For example, the TNF-α polymorphism rs1800629 has been associated with the severity of periportal fibrosis in Sm infected individuals in Brazil [89]. An association between infection intensity and polymorphism at the rs2073342 marker has been observed with respect to ECP [25]. Moreover, ECP also showed significant cytotoxic activity against Sm larvae, with differences in cytotoxicity depending on genetic variants [55, 56, 91].
In schistosome infection, pathology arises primarily from host immune responses directed against eggs trapped in tissues [14]. In morbidity studies of schistosomiasis [8, 58, 59], two major phenotypes are evaluated: (i) the intensity of infection, measured by the number of eggs excreted and/or by the detection of circulating antigens (cathodic circulating antigen in urine and anodic circulating antigen in plasma), and (ii) disease manifestations, including urinary tract lesions (kidney and ureter dilatation) for Sh, and liver lesions (periportal fibrosis, portal hypertension) for Sm [30, 57, 70]. These disease manifestations are detected by ultrasound examinations [79, 80].
Human genetics is known to influence Sm infections, but this is less well studied in Sh infections despite their significant medical implications. Importantly, Sh is the only schistosome species classified as carcinogenic, due to its established association with bladder cancer [13, 35]. In addition, chronic infection frequently results in egg deposition in the genital organs (bladder, cervix) [44, 75], seminal vessels, and prostate [45], which can lead to sterility in adulthood. Furthermore, S. haematobium may contribute to HIV transmission by increasing viral load in semen and inducing hemorrhagic lesions in the cervical mucosa of infected women [60]. In this context, the present study aimed to explore the impact of human genetic background on morbidity associated with bladder and kidney diseases induced by Sh infection. The study focused on immune genes in young men from rural areas in Benin with high schistosomiasis endemicity, specifically the urogenital form. Understanding the influence of genetic factors in the susceptibility and severity of this infection could pave the way for new prevention, treatment, and vaccination strategies [43]. We examined the correlation between the infection levels, the morbidity-related disease manifestations assessed, and the single-nucleotide polymorphisms (SNPs) in the TNF-α and ECP candidate genes. Additionally, studies have shown the roles that these SNPs play in the severity of periportal fibrosis [89] and the intensity of infection [25] in Sm infection. In line with the new WHO roadmap for the elimination of schistosomiasis as a public health problem by 2030 [97], this study provides essential data on the interactions between the genetic diversity of the human host and the morbidity associated with Schistosoma spp. infections.
Materials and methods
Ethics approval
The study was approved by the Comité National d’Éthique pour la Recherche en Santé (CNERS) of the Benin Ministry of Health under authorization reference 063/MS/DC/SGM/CNERS/SA of March 30, 2022. Written consent was obtained from village chiefs and all participants. The parents of patients under 18 years of age were informed and we obtained their oral consent. In cases where those who had to give permission could not read or write, a detailed verbal explanation of the form was given in order to obtain informed consent. Two copies of the written consent form were signed and dated. One copy was retained by each patient, and the second copy was archived in our database. Parasitological and pathological status was communicated to all participants found to have schistosomal infections or other diseases, such as prostatic hypertrophy. Those infected with schistosomiasis were treated with praziquantel (40 mg/kg body weight), in accordance with WHO recommendations [94]. Patients with infections other than schistosomiasis were referred to health centers for more appropriate treatment.
Study sites
This study was carried out in two communes (Sô-Ava and Dangbo) in southern Benin (Fig. 1), where urogenital schistosomiasis transmission is active [85, 86]. Cross-sectional surveys were conducted between March and May 2022 among young male volunteers aged 14 and older in the lakeside villages of Gbéssou, Vêkky, and Houédo (Sô-Ava district) and Késsounou (Dangbo district). These villages are characterized by a sub-equatorial climate with four distinct seasons: (i) a long rainy season from March to June, (ii) a short dry season from July to August, (iii) a short rainy season from September to October, and (iv) a long dry season from November to February.
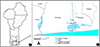 |
Figure 1 Map of the study area showing villages sampled during the survey. A) Geographical situation of Benin. B) Localization of main villages surveyed. |
Sample collection
The sample collection protocol is described in Figure 2. Urine samples were collected from 334 participants in 250 mL containers, and 267 of them also provided approximately 2 mL of venous blood in EDTA tubes. The samples were collected between 10:00 am and 14:00 pm and transported to the laboratory in a refrigerated cooler. For each participant, about 5 mL of homogenized urine was aliquoted and stored at –80 °C within four hours of collection for subsequent measurement of eosinophil cationic protein (ECP). The remaining urine was processed using the filtration method to assess urogenital schistosomiasis infection [73].
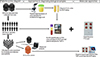 |
Figure 2 Summary of study design. |
Blood plasma was extracted from each sample after centrifugation and was stored at −80 °C to quantify TNF-α levels. Fifty microliters of the blood pellet were then dried using FTA cards for TNF-α and ECP genotyping. Among the participants examined for Schistosoma infection, 146 (including all Sh positive patients (n = 69)) invited participants underwent abdominal-pelvic ultrasound examination.
Urogenital schistosomiasis diagnostic
The urine filtration technique was employed using a 10 mL syringe and a filtration device containing a 13 mm diameter polycarbonate membrane with 12 μm pore size (Polycarbonate (PC) hydrophilic, it4ip, Louvain-la-Neuve, Belgium). For each sample, 10 mL of homogenized urine was drawn with a sterile syringe and passed through the membrane. The membrane was then carefully removed with forceps, placed on a clean glass slide, and stained with a drop of Lugol’s iodine solution. Schistosoma eggs were subsequently counted by microscopic examination.
ECP quantification
Urinary eosinophil cationic protein (ECP) levels were measured using a standard enzyme-linked immunosorbent assay with the MESACUP ECP TEST kit (MBL International, Woburn, MA, USA) [5]. One hundred microliters of prepared urine samples (50 μL of urine sample to 200 μL of assay diluent) were transferred to a 96-well microplate pre-coated with anti-human ECP antibody. After incubation and washing, 100 μL of horseradish peroxidase conjugated anti-human ECP polyclonal antibody was added, followed by tetramethylbenzidine/H2O2 substrate. The absorbance was read at 450 nm using an infinite 200Pro enzyme-linked immunosorbent assay (ELISA) microplate reader (Tecan, Männedorf, Switzerland) and results were interpreted according to the protocol provided by the manufacturer. We also ran the positive and negative controls supplied in the kit in triplicate. Additionally, the results for each plate were validated in accordance with the kit manufacturer’s specifications: a 450 nm absorbance reading of ≤ 0.2 for standard 1 and ≥ 1.2 for standard 6.
TNF-α quantification
Plasma TNF-α levels were measured using a standard ELISA with Invitrogen’s Human TNF-α uncoated kit, following the manufacturer’s protocol. Microtiter plates were coated with 100 μL of anti-human TNF-α antibody, previously diluted according to the manufacturer’s instructions, then incubated overnight at 4 °C. After washing, wells were blocked with the kit’s blocking buffer for 1 hour at room temperature. Serial dilutions of standard TNF-α (recombinant human TNF-α) and samples (patient plasma) were added (100 μL/well), followed by incubation at room temperature for two hours. After a further wash, biotin-conjugated anti-human TNF-α detection antibody and streptavidin-HRP were added. The yellow color obtained after stopping the reaction with tetramethylbenzidine substrate was read at 450 mm in an infinite 200Pro ELISA microplate reader (Tecan).
Assessment of morbidity by ultrasound
A subgroup of patients, both infected (Sh+) and uninfected (Sh−), who provided urine and blood samples were examined by ultrasound. This was done to assess lesions caused by the infection in the urinary tract and at the hepatosplenic level, in order to correlate infection levels with ureteral, renal, and intestinal disorders. The presence of urinary tract and intestinal disorders was determined using a portable ultrasound machine (DP-30, Mindray, Shenzhen, China) equipped with 3.5 MHz convex transducers. The ultrasonographer was unaware of the status of the tested subjects. Half an hour before the ultrasound examination, each patient was given approximately 500 mL of water. The examination was performed only when the bladder was completely full. Subjects were examined in the supine position with the transducer positioned to obtain the best image. Images were recorded and scored for renal size, hydronephrosis, hydroureter, bladder thickening, and bladder irregularities to assess urinary tract disease, while liver, spleen, and abdominal vessel size were scored for bowel disease. All ultrasound image analyses were performed according to the standardized Niamey protocol [79, 80]. The ultrasound data allowed us to determine whether a participant had urinary tract disease by summing the scores as recommended by the WHO according to degree [79, 80]. Participants with a total score of zero (score = 0) and those with a score of at least 1 (score ≥ 1) were classified as having urogenital schistosomiasis with morbidity undetectable by ultrasound (IMU) and with morbidity detectable by ultrasound (IMD), respectively. Other indicators such as the liver image compared with liver patterns A, B, C, D, E, F, X, Y, and U showing the extent of fibrosis or other parenchymal pattern [80], measurements of left and right hepatic lobe size and portal vein internal diameter were adjusted to the patient’s height to assess intestinal lesions. Following WHO guidelines, periportal wall measurements were obtained by assessing the external and luminal diameters of the widest segmental portal branch, measured as near as possible to the branch point [80]. The internal diameter of the portal vein near its entrance into the liver was also measured, and the average of the two measurements was calculated to identify any hepatic periportal thickening. The evaluation of fluid accumulation in the abdomen, known as ascites, as well as the measurements of the sizes of both liver lobes and the portal vein diameter, were used to assess portal hypertension in the patients. Additionally, the height of each patient was measured precisely to the nearest centimeter using a fixed tape measure. Based on the obtained measurements, an index was calculated by dividing the mean participant height by the diameter of the periportal wall, the portal vein, or the size of both liver lobes, in order to correct for patient size differences before assigning scores, as recommended by the WHO [80]. The scores obtained for the portal vein and liver lobes were combined with a score for the presence or absence of ascites to yield a final score for assessing the presence of portal hypertension in the participants. A final score of zero indicates the absence of hepatic portal thickening or portal hypertension.
