| Issue |
Parasite
Volume 21, 2014
|
|
|---|---|---|
| Article Number | 68 | |
| Number of page(s) | 17 | |
| DOI | https://doi.org/10.1051/parasite/2014067 | |
| Published online | 17 December 2014 | |
Review Article
Notes on the genus Tunga (Siphonaptera: Tungidae) II – neosomes, morphology, classification, and other taxonomic notes
Notes sur le genre Tunga (Siphonaptera: Tungidae) II – néosomes, morphologie, classification et autres notes taxonomiques
1
Departamento de Parasitologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais, Caixa Postal 486, Avenida Presidente Antônio Carlos, 6627, Campus UFMG, Belo Horizonte, Minas Gerais
31270-901, Brazil
2
Laboratoire de Parasitologie et Zoologie appliquée, Faculté de Médecine 2, avenue du Professeur Léon Bernard, 35043
Rennes Cedex, France
3
Institut de Parasitologie de l’Ouest, Faculté de Médecine 2, avenue du Professeur Léon Bernard, 35043
Rennes Cedex, France
4
Laboratório de Pesquisas Clínicas, Centro de Pesquisas René Rachou, Fundação Oswaldo Cruz, Belo Horizonte, Minas Gerais, Brazil
5
Laboratoire de Parasitologie, Mycologie et Immunologie parasitaire, Centre Hospitalier Régional Universitaire, 2 rue Henri Le Guilloux, 32033
Rennes Cedex, France
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
19
August
2014
Accepted:
3
December
2014
Abstract
This review focuses on the neosomes, morphology, and taxonomy of adult species of the genus Tunga, complementing the previously published data on the phylogeny, ecology, and pathogenic role. Neosomes are structures formed after penetration of adult females into the skin of hosts resulting in significant enlargement, being the most characteristic and most frequently observed form in hosts. Neosomes can be differentiated by shape, measurements, and sites of attachment to principal hosts. The taxonomic value and morphometric data of the most widely used characteristics to separate species – such as frontal curvature, head chaetotaxy, preoral internal sclerotization, ventral and dorsal genal lobes, eyes, maxillary palps, fusion of pronotum and mesonotum, metacoxae, metatarsi chaetotaxy, spermatheca (females), manubrium, basimere, telomere, and phallosome (males) – are comparatively analyzed. The sexes, individual variations, undescribed species, higher taxa, as well as a proposal for division of the genus into two subgenera (Tunga and Brevidigita) are presented (as previously given by Wang). A key for females, males, and gravid females (neosomes) also is included for identifying the 13 known species. Data on host specificity and geographical distribution may also support the identification of Tunga species because some sand fleas and their hosts may have co-evolved.
Résumé
Ce travail est axé sur les néosomes, la morphologie et la taxonomie des adultes du genre Tunga, complétant les données déjà publiées sur la phylogénie, l’écologie et le rôle pathogène de ces espèces. Les néosomes sont des structures élaborées après la pénétration de la femelle adulte dans les téguments de l’hôte: ils montrent une taille significativement plus grande et sont les formations les plus caractéristiques et les plus fréquemment rencontrées chez les hôtes. Les néosomes peuvent être différenciés par leur forme, leurs dimensions et les zones de localisation sur leurs principaux hôtes. La valeur taxonomique et les données morphométriques des caractères les plus utilisés pour la séparation des espèces – tels que la courbure frontale, la chétotaxie céphalique, les sclérifications préorales internes, le lobe génal interne et externe, la forme et le développement de l’œil, la conformation des palpes maxillaires, le degré de fusion du pronotum et du mesonotum, la forme et la chétotaxie des metacoxae, la chétotaxie des métatarses, chez les femelles la forme de la spermathèque, chez les mâles, la forme du manubrium, du basimère, du télomère et du phallosome, sont comparées et discutées. Les sexes, les variations individuelles, les taxa non décrits, les classifications de plus haut niveau, de même qu’une proposition (déjà donnée par Wang) de diviser ce genre en deux sous-genres (Tunga and Brevidigita), sont exposés. Une clé des 13 espèces décrites est proposée pour les femelles, les mâles et les femelles gravides, ou néosomes. La spécificité et la distribution géographique peuvent également aider à l’identification des espèces de Tunga car diverses puces-chiques semblent avoir co-évolué avec leurs hôtes.
Key words: Tunga / Siphonaptera / Sand fleas / Neosomes / Morphology / Taxonomy
© P.M. Linardi et al., published by EDP Sciences, 2014
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Currently, the genus Tunga comprises 13 species, representing less than 0.5% of the world flea fauna, which consists of approximately 3,000 known species [27]. One species, T. penetrans (L., 1758) [35], presents a wide distribution and a high degree of specificity occurring in the Neotropical region and sub-Saharan Africa, in spite of occasional reports of individuals from the USA [7, 45], Europe [49], and New Zealand [42] in whom sand flea infestations were diagnosed after travel to infested regions. Two species, T. caecigena Jordan and Rothschild, 1921 [24] and T. callida Li and Chin, 1957 [29], parasitize essentially commensal rats in the Oriental Region, while T. monositus Barnes and Radovsky, 1969 [2] infests wild rodents in the southwestern USA [19]. Nine other species occur in the Neotropics; four are found on wild and commensal rodents [6, 20, 37]: T. caecata (Enderlein, 1901) [15]; T. libis Smit, 1962 [46]; T. bossii De Avelar, Linhares, and Linardi, 2012 [13] and T. bonneti Beaucournu and González-Acuña, 2012 [5, 6]; three infest edentates [21, 30, 34]: T. travassosi Pinto and Dreyfus, 1927 [43]; T. bondari Wagner, 1932 [50] and T. terasma Jordan, 1937 [22]; and two occur on domestic Artiodactyla and man [38, 39, 41]: T. trimamillata Pampiglione, Trentini, Fioravanti, Onori, and Rivasi, 2002 [40]; and T. hexalobulata De Avelar, Facury Filho and Linardi, 2013 [11]. It is important to stress that more than 30% of the species have been described since 2002, and 23% of these species were described only in the last 2 years, indicating that opportunities for new findings are abundant.