ECP and TNF-α polymorphism
DNA extraction
We used FTA-card-dried blood samples from patients found to be infected with urogenital schistosomiasis. Genomic DNA was extracted using a QIAamp DNA mini kit (QIAGEN, Hilden, Germany), in accordance with the manufacturer’s instructions.
ECP genotyping
The rs2073342 ECP polymorphism in the ECP gene, located on chromosome 14, was targeted by the restriction fragment length polymorphism (RFLP-PCR) method as previously described [25, 38, 71]. Primers 5′–GTGTGTCATAACCGAGACCGGATAG–3′ and 5′–GGACAGTTGCTGATACCCAGAGTAC–3′ [38] were used for the amplification reaction. Amplification was carried out in a final volume of 25 μL, containing 2 μL of DNA, 1.5 μL of 25 mM MgCl2, 5 μL of 5× buffer (Promega, Madison, WI, USA), 1 μL of each of the 10 μM primers, 0.5 μL of 10 mM dNTP, 0.2 μL of Go Taq polymerase (Promega) and 13.8 μL of milli-Q water. Each cycle included a DNA denaturation step at 95 °C for 40 s, followed by a primer hybridization step at 51 °C for 40 s and an elongation step at 74 °C for 1 min 10 s. The program continued and ended with a final extension step at 74 °C for 5 min. In the next step, 17 μL of PCR products were digested at 37 °C for 15 min using 0.1 μL 10 U PStI restriction enzyme (New England Biolabs, Ipswich, MA, USA), 5 μL of CutSmart buffer, and 27.9 μL of water, in a total reaction volume of 50 μL. The reaction was then inactivated at 80 °C for 20 min. The PStI enzyme cut sites for the ECP-specific SNP rs2073342 were based on the sequence positions 5′–CTGCA↓G–3′ and 3′–G↑ACGTC–5′. This enzymatic digestion cuts the 644 bp ECP fragment into three bands for the heterozygous ECP +434 GC genotype (644 bp, 430 bp, and 214 bp); two bands for the homozygous ECP +434 GG genotype (430 bp and 214 bp), and a single band for the homozygous ECP +434 CC genotype (214 bp) [71]. Digestion of PCR products from the 76 samples that successfully amplified the ECP gene was visualized using 2% agarose electrophoresis gels stained with GelRedTM (Biotium Inc. Darmstadt, Germany). Sequencing confirmed 30 profiles to validate the band profiles obtained from the gels.
TNF-α genotyping
Primer design
To analyze SNPs in the promoter region of the TNF-α gene, two primer pairs were designed using Geneious software, and based on DNA sequences (accession No. NG_007462.1). The primer sequences were as follows: (i) BS_TNF1F: 5′–ATCTGCACCCTCGATGAAGCC–3′ and BS_TNF1R: 5′–ATCTGCACCCTCGATGAAGCC–3′ (amplicon size: 1115 bp); (ii) BS_TNF2F: 5′–ATCAGTCAGTGGCCCAGAAGACC–3′ and BS_TNF2R: 5′–CACCTTCCAGGCATTCAACAGC–3′ (amplicon size: 1,116 bp). Primer specificity was validated using NCBI’s Primer-BLAST, ensuring alignment with the target gene. Furthermore, Sigma Oligoanalyser (https://www.oligoevaluator.com) was used to evaluate potential hairpins, self-dimers, and heterodimers. These two primer pairs collectively covered a 2,122 bp region of the TNF-α gene promoter, allowing comprehensive genotyping of SNPs within the promoter.
DNA amplification and sequencing
For the BS_TNF1F/BS_TNF1R primer pair, the amplification conditions included the activation phase at 95 °C for 5 min, followed by 45 amplification cycles. Each cycle involved a DNA denaturation step at 95 °C for 45 s, followed by a primer hybridization step at 51 °C for 45 s, and an elongation step at 72 °C for 1 min 10 s. The program continued and ended with a final extension phase at 72 °C for 7 min. Amplification conditions for the BS_TNF2F/BS_TNF2R pair consisted of an activation phase at 96 °C for 5 min, followed by 45 amplification cycles. Each cycle included a DNA denaturation step at 96 °C for 45 s, followed by a primer hybridization step at 58 °C for 45 s, and an elongation step at 74 °C for 1 min 10 s. The program continued and ended with a final extension step at 74 °C for 7 min. All PCR products successfully amplified for both primer pairs were purified and sequenced with primers BS_TNF1R: 5′–ATCTGCACCCTCGATGAAGCC–3′ and BS_TNF2R: 5′–CACCTTCCAGGCATTCAACAGC–3′, respectively on an Applied Biosystems genetic analyzer at Genoscreen (Lille, France).
Sequence analysis
Successfully sequenced DNA sequences were assembled and manually edited using Sequencer, version 4.5 (Gene Codes Corporation; http://genecodes.com) to remove ambiguities and sequencing errors. The cleaned sequences were aligned using MUSCLE implemented in MEGA, version 7.0.26 [22, 42]. The two partial TNF-α sequences (TNF1 and TNF2) obtained from the same individual were concatenated using Geneious version 4.8.5 (www.geneious.com) to generate a single consensus sequence. The TNF-α gene is a bi-allelic autosomal gene located on chromosome 6. We used the PHASE module of the DnaSP 6.12.03 program [83] to generate the two allelic sequences for each patient. The program generated two sequences for each of the input sequences, giving a total of 156 sequences for the 78 Sh+. The Haploview program [7] was used to assess genetic linkage between pairs of markers associated with observed morbidity through linkage disequilibrium (LD) analysis. A block matrix was generated based on the degree of linkage using the R2 value (expressed as a percentage), with each number indicating the level of association between marker pairs. To determine whether the distribution of alleles for each SNP conformed to the Hardy-Weinberg equilibrium expectations, and to perform a case-control association analysis (IMD vs IMU), a chi-squared contingency table was constructed and the corresponding values were calculated.
Statistical analysis
A Shapiro test was used to assess the normality of ECP, TNF-α, and Sh infection intensity data. ECP and TNF-α showed a positively skewed distribution. To normalize the distribution and reduce the impact of extreme values, a log10 transformation was applied before statistical analysis. A Kruskal–Wallis test, followed by post-hoc pairwise comparisons using Dunn’s test with Bonferroni adjustment, was also used to assess whether disease status was associated with differences in parasite intensity (egg count), ECP, and plasma TNF-α concentrations.
Differences in immune protein levels (ECP and TNF-α) between patient groups (infected and uninfected) and between marker genotypes were compared using the a Wilcoxon–Mann–Whitney and Kruskal–Wallis tests. Kendall’s correlation was used to determine the association between the mean Schistosoma egg count for Sh and the level of immune proteins (ECP and TNF-α). Chi-square tests with Monte Carlo approximation were used to analyze the distribution of genotypes of markers with three variants between the IMD and IMU groups. Of note, the data were structured in the form of a 2 × 3 contingency table, where the rows represent the groups (IMD and IMU) and the columns the different genotypes. Considering the presence of small numbers for some genotypes, we applied a Monte Carlo approximation with 10,000 simulations in order to obtain a more robust estimate of the p-value. The analysis was performed using the chisq.test() function in R software, with the simulate.p.value option activated to compensate for the small numbers and ensure the validity of the independence test.
A generalized linear model (GLM) was used to perform ANOVA (analysis of variance) to test the effect of genetic variants on the S. haematobium egg count/intensity, and pairwise comparison groups were evaluated using Tukey’s test. The association of alleles with morbidity was also measured using the Chi-square test. Univariate statistical analyses were performed using BiostaTGV (https://biostatgv.sentiweb.fr) and R software. The test was considered significant when the p-value was less than 0.05.
Results
Prevalence of S. haematobium and egg count
Among the participants, 25.4% (85/334) had Sh eggs in their urine. The mean Schistosoma egg count ± standard error of Sh eggs per 10 mL of urine was 16.74 ± 3.27 with the minimum and maximum counts of 1 and 193 eggs/10 mL, respectively.
ECP and TNF-α concentrations in urine and blood
Mean urinary ECP levels were significantly higher in Sh+ than in Sh− (183.4 ± 16.7 ng/mL vs. 3.9 ± 1.3 ng/mL, U = 912.5; p < 0.0001). Similar observations were recorded for mean plasma levels of TNF-α (17.6 ± 9.1 pg/mL vs. 2.5 ± 0.6 pg/mL, U = 5888; p = 0.0098) (Fig. 3a and 3b). Furthermore, there was a significant Kendall correlation between the number of eggs present in urine and ECP levels (Tau = 0.40; p < 0.0001) and TNF-α (Tau = 0.22; p = 0.014) observed in urine and blood, respectively.
 |
Figure 3 Levels of ECP (a) and TNF-α (b) in urine and blood of patients with (Sh+) or without (Sh−) S. haematobium infection. Horizontal bars indicate the geometric means for each group. Log transformations have been used due to the inherent differences in the data for each biomarker (ECP and TNF-α). |
Urinary tract disorders detected on ultrasound
Among the 146 patients examined by ultrasound, 69 (47%) were Sh+ and 77 (53%) were Sh−. The prevalence of urinary tract disorders associated with Sh+ was significantly higher (Fisher Test; p < 0.0001) (36.23%) than in Sh− (7.80%) (Table 1). Our results show that disorders of the bladder (lower urinary tract) were much more frequent than those of the upper urinary tract. Regarding lower urinary tract disorders, bladder thickening was the most frequent (Fig. 4), which accounted for 21 patients (14.38%), followed by bladder irregularities (focal or multifocal) in 8 patients (5.48%). Severe bladder disorders such as masses and polyps were rare, with only one case observed. The prevalence of bladder wall irregularity appeared to be the same in both groups, despite the observed bladder wall thickening, which was significantly more prevalent in Sh+ cases than in Sh− (Fisher Test; p < 0.0001); the prevalence of irregularity appeared to be the same in both groups. As for upper tract disorders, they were observed in the Sh+ group only (Table 1). Dilatation of the kidneys and ureters was observed in four (2.74%) and three (2.05%) patients respectively.
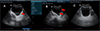 |
Figure 4 Ultrasound images showing some lesions observed in the bladder of patients with (Sh+) or without (Sh−) S. haematobium infection. A: Bladder of irregular shape and thickened wall (red arrow); B: Bladder of irregular shape with pseudopolyps (red arrow); C: Normal bladder; M: Male patient. |
Ultrasound lesion findings in patients with (Sh+) and without (Sh−) S. haematobium infection. The measurements in mm are the size of bladder wall thickening.
The overall severity index scores recorded from the ultrasound ranged from 0 to 20, which enabled the classification of observed conditions into five categories: no disease (score 0), mild (score 1–2), moderate (score 3–4), severe (score 5–6), and very severe (score ≥ 7) (Table 2). Analysis of the morbidity scores observed from ultrasound showed that the severity index was higher and increased (score 0 to score 20) in the Sh+ group compared to the Sh− group (Table 2). The higher frequency of zero morbidity scores as observed in Sh− group revealed the absence of urinary disease.
Overview of the distribution of morbidity scores measured by ultrasound in patients with (Sh+) and without (Sh−) S. haematobium infection.
Concerning lower urinary tract disorders, the observed lesions included abnormal bladder shape, wall irregularities, bladder thickening, weights, and pseudopolyps (Table 1). The first three were more frequent than the last two, with bladder thickening significantly more prevalent in the Sh+ group than in the Sh− group (Fisher test; p < 0.0001). Bladder thickening was classified into four grades, i.e., from 0 to 3 (See Table 1). Most patients in the Sh− group had a normal bladder without thickening (Fisher test; p < 0.0001), except for two cases with mild and severe thickening, respectively. Mild (Fisher test; p = 0.003) and moderate (Fisher test; p = 0.0009) bladder thickening were significantly more frequent in the Sh+ group. Upper urinary tract disorders, including ureteral and renal dilation were only observed in the Sh+ group.
Hepatic disorders detected on ultrasound
Hepatic fibrosis was observed in only one patient with image template D in reference to the Niamey protocol [80]. All other liver images were classified as normal (image template A). No hepatic periportal thickening or portal hypertension were detected in either groups examined (Sh+ and Sh−).
Relationship between urinary tract morbidity and levels of ECP, TNF-α, and parasite intensity
The Kruskal–Wallis test revealed significant differences across disease groups in egg counts (KW = 23.80; p < 0.001) and ECP levels (KW = 19.23; p < 0.001) but not in TNF-α concentrations (KW = 6; p = 0.2) (Figs. 5A to 5C). Post-hoc Dunn’s tests also showed that both the mild and moderate groups had significantly higher egg counts and ECP levels compared to other group (p < 0.05). Urinary ECP levels and the number of eggs counted increased with mild and moderate morbidity, but decreased when morbidity was classified as normal, severe, and very severe.
 |
Figure 5 Relationship between severity of disease (morbidity), ECP levels (A), TNF-α levels (B), and parasite intensity (C) in patients infected with S. haematobium. Score 0 = No disease (normal); Score 1–Score 2 = Mild urinary disease; Score 3–Score 4 = Moderate urinary disease; Score 5–Score 6 = Severe urinary disease; Score ≥ 7 = very severe urinary disease. |
IMD patients (218.27 ± 30.08 ng/mL) had a significantly higher ECP level (W = 1,711; p < 0.0001) than IMU patients (145.83 ± 22.65 ng/mL). Although an elevated mean TNF-α level could be observed in IMD patients (28.69 ± 22.51 pg/mL) compared to IMU patients (14.96 ± 10.49 pg/mL), we were unable to statistically prove this difference (W = 51; p = 0.2763).
ECP and TNF-α gene polymorphisms and their association with the disorder
The genotype frequencies of each marker present on the ECP and TNF-α genes are presented in Table 3. The rs2073342 GC and rs2073342 CC genotypes of the ECP gene were the most common among patients. None of the rs2073342 marker genotypes showed significant differences between the IMD and IMU groups (χ2 = 0.98; p = 0.71) (Table 3). For the TNF-α gene, sequencing of the promoter and a gene fragment showed the presence of (i) five point mutations upstream of the transcription start site, and (ii) two mutations in the first exon (Fig. 6). All seven mutations have previously been reported in other studies, and all have dbSNP identification numbers (−1031 T > C (rs1799964), −863C > A (rs1800630), −857C > T (rs1799724), −308 G > A (rs1800629), −113 C > T/G (rs3093660), +87 G > T (rs2228088), and +489 G > A (rs1800610)). Comparison of the genotype distribution of markers on the TNF-α gene showed no strong evidence of a significant difference between the IMD and UMD groups (Table 3).
 |
Figure 6 Schematic representation of the 2,122 bp promoter and fragment of the sequenced TNF-α gene, showing the location of the various SNPs. |
ECP and TNF-α SNP distribution in patients infected with S. haematobium according to the morbidity level revealed by ultrasound (IMD or IMU).
Adjustment of the data using the linear regression model (GLM) showed that the genetic polymorphisms observed in the ECP and TNF-α genes had a significant effect on the number of eggs observed, except for the rs1800629 marker (Table 4). Tukey’s post-hoc test showed that patients with genotype rs2073342 GG for the ECP gene and rs1799964 CC; rs1800630 AA; rs1799724 TC; rs3093660 GG; rs2228088 GG for the TNF-α gene were associated with a significantly higher number of eggs in the urine compared to the other genotypes found on the same markers (Supplementary file 1). The level of the ECP immune protein was significantly higher in patients with the GG genotype than in those with the CC genotype for the rs3093660 marker (W = 75; p = 0.04) (Supplementary file 2). No significant differences were observed between genotypes for the other ECP markers. There were also no significant differences in plasma TNF-α levels between genotypes (Supplementary file 2).
Summary of the generalized linear model output to determine the effect of TNF-α and ECP polymorphisms on eggs counts.
Linkage disequilibrium (LD) analysis between marker pairs located in the TNF-α gene revealed a weak correlation between rs1799724 and rs1800610 (R2 = 0.13), and a moderate correlation between rs1799964 and rs1800630 (R2 = 0.38). For all other marker pairs, no correlation (R2 = 0) or a negligible level of association (R2 = 0.01) was observed (Fig. 7). A statistically significant deviation from expectations under the Hardy-Weinberg equilibrium could be detected in TNF-α mutations at positions −1031, −113, and +489. Allelic case-control (IMD vs IMU) association analysis revealed that only the G mutant allele at position -133, located on marker rs3093660 within the TNF-α gene, was significantly associated with morbidity (Table 5). Our findings show that the distribution of TNF-α alleles differed significantly between IMD patients and IMU (χ2 = 4.47, p = 0.034). The G allele was more frequent among IMD patients (15.4%) than among IMU patients (3.4%), corresponding to an odds ratio of 5.09 [95% CI: 1.04–24.9]. The C allele on this marker (rs3093660) seemed not to be linked to the severity of infection.
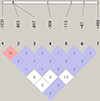 |
Figure 7 Tests for pair-wise linkage disequilibrium tests between SNPs in Sh infected groups in relation to morbidity. The numbers in the boxes are the R2 (%) values. The stronger the association between two SNPs, the higher the R2 value. |
Test for association of TNF-α gene alleles with morbidity and Hardy–Weinberg equilibrium in patients infected with S. haematobium.