Among the Siphonaptera, Tunga is the most specialized genus because the adult females penetrate into the skin of their hosts. Similar to other fleas, both males and females are blood-feeding [18, 53], however, the larvae and adult male of T. monositus do not feed [26].
Recently, a review of the genus Tunga concerning taxonomy, phylogeny, ecology and pathogenic role was presented by Beaucournu et al. [5], although the review does not include T. hexalobulata, which was described subsequently. Similarly, De Avelar et al. [13], when describing T. bossii for the first time, presented a widely used dichotomous key for identifying the known species and their neosomes and excluded T. bonnetti and T. hexalobulata, which were discovered later.
The present study complements this review with regard to the neosomes, morphology, classification, and other taxonomic notes. The taxonomic value and morphometric data of several characteristics are comparatively analyzed. Although the morphological aspects are often not known for many taxa, here we present a new key for identifying the 13 known Tunga species, including a more classical vision and emphasizing neosomal characteristics.
Neosomes
Neosomes are altered organisms resulting from a process characterized by the growth of new tissue and the formation of a new morphological structure accompanied by significant enlargement after adult eclosion [1]. This phenomenon, previously designated by Jordan [23] as teleomorphosis, is known as neosomy and occurs among several arthropods, including queen ants (Formicidae), queen termites (Termitidae), termitophilus beetles (Staphylinidae), Diptera (Phoridae, Streblidae, Carnidae), parasitic Copepoda, Acari (Trombiculidae, Ixodidae [but not Argasidae], Pyemotidae), and some Siphonaptera.
Approximately 90 species of fleas have sessile or semi-sessile females that, after eclosion, remain more or less permanently attached to the integument of the host [44]. Considering the permanence of the hypertrophied females on the hosts, these fleas can be classified as (i) internal, in the host beneath the skin (mesoparasites): Tunga (Tungidae) and Neotunga (Pulicidae); (ii) an external, sessile female, permanently attached as soon as it is on its host (ectoparasites): Echidnophaga (Pulicidae), Hectopsylla (Tungidae); or (iii) an external, non-sessile female, temporarily attached when on its host: Chaetopsylla pro parte, Dorcadia, Vermipsylla (Vermipsyllidae), Parapsyllus pro parte (Rhopalopsyllidae), Glaciopsyllus (Ceratophyllidae), Ancistropsylla (Ancistropsyllidae), Malacopsylla and Phthiropsylla (Malacopsyllidae).
Morphological studies of the genus Tunga have concentrated on the description of the neosomes because they are the most characteristic and most frequently observed form in hosts. Females and males measure approximately 1 mm, but after penetration, the gravid females increase considerably in size, reaching approximately 10 mm (Fig. 1), and expose, on the surface of hosts, only the vital respiratory, anal, and vaginal apertures, which are contained in a caudal disk or conical prominence. The caudal disk exhibits some differences among species: (i) flattened, as in T. penetrans (Fig. 2), T. monositus, and T. bonneti; (ii) conical, as in T. caecata, T. travassosi, T. trimamillata, and T. hexalobulata; or (iii) cylindrical, as in T. terasma, T. bondari, T. caecigena, and T. callida. Otherwise, the caudal disk can be (i) as wide as long, or almost as wide as long, as in T. caecata, T. travassosi, and T. callida; (ii) wider than long, as in T. penetrans, T. monositus, T. trimamillata, T. bossii, and T. hexalobulata; or (iii) longer than wide, as in T. terasma, T. bondari, and T. caecigena. Neosomes can live more than three months attaching to different sites of their respective hosts [14, 26]. Consequently, neosomes are important for identifying the species in the genus Tunga.
 |
Figure 1. A neosome of the caecata group of species: (A) embedded on the ear of Nectomys squamipes (×3); (B) frontal view after extraction (×12). |
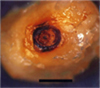 |
Figure 2. A neosome of Tunga penetrans – posterior view. Scale bar = 2 mm. |
A review solely on the neosomes of tungid fleas that infest wild and domestic animals and concentrated mainly on hosts, infestation, sites of attachment, and impact on the hosts was recently presented by Linardi and Avelar [32].
Table 1 shows the known Tunga species with their respective geographic distributions, sexes, stages, neosomes, and sites of attachment to principal hosts. Excepting neosomes, in which the size is indicated in millimeters, all measurements included in other tables are in micrometers.
Tunga species: Geographic distribution, sexes, stages, neosomes, and sites of attachment to their principal true hosts.