Of the 12 haplotypes observed (H1–H12), H1 comprising wild-type alleles at all seven polymorphic sites was the most common, with a significantly higher frequency (Fisher Exact test; p = 0.001) in the IMU group (40%) compared to the IMD group (10%) (Table 6). H2, with the allele mutated at position −308, was present in both groups with a lower frequency than H1, but also higher in the IMU group (8%) than in the IMD group (4%) (Table 6). H4, H7, and H8, with alleles mutated at positions −113, −863, and −1031 had a higher frequency in the IMD group (5%, 5%, and 4%, respectively) than in the IMU group (2%, 2%, and 1%, respectively) (Table 6). H3 and H5, which respectively had two mutated alleles (positions −1031 and −863) and one mutated allele (+87), were higher in the IMU group (Table 6). The rare haplotypes: H6, H9, H10, H11, and H12 occurred at a frequency of 1%. Among these, H6, H9, and H10 were only observed in the IMU group, whereas H11 and H12 appeared exclusively in the IMD group (Table 6). However, there was no observed statistically significant difference between the IMD and IMU groups for the H2 –H12 haplotypes.
Haplotypes (H1 to H12) observed and their frequency according to IMD and IMU groups in patients infected with S. haematobium. Haplotype H1 has only wild-type alleles at the segregation sites, but the other haplotypes (H2 to H12) have at least one mutant allele at one of the segregation sites (see highlighted in black).
Discussion
Our results showed an overall average S. haematobium prevalence of 24.5%, with variations between villages examined. This prevalence is comparable with that observed in other endemic countries, such as Mali [9], Mauritania, and Senegal [1]. However, this prevalence exceeds the national prevalence observed five years earlier, which was 17.6% [69]. This difference may be attributed to the sampling target, as our study involved larger populations, including adults and out-of-school children, not included in current treatment strategies, showing that these non-targeted groups are actively involved in schistosomiasis transmission and harbor high rates of infection. This further emphasizes the need to extend routing preventive chemotherapy treatment to the entire population in endemic areas, such as the communes of Sô-Ava and Dangbo, in order to achieve the schistosomiasis elimination targets set by the WHO [96].
The morbidity parameters associated with S. haematobium infection were validated by both biomarkers and ultrasound analysis. The significant elevation of both ECP and TNF-α concentrations in the urine and plasma of Sh+, respectively supports the idea that Sh infection induces both systemic and local immune responses. These findings are consistent with previous studies, including those by Masamba and Kappo [53], Asuming-Brempong et al. [5], and Wamachi et al. [92], who reported activation of inflammatory response during Schistosoma infections. The significant correlation between parasite intensity and levels of these immune proteins suggests that they could be used as indicators of parasite load in epidemiological monitoring [61, 78]. Considering urinary disorders measured by ultrasound, over 36% of Sh+ showed bladder abnormalities, including bladder wall thickening, frequently observed in chronic Sh infections [24, 26, 37, 40, 48]. In contrast, no liver disease was detected, consistent with previous studies showing that periportal fibrosis is specific to intestinal schistosomiasis [54]. Our data differ from those reported by Agniwo et al. [3], who observed a high prevalence (56–90%) of bladder irregularities in children in Mali. This could indicate that the disease course is age-related, with bladder thickening observed in adults and more frequent irregularities in children. This mechanism is thought to result from host immune response to eggs trapped in the urinary tract, triggering a Th2 response and the formation of granulation tissue around trapped eggs [14, 18, 87, 103]. Consequently, adults may experience regular egg deposition over the years due to repeated reinfections, leading to the continuous formation of granulation and scar tissue in the bladder, which will ultimately result in thickening of the bladder wall. In contrast, bladder disease in children remains confined to changes in bladder shape, which explains the frequent irregularity observed in this group. Our results also show that infected patients with morbidity detected (IMD) by ultrasound have significantly higher ECP levels than infected patients without morbidity detection (IMU). This phenomenon could indicate more marked activation of immune response in patients with severe forms of the disease. However, plasma TNF-α levels showed no significant difference between IMD and IMU groups, which is similar to the observations of Wamachi et al. [92]. The low prevalence of severe disease, such as dilatation of the ureters (3 patients) or kidneys (4 patients), and masses and polyps (2 patients), could suggest that the infection is in a less advanced phase, or that acquired immunity limits the progression of lesions. This could explain the low prevalence of these serious diseases, even though biomarkers indicate immune activation. The observed bladder disorder in Sh− participants could also be associated with early treatment, while pathology reversal is still in process, which in most cases takes longer (3 to 6 months in school-age children [10] and 1 to 2 years in adults [49]) than the elimination of urinary egg excretion.
Our findings also highlight a significant association between morbidity status and both the parasite burden and eosinophil-mediated immune activity, but not with systemic TNF-α concentrations. Specifically, individuals classified with mild and moderate morbidity exhibited significantly higher egg counts and elevated ECP levels compared to those in the normal, severe, and very severe groups. This unexpected pattern, where moderate morbidity is associated with higher egg excretion than severe morbidity, may reflect a phenomenon well described in chronic schistosomiasis where the fibrous lesions progressively trap the eggs in the tissues, reducing their excretion in the urine despite the persistence of disease [15, 17, 68, 82]. Similar cases have been observed in patients with S. mansoni intestinal schistosomiasis, with advanced liver damage due to chronic inflammation and periportal fibrosis [31]. These findings underscore the limited sensitivity of egg detection methods in chronic stages and the need for complementary biomarkers. The observation of higher ECP levels in mild and moderate groups compared to severe groups points to the key role that active eosinophilic inflammation plays in early or transitional disease [76, 77]. These results are in contrast to the findings of Reimert et al. [78] and Leutscher et al. [46], who reported a significant increase in urinary ECP levels with progression from mild to severe schistosomiasis morbidity in patients older than 5 years, whereas higher ECP concentrations were observed in individuals between 6 and 19 years than those over 20 years of age [78]. ECP level decline in severe morbidity groups may reflect a shift from inflammatory to fibrotic responses, possibly regulated by immunosuppressive mechanisms such as regulatory T cells or altered cytokine signaling [33, 39, 100]. In contrast, no significant variation in TNF-α levels was observed between morbidity categories. TNF-α plays a central role in granuloma formation and fibrosis in experimental models of schistosomiasis [16]. However, systemic measures may not adequately reflect localized immune responses or may be masked by host immune modulation mechanisms [47], co-infections [72], or chronic immune exhaustion in endemic populations [101]. These results are consistent with previous reports indicating that TNF-α is not a strong systemic marker of disease severity in chronic schistosomiasis [66, 93].
Analysis of ECP and TNF-α polymorphisms revealed interesting associations between certain genotypes and the parasite egg counts or disease severity. Concerning ECP, the different genotypes of the rs2073342 marker were not associated with disease severity. However, patients carrying the rs2073342 GG genotype exhibited significantly higher egg counts in their urine compared to those with other genotypes. Considering that ECP is known to be a marker of eosinophil activation in parasitic infections [4, 84], this suggests that the GG genotype might play a role in modulating parasite intensity, potentially by influencing eosinophil activity. These results are consistent with previous studies that have highlighted the involvement of ECP in immune response, particularly in helminth infections where eosinophils play a critical role in controlling parasite burdens [12, 50, 62, 77].
Genetic variations in the TNF-α promoter gene are increasingly recognized as a key factor influencing host susceptibility to severe helminthic infections. In this study, we examined seven polymorphisms previously described in the literature. The absence of significant linkage disequilibrium between markers rs1800629, rs3093660, and rs2228088 located at positions −308, −113, and +87, respectively and the other SNPs analyzed suggests that these variants are not co-segregating. This result confirms the hypothesis that each of these polymorphisms can independently influence host immune response to Sh infection. Of these, the G allele at position −113 (rs3093660) was significantly associated with increased morbidity. Individuals homozygous for the G allele (GG genotype) were five times more likely to develop serious clinical manifestations than individuals homozygous for the C allele (CC genotype). This observation is consistent with previous reports suggesting that TNF-α promoter polymorphisms can influence TNF-α expression, promoting excessive inflammation and tissue damage [89]. In particular, elevated TNF-α levels have been linked to liver fibrosis in schistosomiasis [16, 33]. Other promoter variants such as rs1800629 (−308G > A) and rs361525 (−238G > A) have been shown to alter TNF-α transcription and overproduction [99]. Although rs3093660 has been less explored, this study reinforces its potential role in driving granulomatous inflammation and severe immunopathology during helminthic infections. Surprisingly, this study found no significant difference in plasma TNF-α levels between patients carrying the CC and GG genotypes of the rs3093660 polymorphism (Supplementary file 2). However, ECP levels were significantly higher in patients carrying the GG genotype than in those carrying the CC genotype. These results suggest that this variant of the TNF-α gene promoter may modulate ECP expression by indirect mechanisms. This observation raises an important question: how could the G variant of the rs3093660 gene influence ECP levels without altering systemic TNF-α concentrations? Several plausible mechanisms can be envisaged: (i) local modulation: the G allele of rs3093660, located in the TNF-α promoter region, may selectively affect local cytokine expression and eosinophil activation in inflamed tissues [27, 67]; (ii) cytokine synergy: the variant can indirectly modulate eosinophil responses by interacting with the IL-4 and IL-5 signaling pathways, which are known to lead to the release of ECP [28, 63]; and (iii) post-transcriptional regulation: TNF-α is subject to complex regulatory mechanisms, including mRNA stability, translation efficiency, and protein turnover, which can dissociate the effects of promoter polymorphism from plasma cytokine levels [23, 36, 41, 52].