Morphology
Excepting neosomes, the classical structures more frequently used to separate or group the species of Tunga are discussed as follows:
Head (Fig. 3)
The cephalic capsule includes the majority of the characteristics used for identifying these species. Members of the genera Tunga, Hectopsylla (with the exception of the female of Hectopsylla coniger), Echidnophaga, Phacopsylla, and Neotunga euloidea are always characterized by an angular profile and a well-pronounced frontal tubercle, as in T. caecigena (Fig. 3C), T. travassosi (Fig. 3D), T. bondari (Fig. 3E), T. trimamillata (Fig. 3J), T. bossii (Fig. 3K), and T. hexalobulata (Fig. 3M). The front curvature varies little but is gently convex or sharper as in T. caecigena (Fig. 3C) or almost straight as in T. libis (Fig. 3H). Generally, the ventral profile of the cephalic capsule shows a genal lobe, which is often more pronounced in females; it seems absent in bossii, but is very clear in caecata, caecigena, travassosi, bondari, terasma, and monositus. Preoral internal sclerotization presents as the posterior arm longer than the anterior arm (caecata, monositus, trimamillata, and hexalobulata) or with the posterior arm subequal to or shorter than the anterior arm (caecigena, penetrans, bondari, bossii), or much shorter than the anterior arm (callida). A dorsal genal lobe either covers part of the antennal segment III (bondari, travassosi, caecigena, callida) or not, as in other species. The number of bristles on the postantennal region, posterior region to the frontal tubercle, antennal segment II, and base of the maxilla may be a differential characteristic. Inside the cephalic capsule, the eyes are the most characteristic structures for species identification. They may be absent in caecigena and callida or present in other species, or they may be pigmented (penetrans, bondari, terasma, libis, trimamillata, bonneti, and hexalobulata) or without black pigment (caecata, travassosi, monositus, and bossii). When present, the eyes can be small (caecata, travassosi, and monositus), or large, in the case of other species. In some species such as penetrans, bondari, trimamillata, the eyes also have a recess into their internal margins. The greatest eye diameters and the numeric variations of bristles on some structures of Tunga species are presented in Table 2.
 |
Figure 3. The heads of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. callida; (H) T. libis; (I) T. monositus; (J) T. trimamillata; (K) T. bossii; (L) T. bonneti; (M) T. hexalobulata. |
Eye and numeric variations of bristles on some structures of Tunga species.
Maxillary palps (Fig. 4) with segments vary in size and chaetotaxy; in caecigena, callida, and bonneti, segment IV is longer than segment I, though the reverse is true in T. trimamillata and T. hexalobulata. In other species, the palps are approximately the same length. In T. bossii, segment I was described as very small and segments III and IV as being incompletely divided; however, a new observation showed that, in fact, what was thought to be “segment I” was a strengthened area at the base of the palp and an incomplete division occurred between segments II and III and between segments III and IV.
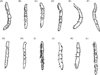 |
Figure 4. The maxillary palps of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. callida; (H) T. monositus; (I) T. trimamillata; (J) T. bossii; (K) T. bonneti; (L) T. hexalobulata. |
Morphometric data regarding the maxillary palps, including new measurements for T. bossii, laciniae and preoral internal sclerotization are indicated in Table 3.
Morphometric variations in maxillary palps and laciniae in Tunga species.
Thorax
As Smit [46] noted, the fusion of the pronotum and mesonotum can be dorsally complete, a characteristic of the penetrans group or incomplete, as in the caecata group. Currently, the penetrans group includes T. penetrans, T. travassosi, T. bondari, T. terasma, T. trimamillata, and T. hexalobulata [11, 13]. In the caecata group, the following species are included: T. caecata, T. caecigena, T. callida, T. libis, T. monositus, T. bossii, and T. bonneti. The chaetotaxy is generally sparse, consisting of 1–7 bristles on the prothorax. Legs III show the classical increase in length, compared to the first two pairs, typical of Siphonaptera. However, these legs are slender, and members of the Tunga genus are poor jumpers despite the existence of a pleural arch. At this leg, Beaucournu et al. [5] noted an external guard tooth, whose size is variable depending on the species and ranges from completely absent to normally developed. The coxae are always preserved regardless of the autotomy of the legs. The metacoxae (Fig. 5) project downward at an anterodistal angle, like a wide tooth, and exhibit a variable number of bristles both on the external and internal surfaces. Species such as penetrans, trimamillata, and hexalobulata present a greater number of bristles. In the species of the caecata group, the metacoxae are slightly wider at the base than at the apex (Figs. 5E, H, I), though in the penetrans group, excepting T. terasma (Fig. 5F), the basal part is nearly two times wider than the apical part (Figs. 5A, B, H, J, K). According to Hopkins and Rothschild [20], T. penetrans is characterized by a slower and less complete deterioration of the legs than the other species known at that time. The chaetotaxy of tibiae, tarsi, and especially of the distal segment of leg III are discriminant characteristics. According to Smit [46], the chaetotaxy of the distal tarsal in the species belonging to the penetrans group is strongly reduced, with only two pairs of hair-like lateral plantar bristles and no patch of minute plantar setae segment bristles (Fig. 6A). In contrast, the chaetotaxy is only slightly reduced in the species of the caecata group, which exhibit three or four pairs of stiff subspiniform lateral plantar bristles and a patch of minute plantar bristles (Figs. 6B–F). Unfortunately, these articles are often missing even when studying a female that is recently embedded.