In terms of haplotype analysis, in the study, we identified 12 haplotypes based on the seven mutations in the TNF-α gene. The H1 haplotype, carrying wild-type alleles at all seven mutation sites, was the most common and showed a significantly higher frequency in the IMU group compared to the IMD group. This suggests that the H1 haplotype may be protective or associated with less severe disease. In contrast, haplotypes H4, H7, and H8, which carried mutations at positions −113, −863, and −1031, were more frequent in the IMD group, suggesting that these haplotypes may be linked to morbidity. Our results highlight the complex influence of host genetic variation on immune regulation and progression of schistosomiasis, ultimately shaping patterns of morbidity and clinical severity.
Although this study provides valuable information on the genetic and immunological mechanisms underlying schistosomiasis, it has several limitations that should be taken into account in future research. First, the ultrasound assessments were carried out on only 146 patients, which may limit the generalizability of the results. To address this, future studies should aim to include larger and more diverse populations in different endemic regions, allowing for more robust and representative assessments of morbidity patterns. Second, TNF-α levels were not measured directly at sites of infection (e.g., in urine samples), limiting our ability to assess local cytokine activity. Incorporating tissue-specific or excretory biomarker measurements into future research would provide clearer data on local immune responses and inflammation. Third, the overall sample size included in the genetic association analysis was relatively small, potentially limiting the statistical power to detect subtle genetic associations. Increasing the size of the cohort through multicenter collaborations would increase analytical power and improve detection of genotype-phenotype correlations. Although our genetic analysis focused on known polymorphisms, it might have missed rare or novel variants. Therefore, the use of high-throughput genomic approaches, such as next-generation sequencing (NGS), could enable more comprehensive exploration of the genetic loci underlying disease severity. Fourth, the cross-sectional nature of the study does not allow us to understand the temporal dynamics of infection or immune responses. Longitudinal cohort studies that monitor changes in biomarkers and morbidity over time would help to clarify causal relationships and the progression of disease.
In conclusion, this study highlights the complex interaction between Sh infection, immune response, and genetic factors. Biomarkers such as ECP and TNF-α could potentially be used as reliable indicators to assess disease severity. Furthermore, genetic polymorphisms, in particular the rs3093660 variant of the TNF-α gene, are emerging as relevant predictive markers for identifying patients at risk of severe forms, enabling clinicians to adapt therapeutic strategies. Finally, the study highlights the determinant role of genetic factors in susceptibility to urinary schistosomiasis and its clinical course. Further research is still needed to validate these genetic associations and clarify their impact on disease progression.
Abbreviations
Sh : Schistosoma haematobium
Sh+: Patients infected with Sh
Sh−: Patients uninfected with Sh
Sm : Schistosoma mansoni
IL: interleukin
DNA: Deoxyribonucleic acid
SNP: Single nucleotide polymorphism
TNF-α: Tumor necrosis factor-alpha
ECP: Eosinophil cationic protein
CNERS: Comité national d’éthique pour la recherche en santé
IMU: Infected with morbidity undetected on ultrasound
IMD: Infected with morbidity detected on ultrasound
p : p-value
LD: linkage disequilibrium
RFLP-PCR: Restriction fragment length polymorphism - polymerase chain reaction
U: Mann–Whitney statistic test
KW: Kruskal–Wallis statistic test
GLM: Generalized linear models
χ2: Chi-Square test
Acknowledgments
We would like to thank Campus France (France) for the MOPGA4 grant.
The authors would like to thank Dr. Rafiou Adamou and Dr. Paulin Sonon for their assistance in demonstrating the use of the ELISA system and the Haploview software, which were used for data analysis. We would particularly like to thank the local authorities and the medical/nursing chief officers of the villages of Gbéssou, Vêkky, Houédo (Sô-Ava district), and Késsounou (Dangbo district) for their support in carrying out the field activities.
Funding
This research was funded by the program AFZOONS (ANR-CE35-6032) from the French Research National Agency. This study is set within the framework of the “Laboratoires d’Excellence (LABEX)” TULIP (ANR-10-LABX-41) and the International joint laboratory CONS-HELM.
Conflicts of interest
All authors approved the final version of this manuscript and declare that they have no competing interests.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Author contribution statement
B.A.E.S. Conceptualization, Formal Analysis, Investigation, Methodology, Visualization, Writing – Original Draft Preparation, Writing – Review & Editing. S-D.K.M., D.B., and G.H. Investigation, Writing – Review & Editing N.L.A and E.T. Formal Analysis, Writing – Review & Editing M.I and A.M. Formal Analysis, Methodology, Supervision, Writing – Review & Editing. D.C. Formal Analysis, Methodology, Writing – Review & Editing. J.B. Conceptualization, Formal Analysis, Project Administration, Supervision, Writing – Review & Editing
Supplementary material
 |
Supplementary file 1: SNP marker genotype structuring analysis based on average number of eggs in patients infected with S. haematobium. |
Analysis of the structure of the SNP marker genotypes was carried out according to the average number of eggs obtained after the GLM analysis (see Table 4). On the histograms of the genotypes, the presence of identical letters (“a” or “b”) above the bars indicates that there is no significant difference between these genotypes (e). On the other hand, different letters indicate a statistically significant difference (f).
 |
Supplementary file 2: Comparative analysis of urinary ECP (left) and plasma TNF-α concentrations (right) according to the genotypes identified for each marker in samples from patients infected with S. haematobium. |
References
- Abdellahi M, Ndir O, Niang S.. 2016. Évaluation de la prévalence des bilharzioses auprès des enfants de 5 à 14 ans après plusieurs années de traitement de masse dans le bassin du fleuve Sénégal. Santé Publique, 28(4), 535–540. [Google Scholar]
- Abel L, Dessein AJ. 1997. The impact of host genetics on susceptibility to human infectious diseases. Current opinion in Immunology, 9(4), 509–516. [Google Scholar]
- Agniwo P, Sidibé B, Diakité A, Niaré SD, Guindo H, Akplogan A, Ibikounlé M, Boissier J, Dabo A. 2023. Ultrasound aspects and risk factors associated with urogenital schistosomiasis among primary school children in Mali. Infectious Diseases of Poverty, 12(1), 40. [CrossRef] [PubMed] [Google Scholar]
- Amoani B, Adu B, Frempong MT, Sarkodie-Addo T, Nuvor SV, Wilson MD, Gyan B. 2019. Levels of serum eosinophil cationic protein are associated with hookworm infection and intensity in endemic communities in Ghana. PLoS One, 14(9), e0222382. [Google Scholar]
- Asuming-Brempong E, Gyan B, Amoah AS, van der Puije W, Bimi L, Boakye D, Ayi I. 2015. Relationship between eosinophil cationic protein and infection intensity in a schistosomiasis endemic community in Ghana. Research and reports in Tropical Medicine, 6, 1–10. [Google Scholar]
- Ayabina DV, Clark J, Bayley H, Lamberton PH, Toor J, Hollingsworth TD. 2021. Gender-related differences in prevalence, intensity and associated risk factors of Schistosoma infections in Africa: A systematic review and meta-analysis. PLoS Neglected Tropical Diseases, 15(11), e0009083. [Google Scholar]
- Barrett JC, Fry B, Maller J, Daly MJ. 2005. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics, 21(2), 263–265. [CrossRef] [PubMed] [Google Scholar]
- Bethony JM, Quinnell RJ. 2008. Genetic epidemiology of human schistosomiasis in Brazil. Acta Tropica, 108(2–3), 166–174. [Google Scholar]
- Bintou L, Sodio B, Sacko M. 2019. Persistance de la schistosomiase urinaire en zones endémiques soumises aux traitements de masse répétés au Mali. International Journal of Biological and Chemical Sciences, 13(1), 369–381. [Google Scholar]
- Bocanegra C, Pintar Z, Mendioroz J, Serres X, Gallego S, Nindia A, Aznar ML, Soriano-Arandes A, Salvador F, Gil E. 2018. Ultrasound evolution of pediatric urinary schistosomiasis after treatment with praziquantel in a highly endemic area. American Journal of Tropical Medicine and Hygiene, 99(4), 1011. [Google Scholar]
- Boissier J, Mouahid G, Moné H. 2019. Rose JB, Jiménez-Cisneros B, Schistosoma spp., in: Global Water Pathogen Project, Michigan State University, E. Lansing, MI, UNESCO. [Google Scholar]
- Boix E, Torrent M, Sánchez D, Nogues MV. 2008. The antipathogen activities of eosinophil cationic protein. Current pharmaceutical biotechnology, 9(3), 141–152. [Google Scholar]
- Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L. 2009. A review of human carcinogens – Part B: biological agents. Lancet Oncology, 10(4), 321–322. [Google Scholar]
- Burke M, Jones M, Gobert G, Li Y, Ellis M, McManus D. 2009. Immunopathogenesis of human schistosomiasis. Parasite Immunology, 31(4), 163–176. [Google Scholar]
- Cheever AW, Duvall RH. 1981. Bladder calcification and obstructive uropathy in a gibbon infected with Schistosoma haematobium. American Journal of Tropical Medicine and Hygiene, 30(3), 604–608. [Google Scholar]
- Cheever AW, Hoffmann KF, Wynn TA. 2000. Immunopathology of schistosomiasis mansoni in mice and men. Immunology Today, 21(9), 465–466. [Google Scholar]
- Cheever AW, Young SW, Shehata A. 1975. Calcification of Schistosoma haematobium eggs: relation of radiologically demonstrable calcification to eggs in tissues and passage of eggs in urine. Transactions of the Royal Society of Tropical Medicine and Hygiene, 69(4), 410–414. [Google Scholar]
- Colley D, Secor W. 2014. Immunology of human schistosomiasis. Parasite Immunology, 36(8), 347–357. [CrossRef] [PubMed] [Google Scholar]
- Colley DG, Bustinduy AL, Secor WE, King CH. 2014. Human schistosomiasis. Lancet, 383(9936), 2253–2264. [CrossRef] [PubMed] [Google Scholar]
- Dessein A, Rihet P, Demeure C, Couissinier P, Bacellar O, Carvalho EM, Kohlstaedt S, Dessein H, Souza A, Prata A. 1992. Facteurs génétiques et immunologiques déterminant la résistance à la bilharziose en région d’endémie. Médecine Sciences, 8, 108–118. [Google Scholar]
- Dessein H, Duflot N, Romano A, Opio C, Pereira V, Mola C, Kabaterene N, Coutinho A, Dessein A. 2020 Genetic algorithms identify individuals with high risk of severe liver disease caused by schistosomes. Human Genetics, 139, 821–831. [Google Scholar]
- Edgar RC. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32(5), 1792–1797. [CrossRef] [PubMed] [Google Scholar]
- El-Tahan RR, Ghoneim AM, El-Mashad N. 2016. TNF-α gene polymorphisms and expression. Springerplus, 5, 1–7. [CrossRef] [PubMed] [Google Scholar]
- Elmadani AE, Hamdoun AO, Monis A, Karamino NE, Gasmelseed N. 2013. Ultrasound findings in urinary shistosomaisis infection in school children in the Gezira State Central Sudan. Saudi Journal of Kidney Diseases and Transplantation, 24(1), 162–167. [Google Scholar]
- Eriksson J, Reimert CM, Kabatereine NB, Kazibwe F, Ireri E, Kadzo H, Eltahir HB, Mohamed AO, Vennervald BJ, Venge P. 2007. The 434 (G > C) polymorphism within the coding sequence of Eosinophil Cationic Protein (ECP) correlates with the natural course of Schistosoma mansoni infection. International Journal for Parasitology, 37(12), 1359–1366. [Google Scholar]
- Fall CB, Lambert S, Léger E, Yasenev L, Garba AD, Diop SD, Borlase A, Catalano S, Faye B, Walker M. 2021. Hybridized zoonotic Schistosoma infections result in hybridized morbidity profiles: A clinical morbidity study amongst co-infected human populations of Senegal. Microorganisms, 9(8), 1776. [Google Scholar]
- Fettrelet T, Gigon L, Karaulov A, Yousefi S, Simon H-U. 2021. The enigma of eosinophil degranulation. International Journal of Molecular Sciences, 22(13), 7091. [Google Scholar]
- Fulkerson PC, Rothenberg ME. 2013. Targeting eosinophils in allergy, inflammation and beyond. Nature reviews Drug Discovery, 12(2), 117–129. [Google Scholar]
- Fumagalli M, Pozzoli U, Cagliani R, Comi GP, Bresolin N, Clerici M, Sironi M. 2010. The landscape of human genes involved in the immune response to parasitic worms. BMC Evolutionary Biology, 10, 1–15. [Google Scholar]
- Gaye AM, Doh K, Thiam I, Bentefouet L, Woto-Gaye G. 2016. Bilharziose et cancer: une association fortuite ou une relation de cause à effet. Bulletin du Cancer, 103(9), 806–807. [Google Scholar]
- Gryseels B, Polman K, Clerinx J, Kestens L. 2006. Human schistosomiasis. Lancet, 368(9541), 1106–1118. [CrossRef] [PubMed] [Google Scholar]
- Gurarie D, Yoon N, Li E, Ndeffo-Mbah M, Durham D, Phillips AE, Aurelio HO, Ferro J, Galvani AP, King CH. 2015. Modelling control of Schistosoma haematobium infection: predictions of the long-term impact of mass drug administration in Africa. Parasites & vectors, 8(1), 1–14. [Google Scholar]
- Hesse M, Piccirillo CA, Belkaid Y, Prufer J, Mentink-Kane M, Leusink M, Cheever AW, Shevach EM, Wynn TA. 2004. The pathogenesis of schistosomiasis is controlled by cooperating IL-10-producing innate effector and regulatory T cells. Journal of Immunology, 172(5), 3157–3166. [Google Scholar]
- Huyse T, Van den Broeck F, Hellemans B, Volckaert F, Polman K. 2013. Hybridisation between the two major African schistosome species of humans. International Journal for Parasitology, 43(8), 687–689. [CrossRef] [PubMed] [Google Scholar]
- IA RC. 2012. IARC. Biological Agents: Monographs on the evaluation of carcinogenic risks to humans. International Agency for Research on Cancer, Lyon, France, 100B, 499. [Google Scholar]
- Imarenezor E, Nmorsi O, Brown S, Abhadionmhen OYaO. 2016. Interleukin (IL)–10 and tumour necrosis factor–alpha (TNF–α) profiles of individuals with Schistosoma haematobium infection in Ewan community, Edo state, Nigeria. FUW Trends in Science & Technology Journal, 1, 24–26. [Google Scholar]
- Joekes E, McMonnies K, Blanshard A, Mutuku FM, Ireri E, Mungai P, Stothard JR, Bustinduy AL, King CH. 2023. A 14-year follow-up of ultrasound-detected urinary tract pathology associated with urogenital schistosomiasis in women living in the Msambweni region of coastal Kenya. Transactions of The Royal Society of Tropical Medicine and Hygiene, 117(9), 637–644. [Google Scholar]
- Jönsson UB, Byström J, Stålenheim G, Venge P. 2002. Polymorphism of the eosinophil cationic protein‐gene is related to the expression of allergic symptoms. Clinical & Experimental Allergy, 32(7), 1092–1095. [Google Scholar]
- Kamdem SD, Moyou-Somo R, Brombacher F, Nono JK. 2018. Host regulators of liver fibrosis during human schistosomiasis. Frontiers in Immunology, 9, 2781. [Google Scholar]
- King CH. 2002 Ultrasound monitoring of structural urinary tract disease in Schistosoma haematobium infection. Memórias do Instituto Oswaldo Cruz, 97, 149–152. [Google Scholar]
- King CL, Malhotra I, Mungai P, Wamachi A, Kioko J, Muchiri E, Ouma JH. 2001. Schistosoma haematobium induced urinary tract morbidity correlates with increased tumor necrosis factor-α and diminished interleukin-10 production. Journal of Infectious Diseases, 184(9), 1176–1182. [Google Scholar]
- Kumar S, Stecher G, Tamura K. 2016. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33(7), 1870–1874. [CrossRef] [PubMed] [Google Scholar]
- Kwok AJ, Mentzer A, Knight JC. 2021. Host genetics and infectious disease: new tools, insights and translational opportunities. Nature Reviews Genetics, 22(3), 137–153. [Google Scholar]
- Leutscher P, Raharisolo C, Pecarrere J, Ravaoalimalala V, Serieye J, Rasendramino M, Vennervald B, Feldmeier H, Esterre P. 1997. Schistosoma haematobium induced lesions in the female genital tract in a village in Madagascar. Acta Tropica, 66(1), 27–33. [Google Scholar]
- Leutscher P, Ramarokoto C-E, Reimert C, Feldmeier H, Esterre P, Vennervald BJ. 2000. Community-based study of genital schistosomiasis in men from Madagascar. Lancet, 355(9198), 117–118. [Google Scholar]
- Leutscher PD, Reimert CM, Vennervald BJ, Ravaoalimalala VE, Ramarokoto CE, Serieye J, Raobelison A, Rasendramino M, Christensen NO, Esterre P.. 2000. Morbidity assessment in urinary schistosomiasis infection through ultrasonography and measurement of eosinophil cationic protein (ECP) in urine. Tropical Medicine & International Health, 5(2), 88–93. [Google Scholar]
- Lundy SK, Lukacs NW. 2013. Chronic schistosome infection leads to modulation of granuloma formation and systemic immune suppression. Frontiers in Immunology, 4, 39. [CrossRef] [PubMed] [Google Scholar]
- Ma SM, Adamu B. 2015. Pattern of urinary bladder sonographic findings in patients evaluated for urinary schistosomiasis. West African Journal of Radiology, 22(2), 92–96. [Google Scholar]