 |
Figure 5. The metacoxae of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. monositus; (H) T. trimamillata; (I) T. bossii; (J) T. bonneti; (K) T. hexalobulata. |
 |
Figure 6. The metatarsi of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. callida; (D) T. libis; (E) T. monositus; (F) T. bonneti. |
Abdomen
Externally, the most striking distinguishing feature between the penetrans and caecata groups is the variation in the size of the spiracles of the hypertrophied females. In the penetrans group, the spiracles of terga II–IV have disappeared, while in the caecata group, they are smaller than the others, though present [46]. The spermatheca presents a certain enigma. In the female neonate or a female not yet embedded, this structure is invisible upon first examination, as far as we know for the fleas that have a body or bulga, is most often sclerotized, and is a distal appendage, tail, or hilla that acts as a pump for sperm. In fleas that are not attached, a thorough examination will reveal, however, a transparent area called cribiform area, which is riddled with multiple orifices, at the base of the bulga. As in other genera, it is connected to the vagina through a duct, the ductus spermathecae, a visible though poorly defined structure. The spermatheca itself is diaphanous, and more or less conical in some species such as penetrans or trimamillata. In contrast, this structure is heavily sclerotized in neosomatic and fertilized females and has a consistent shape within a given species. Both Karsten [25] and Bonnet [8] described the spermatheca perfectly in the penetrans group, although Bonnet was not able to identify its purpose. Bonnet also noted the presence of an occasional double spermatheca in T. penetrans, which was omitted by Beaucournu et al. [4] and is, thus far, unique to this family.
Figure 7 shows the spermathecae of 11 species. T. travassosi, T. bondari, T. terasma, T. callida, T. monositus, and T. bossii exhibit bulgae that are rounded and spherical, while in T. penetrans, T. caecata, T. libis, T. monositus, T. trimamillata, and T. hexalobulata they are elongated and ellipsoidal. Spermatecae with short hillae are seen in penetrans, trimamillata, and hexalobulata, while long hillae are found in caecata, travassosi, bondari, terasma, callida, libis, and bossii. On the other hand, the width of the hilla may be thin (penetrans, terasma, monositus, trimamillata, and hexalobulata) or thick (bondari, callida, and libis). The measurements of the bulga and hilla of the spermathecae are presented in Table 4.
 |
Figure 7. The spermathecae of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. travassosi; (D) T. bondari; (E) T. terasma; (F) T. callida; (G) T. libis; (H) T. monositus; (I) T. trimamillata; (J) T. bossii; (K) T. hexalobulata. |
Morphological and morphometric data of modified segments of Tunga species.
In males, segment IX (clasper) and the phallosome are the most striking features for differentiating species. Similar to other fleas, segment IX is divided into two processes, the basimere (also often called the immovable or fixed process, or even p1) and the telomere (also called the movable process, movable finger, or even p2) which are connected to another structure, the manubrium. Figure 8 shows the shape and chaetotaxy of segment IX for the eight species whose males are known. The ratios between the width of the basimere and telomere in its median portion and the length of basimere/manubrium, and the shape of the basal and dorsal part of the manubrium are observed in Table 4. The phallosome consists of proximal and distal arms articulated in the middle, just before the ductus ejaculatorius and is almost as long as the penis-plate.
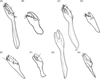 |
Figure 8. Segment IX of the males of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. terasma; (D) T. callida; (E) T. libis; (F) T. monositus; (G) T. trimamillata; (H) T. bonneti. |
The size ratio and the angle between the two arms are differential features for the species, as seen in Figure 9. In T. penetrans and T. monositus, the two arms are angled at approximately 90°, whereas in T. callida and T. caecigena, they are connected in a nearly straight line. Table 4 exhibits some morphologic features and morphometric data of the modified segments of the male and the spermathecae of the females.
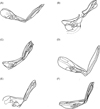 |
Figure 9. Male phallosomes of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. callida; (D) T. monositus; (E) T. trimamillata; (F) T. bonneti. |
Taxonomy
The principal synonymies, sexes, individual variations, undescribed species, subgenera, higher taxa and a key for adult species and neosomes are included and discussed below.
Synonymies
It is interesting to note that the older the description of the species, the greater the number of existing synonymies. Thus, T. penetrans presents seven major synonyms followed by T. caecata with two of them. All the synonymies for the genus were already cited by Beaucournu et al. [5].
Sexes
As shown in Table 1, the species caecata, travassosi, bondari, bossii, and hexalobulata are known only through their hypertrophied females. T. penetrans, T. callida, T. monositus, T. trimamillata, and T. bonneti are species in which the holotype and the allotype were described simultaneously. In caecigena and libis, the allotype males were described 37 (1958) and 6 (1968) years, respectively, after the holotype females. The males of T. terasma were described incorrectly by Fonseca [17] as males of T. travassosi. Interestingly, the holotype female of T. terasma was described the following year by Jordan. It is possible that this is the only case in the entire order of Siphonaptera in which the allotype was known before the holotype.