- Magak P, Chang-CojulunA, Kadzo H, Ireri E, Muchiri E, Kitron U, King CH. 2015. Case–control study of posttreatment regression of urinary tract morbidity among adults in Schistosoma haematobium endemic communities in Kwale County, Kenya. American Journal of Tropical Medicine and Hygiene, 93(2), 371. [Google Scholar]
- Mahmoud AA. 1982. The ecology of eosinophils in schistosomiasis. Journal of Infectious Diseases, 145(5), 613–622. [Google Scholar]
- Marquet S, Abel L, Hillaire D, Dessein A. 1999. Full results of the genome-wide scan which localises a locus controlling the intensity of infection by Schistosoma mansoni on chromosome 5q31–q33. European Journal of Human Genetics, 7(1), 88–97. [Google Scholar]
- Marume A, Chimponda T, Vengesai A, Mushayi C, Mann J, Mduluza T. 2021. Effects of TNF-α and IL-10–819 T > C single nucleotide polymorphisms on urogenital schistosomiasis in preschool children in Zimbabwe. African Journal of Laboratory Medicine, 10(1), 1–7. [Google Scholar]
- Masamba P, Kappo AP. 2021. Immunological and biochemical interplay between cytokines, oxidative stress and schistosomiasis. International Journal of Molecular Sciences, 22(13), 7216. [Google Scholar]
- Mawa PA, Kincaid-Smith J, Tukahebwa EM, Webster JP, Wilson S. 2021. Schistosomiasis morbidity hotspots: Roles of the human host, the parasite and their interface in the development of severe morbidity. Frontiers in Immunology, 12, 751. [Google Scholar]
- McLaren DJ, McKean J, Olsson I, Venge P, Kay A. 1981. Morphological studies on the killing of schistosomula of Schistosoma mansoni by human eosinophil and neutrophil cationic proteins in vitro. Parasite Immunology, 3(4), 359–373. [Google Scholar]
- McLaren DJ, Peterson C, Venge P. 1984. Schistosoma mansoni: further studies of the interaction between schistosomula and granulocyte-derived cationic proteins in vitro. Parasitology, 88(3), 491–503. [Google Scholar]
- McManus D, Dunne DW, Sacko M, Utzinger J, Vennervald BJ, Zhou X-N. 2018. Schistosomiasis. Nature Reviews Diseases Primers, 4, 13. [Google Scholar]
- Mewamba EM, Noyes H, Tiofack AAZ, Kamga RMN, Kamdem CN, Mengoue LET, Ofon E, Ngassam RIK, Nyangiri O, Bucheton B. Association between polymorphisms of IL4, IL13, IL10, STAT6 and IFNG genes, cytokines and immunoglobulin E levels with high burden of Schistosoma mansoni in children from schistosomiasis endemic areas of Cameroon. Infection, Genetics and Evolution, 111, 105416. [Google Scholar]
- Mewamba EM, Nyangiri OA, Noyes HA, Egesa M, Matovu E, Simo G. 2021. The genetics of human schistosomiasis infection intensity and liver disease: A review. Frontiers in Immunology, 12, 613468. [Google Scholar]
- Midzi N, Mduluza T, Mudenge B, Foldager L, Leutscher PD. 2017. Decrease in seminal HIV-1 RNA load after praziquantel treatment of urogenital schistosomiasis coinfection in HIV-positive men an observational study. Open Forum Infectious Diseases. US: Oxford University Press. [Google Scholar]
- Midzi N, Ndhlovu PD, Nyanga L, Kjetland E, Reimert C, Vennervald B, Gomo E, Mudenge G, Friis H, Gundersen S. 2003. Assessment of eosinophil cationic protein as a possible diagnostic marker for female genital schistosomiasis in women living in a Schistosoma haematobium endemic area. Parasite Immunology, 25(11–12), 581–588. [Google Scholar]
- Midzi N, Ndhlovu PD, Nyanga L, Kjetland EF, Reimert CM, Vennervald BJ, Gomo E, Mudenge G, Friis H, Gundersen SG, Mduluza T. 2003. Assessment of eosinophil cationic protein as a possible diagnostic marker for female genital schistosomiasis in women living in a Schistosoma haematobium endemic area., Parasite Immunology, 25(11–12), 581–588. [Google Scholar]
- Molfino N, Gossage D, Kolbeck R, Parker J, Geba G. 2012. Molecular and clinical rationale for therapeutic targeting of interleukin‐5 and its receptor. Clinical & Experimental Allergy, 42(5), 712–737. [Google Scholar]
- Müller-Myhsok B, Stelma FF, Guisse-Sow F, Muntau B, Thye T, Burchard GD, Gryseels B, Horstmann RD. 1997. Further evidence suggesting the presence of a locus, on human chromosome 5q31–q33, influencing the intensity of infection with Schistosoma mansoni. American Journal of Human Genetics, 61(2), 452. [Google Scholar]
- Mutapi F, Burchmore R, Mduluza T, Midzi N, Turner CMR, Maizels RM. 2008. Age-related and infection intensity-related shifts in antibody recognition of defined protein antigens in a schistosome-exposed population. Journal of Infectious Diseases, 198(2), 167–175. [Google Scholar]
- Mutengo MM, Mduluza T, Kelly P, Mwansa JC, Kwenda G, Musonda P, Chipeta J. 2018. Low IL‐6, IL‐10, and TNF‐α and high IL‐13 cytokine levels are associated with severe hepatic fibrosis in Schistosoma mansoni chronically exposed individuals. Journal of Parasitology Research, 2018(1), 9754060. [Google Scholar]
- Netea MG, Balkwill F, Chonchol M, Cominelli F, Donath MY, Giamarellos-Bourboulis EJ, Golenbock D, Gresnigt MS, Heneka MT, Hoffman HM. 2017. A guiding map for inflammation. Nature Immunology, 18(8), 826–831. [Google Scholar]
- Olveda DU, Ross AG. 2017. Chronic schistosomiasis, in: Schistosoma. CRC Press. pp. 368–386. [Google Scholar]
- Onzo-Aboki A, Ibikounlé M, Boko PM, Savassi BS, Doritchamou J, Siko EJ, Daré A, Batcho W, Massougbodji A, Tougoue JJ. 2019. Human schistosomiasis in Benin: Countrywide evidence of Schistosoma haematobium predominance. Acta Tropica, 191, 185–197. [Google Scholar]
- Palumbo E. 2007. Association between schistosomiasis and cancer: a review. Infectious Diseases in Clinical Practice, 15(3), 145–148. [Google Scholar]
- Pereira MC, Oliveira DT, Olivieri EH, Rogatto SR, Carvalho AL, Landman G, Kowalski LP. 2010. The 434 (G > C) polymorphism in the eosinophil cationic protein gene and its association with tissue eosinophilia in oral squamous cell carcinomas. Journal of Oral Pathology & Medicine, 39(1), 56–62. [Google Scholar]
- Perera DJ, Koger-Pease C, Paulini K, Daoudi M, Ndao M. 2024. Beyond schistosomiasis: unraveling co-infections and altered immunity. Clinical Microbiology Reviews, 37(1), e00098-23. [Google Scholar]
- Peters P, Mahmoud AA, Warren K, Ouma J, Siongok TA. 1976. Field studies of a rapid, accurate means of quantifying Schistosoma haematobium eggs in urine samples. Bulletin of the World Health Organization, 54(2), 159. [Google Scholar]
- Pinot de Moira A, Fulford AJ, Kabatereine NB, Ouma JH, Booth M, Dunne DW. 2010. Analysis of complex patterns of human exposure and immunity to Schistosomiasis mansoni: the influence of age, sex, ethnicity and IgE. PLoS Neglected Tropical Diseases, 4(9), e820. [CrossRef] [PubMed] [Google Scholar]
- Randrianasolo BS, Jourdan PM, Ravoniarimbinina P, Ramarokoto CE, Rakotomanana F, Ravaoalimalala VE, Gundersen SG, Feldmeier H, Vennervald BJ, Van Lieshout L. 2015. Gynecological manifestations, histopathological findings, and schistosoma-specific polymerase chain reaction results among women with Schistosoma haematobium infection: a cross-sectional study in Madagascar. Journal of Infectious Diseases, 212(2), 275–284. [Google Scholar]
- Reimert C, Venge P, Kharazmi A, Bendtzen K. 1991. Detection of eosinophil cationic protein (ECP) by an enzyme-linked immunosorbent assay. Journal of Immunological Methods, 138(2), 285–290. [Google Scholar]
- Reimert CM, Fitzsimmons CM, Joseph S, Mwatha JK, Jones FM, Kimani G, Hoffmann KF, Booth M, Kabatereine NB, Dunne DW, Vennervald BJ. 2006. Eosinophil activity in Schistosoma mansoni infections in vivo and in vitro in relation to plasma cytokine profile pre- and posttreatment with Praziquantel. Clinical and Vaccine Immunology, 13(5), 584–593. [Google Scholar]
- Reimert CM, Mshinda HM, Hatz CF, Kombe Y, Nkulila T, Poulsen LK, Christensen N, Vennervald B. 2000. Quantitative assessment of eosinophiluria in Schistosoma haematobium infections: a new marker of infection and bladder morbidity. American Journal of Tropical Medicine and Hygiene, 62(1), 19–28. [Google Scholar]
- Richter J, Botelho M, Holtfreter M, Akpata R, El Scheich T, Neumayr A, Brunetti E, Hatz C, Dong Y, Dietrich CF. 2016 Ultrasound assessment of schistosomiasis. Zeitschrift für Gastroenterologie, 54(07), 653–660. [Google Scholar]
- Richter J, Hatz C, Campagne G, Bergquist N, Jenkins JM. 2000. Ultrasound in schistosomiasis: a practical guide to the standard use of ultrasonography for assessment of schistosomiasis-related morbidity: Second international workshop, October 22–26 1996. Niamey, Niger: World Health Organization [Google Scholar]