Individual variations
Regardless of sexual dimorphism (length, size of spiracles, chaetotaxy, maxillary palps), most of the individual variations are found in the modified segments, especially among the males. Hopkins and Rothschild [20] illustrate variations in the manubrium of T. penetrans and T. terasma. The illustrations of the male of T. caecigena presented by Chen and Ku [9] also show variations in the shape of the manubrium, basimere, and telomere. The figures of Wang [51] stress variations in the form of denticles on the distal portion of the basimere of T. caecigena. Similarly, when describing T. monositus, Barnes and Radovsky [2] illustrated and drew attention to the fact that the manubrium presents a highly variable shape, even on two sides of the same individual, with the proximal portion ranging from broad and blunt to slender, curved, and acuminate.
Undescribed species
-
As reported in Linardi and Guimarães [34], hypertrophied females of the caecata group were observed by Linardi and Botelho [31] to parasitize Oryzomys nigripes (currently Oligoryzomys nigripes) and Nectomys squamipes from Caratinga, Minas Gerais State, Brazil, with the neosomes located near the base of the dorsal surface of the ears. The neosomes of O. nigripes are spherical and 8–9 mm in diameter, in contrast to those observed in N. squamipes that present three anterior humps and measure 8 mm in diameter (Figs. 1A, B). For these authors, the neosomes were two undescribed new species, despite the form found on O. nigripes having been confused with T. monositus. At that time, T. trimamillata, with its three anterior humps in the neosome had not yet been described – that was to happen in 2002.
-
Beaucournu et al. [5] commented that T. bossii had been seen, but not described, in the province of Minas Gerais, by Reinhardt in 1853 (teste Burmeister, 1854, quoted and commented by Smit [46], who wrote “Mesomys spinosus (taxon not known to us) suffers from sand-fleas which preferably burrow near the anus and genitalia, at the base of the tail. This is very reminiscent of the habit of the recently described Chinese Tunga callida and I wonder whether indeed similar species do occur in Brazil”. In the same paragraph, Burmeister informs us that Dr. Reinhardt “Zeigte mir in Lagoa Santa (Minas Gerais) eine Hausmaus (= Mus musculus), die 13 solcher Flöhe an einem und 12 am anderen Ohr hatte” (He showed me a house mouse (Mus musculus) in Lagoa Santa (Minas Gerais, Brazil) that had 13 of these fleas on one ear and 12 on the other). Burmeister and Reinhardt apparently caught sight of the two Tunga parasites of rodents in Brazil, caecata and bossii!
-
When defining neosomy, Audy et al. [1] included the figure of a neosome of an undescribed species that is morphologically similar to the neosome of T. bonneti. Subsequently, Frank Radovsky, one of the authors of this work (in litt., November 13, 1979 addressed to P.M. Linardi), reported that “Incidentally, I have preserved material of Tunga in Phyllotis from Peru which I hope to describe. There are 2 undescribed species, one on the ear pinna as in T. monositus and your Tunga and one in the tail!” However, these two species were ultimately not described by Radovsky. Given the morphological similarities, the hosts, and geographical distribution, it is possible that these two species were, in reality, T. libis and T. bonneti!
-
A new species of Tunga and belonging to the “penetrans group” was found in Argentina [16]. Subsequently Marcela Lareschi (e-mail, May 30, 2014 addressed to P.M. Linardi) reported that this species is now being described and that it forms a discoid neosome in the carapace of Zaedyus pichiy perforating the osteoderms.
Subgenera
When Smit [46] divided the genus Tunga into two groups of species, he considered morphological characteristics such as the dorsal fusion of the pronotum and the mesonotum, the chaetotaxy of the fifth tarsal segment and the presence or the size of spiracular fossae on terga II–IV of the females, in addition to the parasitized host groups. At that time, only eight species were known. Four were included in the penetrans group (T. penetrans, T. travassosi, T. bondari, and T. terasma) and four others in the caecata group (T. caecata, T. caecigena, T. callida, and T. libis). Later, another proposal by Wang [51], based solely on geographical distribution, included the two known Chinese species, caecigena and callida, in a distinct subgenus (Brevidigita). When presenting the supraspecific classification for the genus Tunga, Lewis [28] accepted Wang’s proposal, but seems to have taken into consideration only those Tunga species that parasitize commensal rats for inclusion in Brevidigita because only T. caecata was added, though two other species, T. monositus and T. libis, were already known and were improperly left in the subgenus Tunga. Currently, with 13 known species, we consider the genus divided into two subgenera, Tunga and Brevidigita, though this division may be debatable:
-
Subgenus Tunga: T. (T.) penetrans, T. (T.) travassosi, T. (T.) bondari, T. (T.) terasma, T. (T.) trimamillata, and T. (T.) hexalobulata.
-
Subgenus Brevidigita: T. (B.) caecata, T. (B.) caecigena, T. (B.) callida, T. (B.) libis, T. (B.) monositus, T. (B.) bossii, and T. (B.) bonneti.
Higher taxa
The vast majority of the Siphonaptera show a certain consistency for certain wildcard characteristics; for example, the profile of the cephalic capsule, development of the eye (unless a host to a different ecology), the proportions of the various articles of the palp, or the form of the spermatheca. However, in the genus Tunga, these rules are not respected. As has been done in the genus Pulex (Pulex, Juxtapulex…), further divisions may be required when the puzzle of taxa known as Tunga is complemented by a comparative study of all males.
Beaucournu (in Aberlenc) [3]) recognizes Siphonaptera as
“insect holometabolous, wingless larvae apoda detritiphages but with mandatory addition of blood in their diet in adulthood latero-lateral, flattened to hind legs longer than the 2 prior to biting mouthparts (hematophagous regime), size ranging, fasting and/or before fertilization, from 0.75 to 8 mm. Now, we are also including “bristles directed backward and brownish colour”.
- During copulation, the male is always under the female. The phallosome has no middle joint, except in Hectopsylla. The female is vagile, more rarely sessile, and never cystic in the dermis of the host (except in Neotunga euloidea [Pulicidae] in which the male is not known); the female’s spermatheca is completely visible before any meal or copulation; the male’s abdomen may become neosomic, but it will always remain outside the host (except in N. euloidea); the abdominal spiracles do not change in shape, size or location during the life of the imago even in the case of neosomy… Pulicoidea, Vermipsylloidea, Malacopsylloidea, Ceratophylloidea and Hystrichopsylloidea
- During copulation, the male is always on the female. The phallosome presents a middle joint. The female is vagile only while searching for a host, and upon finding one it becomes completely encapsulated; the female’s spermatheca is visible in its entirety, only when encapsulated (bulga and hilla); female neosomy is very important and compulsory; abdominal stigma in the neosomes, are very enlarged and displaced and form on the terminal tergites… Tungoidea
For this author, the Tungoidea comprises only the genus Tunga. Hectopsylla remains in the Pulicoidea. Linardi asserts (as seen in Linardi and Guimarães [34]) that Hectopsylla must be retained in Tungidae, as evidenced by DNA analysis [52].
Key for adults and neosomes
The first dichotomous key for this genus was that of Hopkins and Rothschild [20] and did not include six species that were subsequently described (callida, libis, monositus, trimamillata, bossii, bonneti, and hexalobulata). While it is no longer usable, it proposed to differentiate Tungidae, but using characteristics that have proved worthless, for example, the indentation of the eye. This same key, excluding the two Chinese Tunga, was later reproduced by Johnson [21] to identify South American sand fleas. Barnes and Radovsky [2], when describing T. monositus, presented a key exclusively for the caecata group species known at that time.
Recently, De Avelar et al. [13], when describing Tunga bossii, proposed a new key, including all known taxa (both bonneti and hexalobulata are absent since their descriptions appeared after bossii) in which the appearance of the neosome is widely used. Although often the morphological aspects of many taxa that we would like to use in classification are not known (the autotomy of legs is mainly in females), we attempt to provide a more classical vision of differentiation, with emphasis on independent characteristics of neosomes. Thus, this new key also includes the means to differentiate known male characteristics.
-
Pronotum and mesonotum fused dorsally. Fifth tarsomere of the metatarsus with chaetotaxy reduced and exhibiting only two pairs of lateral plantar bristles (Fig. 6A). Males have a manubrium approximately two times longer than the basimere and exhibit a small constriction between them (Figs. 8F–H) (Subgenus Tunga ) ..............................................................................2
-
Pronotum and mesonotum not completely fused dorsally. Fifth tarsomere of the metatarsus with chaetotaxy not reduced and exhibiting more than three pairs of lateral plantar bristles (Figs. 6B–F). Males have a manubrium a little longer or as long as the basimere and without constriction between them (Figs. 8A–E) (Subgenus Brevidigita )........................................................................7
-
Eye without black pigment (Fig. 3D). Base of maxilla with only a micro-bristle. Maxillary palp as in Figure 4D. Metacoxa as in Figure 5D. Spermatheca as in Figure 7C. Globular neosome with head invaginated within abdomen, measuring (mm) 12–13 (length), 8 (width), and 10 (height). Male unknown...................................................... T. (T.) travassos i Pinto and Dreyfus.
-
Eye pigmented (Figs. A, E, F, J, M). Base of maxilla with two or more bristles. Neosome with head not invaginated within abdomen or laterally visible........................................................................3
-
Eye small, its greatest diameter only slightly exceeding half the distance from eye to dorsal margin of head (Fig. 3E). Dorsal genal lobe covering part of antennal segment III. Segment IV of maxillary palp longer than segment I (Fig. 4E). Metacoxa as in Figure 5E. Spermatheca with bulga wider than long (Fig. 7D). Mushroom-shaped neosome with a stem and conical posterior region and measuring (mm) 6 (length), 6 (width), and 5 (height). Males unknown ...................................................................................................................... T. (T.) bondari Wagner.
-
Eye large, its greatest diameter nearly equal to the distance from eye to dorsal margin of head. Dorsal genal lobe not covering the antennal segment III. Segment I of maxillary palp longer than segment IV. Spermatheca with a bulga longer than wide. Neosome globular or with prominent lobes.................................................................................................................................................4
-
Frontal tubercle slightly pronounced (Fig. 3F). Antennal segment II with only two bristles. Maxillary palp with few bristles (Fig. 4F). Metacoxa with proximal portion as wide as distal portion (Fig. 5F). Manubrium with ventral margin concave and dorsal margin convex (Fig. 8C). Spermatheca with globular bulga two times longer than hilla (Fig. 7D). Hypertrophied female not globular and containing four prominent lobes, measuring (mm) 10 (length), 9 (width), and 13 (height) and with cylindrical caudal disk, longer than wide................................................................................................................ T. (T.) terasma Jordan.