- Rollinson D, Southgate VR. 1987. The genus Schistosoma: a taxonomic appraisal, in: The biology of schistosomes from genes to latrines, Academic: London. pp. 350. [Google Scholar]
- Ross A, Bartley P, Sleigh A. 2002. Schistosomiasis. New England Journal of Medicine, 346, 1212–1220. [Google Scholar]
- Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A. 2017. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Molecular Biology and Evolution, 34(12), 3299–3302. [CrossRef] [PubMed] [Google Scholar]
- Sakyi SA, Amoani B, Opoku S, Dzata L, Aniagyei W, Senu E, Dankwa K, Wilson MD. 2022. Assessing the role of eosinophil-mediated immune response markers in detecting hookworm infection: A case-control study in Kintampo, Ghana. Health Science Reports, 5(4), e674. [Google Scholar]
- Savassi BA, Dobigny G, Etougbétché JR, Avocegan TT, Quinsou FT, Gauthier P, Ibikounlé M, Moné H, Mouahid G. 2021. Mastomys natalensis (Smith, 1834) as a natural host for Schistosoma haematobium (Bilharz, 1852) Weinland, 1858 x Schistosoma bovis Sonsino, 1876 introgressive hybrids. Parasitology Research, 120(5), 1755–1770. [CrossRef] [PubMed] [Google Scholar]
- Savassi BAES, Mouahid G, Lasica C, Mahaman SK, Garcia A, Courtin D, Allienne JF, Ibikounlé M, Moné H. 2020. Cattle as natural host for Schistosoma haematobium (Bilharz, 1852) Weinland, 1858 x Schistosoma bovis Sonsino, 1876 interactions, with new cercarial emergence and genetic patterns. Parasitology Research, 119(7), 2189–2205. [CrossRef] [PubMed] [Google Scholar]
- Shebel HM, Elsayes KM, Abou El Atta HM, Elguindy YM, El-Diasty TA. Genitourinary schistosomiasis: life cycle and radiologic-pathologic findings. Radiographics, 32(4), 1031–1046. [Google Scholar]
- Shen Z, Luo H. 2025 The impact of schistosomiasis on the Global Disease Burden: a systematic analysis based on the 2021 Global Burden of Disease study. Parasite, 32, 12. [Google Scholar]
- Silva PCV, Gomes AV, de Britto LRPB, de Lima ELS, da Silva JL, Montenegro SML, Muniz MTC, Domingues ALC. 2017. Influence of a TNF-α polymorphism on the severity of schistosomiasis periportal fibrosis in the northeast of Brazil. Genetic Testing and Molecular Biomarkers, 21(11), 658–662. [Google Scholar]
- Trienekens SC, Faust CL, Meginnis K, Pickering L, Ericsson O, Nankasi A, Moses A, Tukahebwa EM, Lamberton PH. 2020. Impacts of host gender on Schistosoma mansoni risk in rural Uganda — A mixed-methods approach PLoS Neglected Tropical Diseases, 14(5), e0008266. [Google Scholar]
- Trulson A, Byström J, Engström Å, Larsson R, Venge P. 2007. The functional heterogeneity of eosinophil cationic protein is determined by a gene polymorphism and post‐translational modifications. Clinical & Experimental Allergy, 37(2), 208–218. [Google Scholar]
- Wamachi AN, Mayadev JS, Mungai PL, Magak PL, Ouma JH, Magambo JK, Muchiri EM, Koech DK, King CH, King C. 2004. Increased ratio of tumor necrosis factor-α to interleukin-10 production is associated with Schistosoma haematobium-induced urinary-tract morbidity. Journal of Infectious Diseases, 190(11), 2020–2030. [Google Scholar]
- Wang X, Tang Q, Bergquist R, Zhou X, Qin Z. 2023. The cytokine profile in different stages of schistosomiasis japonica. Pathogens, 12(10), 1201. [Google Scholar]
- WHO. 2002. Prevention and control of schistosomiasis and soil-transmitted helminthiasis: report of a WHO expert committee. World Health Organization. [Google Scholar]
- WHO. 2011. Report of a meeting to review the results of studies on the treatment of schistosomiasis in preschool-age children. p. 32. [Google Scholar]
- WHO. 2020. Ending the neglect to attain the Sustainable Development Goals: a road map for neglected tropical diseases 2021–2030. World Health Organization. [Google Scholar]
- WHO. 2022. Ending the neglect to attain the sustainable development goals: One health: approach for action against neglected tropical diseases 2021–2030. World Health Organization. [Google Scholar]
- WHO. 2022. WHO guideline on control and elimination of human schistosomiasis. World Health Organization. [Google Scholar]
- Wilson AG, Symons JA, McDowell TL, McDevitt HO, Duff GW. 1997. Effects of a polymorphism in the human tumor necrosis factor α promoter on transcriptional activation. Proceedings of the National Academy of Sciences, 94(7), 3195–3199. [Google Scholar]
- Wilson MS, Mentink-Kane MM, Pesce JT, Ramalingam TR, Thompson R, Wynn TA. 2007. Immunopathology of schistosomiasis. Immunology and cell Biology, 85(2), 148–154. [Google Scholar]
- Wilson RA, Coulson PS. 2009. Immune effector mechanisms against schistosomiasis: looking for a chink in the parasite’s armour. Trends in Parasitology, 25(9), 423–431. [Google Scholar]
- Woolhouse ME, Dye C, Etard J-F, Smith T, Charlwood J, Garnett G, Hagan P, Hii Jx, Ndhlovu P, Quinnell R. 1997. Heterogeneities in the transmission of infectious agents: implications for the design of control programs. Proceedings of the National Academy of Sciences, 94(1), 338–342. [Google Scholar]
- Wynn TA, Thompson RW, Cheever AW, Mentink‐Kane MM. 2004. Immunopathogenesis of schistosomiasis. Immunological Reviews, 201(1), 156–167. [Google Scholar]
Cite this article as: Savassi BSAE, Mahaman S-DK, Biaou D, Luviano Aparicio N, Ibikounl M, Toulza E, Courtin D, Hounkanrin G, Massougbodji A & Boissier J.. 2026. Host genetic background influences the severity of disease in Schistosoma haematobium infections. Parasite 33, 14. https://doi.org/10.1051/parasite/2026013.
All Tables
Ultrasound lesion findings in patients with (Sh+) and without (Sh−) S. haematobium infection. The measurements in mm are the size of bladder wall thickening.
Overview of the distribution of morbidity scores measured by ultrasound in patients with (Sh+) and without (Sh−) S. haematobium infection.
ECP and TNF-α SNP distribution in patients infected with S. haematobium according to the morbidity level revealed by ultrasound (IMD or IMU).
Summary of the generalized linear model output to determine the effect of TNF-α and ECP polymorphisms on eggs counts.
Test for association of TNF-α gene alleles with morbidity and Hardy–Weinberg equilibrium in patients infected with S. haematobium.
Haplotypes (H1 to H12) observed and their frequency according to IMD and IMU groups in patients infected with S. haematobium. Haplotype H1 has only wild-type alleles at the segregation sites, but the other haplotypes (H2 to H12) have at least one mutant allele at one of the segregation sites (see highlighted in black).
All Figures
 |
Figure 1 Map of the study area showing villages sampled during the survey. A) Geographical situation of Benin. B) Localization of main villages surveyed. |
| In the text | |
 |
Figure 2 Summary of study design. |
| In the text | |
 |
Figure 3 Levels of ECP (a) and TNF-α (b) in urine and blood of patients with (Sh+) or without (Sh−) S. haematobium infection. Horizontal bars indicate the geometric means for each group. Log transformations have been used due to the inherent differences in the data for each biomarker (ECP and TNF-α). |
| In the text | |
 |
Figure 4 Ultrasound images showing some lesions observed in the bladder of patients with (Sh+) or without (Sh−) S. haematobium infection. A: Bladder of irregular shape and thickened wall (red arrow); B: Bladder of irregular shape with pseudopolyps (red arrow); C: Normal bladder; M: Male patient. |
| In the text | |
 |
Figure 5 Relationship between severity of disease (morbidity), ECP levels (A), TNF-α levels (B), and parasite intensity (C) in patients infected with S. haematobium. Score 0 = No disease (normal); Score 1–Score 2 = Mild urinary disease; Score 3–Score 4 = Moderate urinary disease; Score 5–Score 6 = Severe urinary disease; Score ≥ 7 = very severe urinary disease. |
| In the text | |
 |
Figure 6 Schematic representation of the 2,122 bp promoter and fragment of the sequenced TNF-α gene, showing the location of the various SNPs. |
| In the text | |
 |
Figure 7 Tests for pair-wise linkage disequilibrium tests between SNPs in Sh infected groups in relation to morbidity. The numbers in the boxes are the R2 (%) values. The stronger the association between two SNPs, the higher the R2 value. |
| In the text | |
 |
Supplementary file 1: SNP marker genotype structuring analysis based on average number of eggs in patients infected with S. haematobium. |
| In the text | |
 |
Supplementary file 2: Comparative analysis of urinary ECP (left) and plasma TNF-α concentrations (right) according to the genotypes identified for each marker in samples from patients infected with S. haematobium. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