-
Frontal tubercle conspicuous (Figs. 3A, J, M). Antennal segment II with more than two bristles. Maxillary palp with numerous bristles (Figs. 3A, J, M). Metacoxa with proximal portion wider than distal portion (Figs. 5A, H, M). Manubrium with ventral margin straight and dorsal margin a little convex (Figs. 8A, G). Spermatheca with ellipsoidal bulga and more than three times longer than hilla. Female is globular and hypertrophied or exhibits three or six anterior lobes, with flattened or conical caudal disk, wider than long............................................................................5
-
Head with pronounced ventral genal lobe (Fig. 3A). Anterior base of maxilla with three thick bristles. Segment II of maxillary palp longer than segment I (Fig. 4A). Anteromedial surface of posterior tibia without bristles. Manubrium with proximal portion wide (Fig. 8A). Phallosome as Figure 9A. Bulga of spermatheca approximately 3.5 times longer than hilla (Fig. 7A). Globular hypertrophied female without projections or lobes and with flattened caudal disk, measuring (mm) 6 (length), 5 (width), and 5 (height).................. T. (T.) penetrans (L.).
-
Head with no evidence of ventral genal lobe (Figs. 3J, M). Anterior base of maxilla with 6–7 bristles. Segment I of maxillary palp longer than segment II (Figs. 4I, L). Bulga of spermatheca almost six times longer than hilla (Figs. 7I, K). Globular hypertrophied female with three or six anterior lobes and conical caudal disk .........................................................................................................6
-
Head with the postantennal region exhibiting 17–19 bristles (Fig. 3I). Antennal segment II with four bristles. Segment IV of maxillary palp almost as long as segment II (Fig. 4I). Metacoxa without a projection at the margin of the proximal portion (Fig. 5H). Spermatheca as in Figure 7I, with curved hila and bulga nine times wider than hilla. Anteromedial surface of posterior tibia with row of 9–12 bristles. Manubrium with proximal portion tapering and facing up (Fig. 8G). Phallosome as Figure 9E. Neosome with three lobes located anteriorly, measuring (mm) 12 (length), 5 (width), and 5 (height)............................................................................................................... T. (T.) trimamillata Pampiglione, Trentini, Fioravanti, Onore, and Rivasi.
-
Head with the postantennal region exhibiting nine bristles (Fig. 3M). Antennal segment II with three bristles. Metacoxa with a projection at the margin of the proximal portion (Fig. 5K). Spermatheca as Figure 7K, with straight hilla and bulga six times wider than hilla. Males unknown. Neosome with six lobes located anteriorly, measuring (mm) 4 (length), 4 (width), and 4 (height).................................................T. (T.) hexalobulata De Avelar, Facury Filho, and Linardi.
-
Eye absent. Males with the basal and apical tubes of the phallosome articulated almost in a straight line (Figs. 9B–C).................................................................................................................8
-
Eye present. Males with the basal and apical tubes of the phallosome forming a right angle (Figs. 9D, F)...............................................................................................................................................9
-
Occipital region with 8–10 bristles (Fig. 3C). Preoral internal sclerotization with anterior arm extending to half the distance between frontal tubercle and base of maxillary palp. Lacinia as long as maxillary palp. Manubrium almost as long as basimere (Fig. 8B). Phallosome as in Figure 9B. Elliptical neosome, with dorsal and ventral portions of similar dilatation, measuring (mm) 7–10 (length), 5 (width), and 6 (height) and caudal disk longer than wide................................................................................... T. (B.) caecigena Jordan and Rothschild.
-
Occipital region with 3–5 bristles (Fig. 3G). Preoral internal sclerotization with anterior arm extending near the base of maxillary palp. Lacinia longer than maxillary palp. Manubrium slightly longer than basimere (Fig. 8D). Phallosome as in Figure 9C. Spherical neosome, with the dorsal portion more swelled than the ventral portion, measuring (mm) 4.5 (length), 4.5 (width), and 4.5 (height) and caudal disk as long as wide....................... T. (B.) callida Li and Chin.
-
Eye pigmented. Head with the frontal curvature almost straight (Figs. 3I, L). Manubrium with the proximal portion tapering, acuminate (Figs. 8E, H). Elliptical gravid female........................10
-
Eye without black pigment. Head with the frontal curvature gently convex (Figs. 3B, I, K). Manubrium with the proximal portion truncate. Globular or bell-shaped gravid female..............11
-
Fifth metatarsomere with numerous minute plantar bristles (Fig. 6D). Segment II of the maxillary palp longer than segment III. Telomere as wide as basimere (Fig. 8E). Vertically elliptical neosome, higher than long and without lobes........................................... T. (B.) libis Smit.
-
Fifth metatarsomere of the metatarsus with few minute plantar bristles (Fig. 6F). Segment II of the maxillary palp as long as segment III (Fig. 4K). Telomere wider than basimere (Fig. 8H). Horizontally elliptical neosome, longer than high, rugby-ball shaped and without lobes..................................................................... T. (B.) bonneti Beaucournu and González-Acuña.
-
Posterodorsal lobe of proepimeron large, strongly projecting. Base of maxilla with no bristles. Metacoxa with basal portion much wider than apical and with anterior basal corners dilated (Fig. 5G). Spermatheca with bulga four times longer than hilla (Fig. 7H). Males with segment IX as in Figure 8F and phallosome as in Figure 9D. Bell-shaped gravid female with eight lobes, arranged as four large outer lobes and four small inner lobes...................................................................................................................... T. (B.) monositus Barnes and Radovsky.
-
Posterodorsal lobe of proepimeron small, rounded. Base of maxilla with one or two bristles. Metacoxa with basal portion a little wider than apical and without dilatation in the anterior basal corners (Figs. 5B, I). Spermatheca with bulga that is two times longer than hilla (Figs. 7B, J). Males unknown. Globular gravid female without lobes................................................................12
-
Preoral internal sclerotization with posterior arm more than eight times the length of anterior arm (Fig. 3B). Frontal tubercle slightly pronounced. Eye with the greatest diameter less than half the length of the antennal segment II. Base of maxilla with one bristle. Segment IV of the maxillary palp less than the length of segments II + III (Fig. 4B). Metacoxa as in Figure 5B. Spermatheca with bulga longer than wide and hilla two times longer than wide (Fig. 7B). Neosome, measuring (mm) 7 (length), 6 (width), and 6 (height) and conical caudal disk, almost as wide as long......................................................................................................... T. (B.) caecata (Enderlein).
-
Preoral internal sclerotization with posterior arm less than four times the length of anterior arm (Fig. 3K). Conspicuous frontal tubercle. Eye with the greatest diameter almost as long as antennal segment II. Base of maxilla has two bristles. Segments II, III, and IV of the maxillary palp undivided, but apparently IV greater than II + III (Fig. 4J). Metacoxa as in Figure 5I. Spermatheca with bulga wider than long and hilla about four times longer than wide (Fig. 7J). Neosome, measuring (mm) 9 (length), 8 (width), and 7 (height) and flattened caudal disk, wider than long...................................................... T. (B.) bossii De Avelar, Linhares, and Linardi.
Final remarks
The geographical center of the distribution of the genus Tunga is concentrated in South America, including 77% of the species that most likely originated there. Because approximately 23% of the species were described in the last 2 years from Ecuador, Brazil, and Chile, the biomes and regions contained in these countries offer great opportunities for new findings. There are at least three or four undescribed species, with one of them awaiting description and another two or three remaining confused with species that are already known.
Thirty-nine percent of the species are known only by the characteristics of the embedded females (neosomes). Only two out of the 13 described species of sand fleas have known larvae, although several larvae of the 1st instar of an undescribed species were obtained in the laboratory by Linardi and Botelho [34] from hypertrophied females infesting Brazilian wild rodents. Consequently, investigation of the alternate sexes and immature forms provides promising lines of research in the respective regions of their occurrences.
Because larval development occurs in fine-grained soils and the sand fleas are univoltine, the best results can be obtained in the dry-cool season.
Given the epidemiological and economic importance of T. penetrans and T. trimamillata, which infest both domestic animals and humans, the correct identification of species is indispensable. T. penetrans has been found parasitizing at least 28 genera of hosts [10], although some occurrences are incorrect records [32, 33]. Morphological variations must be used carefully for taxonomic purposes. Molecular biology should also be used for such purposes. As showed by De Avelar and Linardi [12] the Multiple Displacement Amplification technique (MDA) may be a valuable tool for molecular studies involving samples of sand fleas that are preserved in scientific collections.
Rodents and edentates are the main hosts of Siphonaptera, housing approximately 85% of the known species.
Data on host specificity and geographical distribution may support the identification of Tunga species because some sand fleas and their hosts may have co-evolved. According to Traub [48], most fleas have clearly evolved with their hosts and primitive hosts tend to have primitive fleas [47]. Mammals such as Edentata might have been the primitive hosts of tungids because they are devoid of incisor teeth and nails to remove the neosomes attached on toes and on the ventral abdominal, regions regularly in contact with the soil and of great difficulty for the hosts to dislodge the parasites by grooming or eating. On the other hand, based on the molecular phylogeny, Whiting et al. [52] placed Tunga at the base of flea phylogeny and its association with basal mammal hosts suggests that the origin and diversification of Siphonaptera coincided with basal mammal diversification. Sloths (Pilosa) and armadillos (Cingulata) belong to an ancient stock of mammalians and constitute the majority of the natural mammalian hosts of the genus Tunga [52].
Conflict of Interest
No conflict of interest.
Acknowledgments
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/Brasil) for the research fellowship granted to P.M.L. Some data from this research constituted part of the PhD thesis of D.M.A in the Parasitology/Post-graduate program in Parasitology/Instituto de Ciências Biológicas/Universidade Federal de Minas Gerais/UFMG, Brazil.
References
- Audy JR, Radovsky FJ, Vercammen-Grandjean PH. 1972. Neosomy: radical intrastadial metamorphosis associated with arthropod symbioses. Journal of Medical Entomology, 9, 487–494. [PubMed] [Google Scholar]
- Barnes AM, Radovsky FJ. 1969. A new Tunga (Siphonaptera) from the nearctic region with description of all stages. Journal of Medical Entomology, 6, 19–36. [CrossRef] [PubMed] [Google Scholar]
- Beaucournu J-C. 2015. Ordre Siphonaptera, in Les Insectes du Monde. Biodiversité. Classification. Clés de détermination des familles, Aberlenc H-P, Editor. Quae, Opie, Biotope: Montpellier & Mèze [Google Scholar]
- Beaucournu J-C, Launay H, Sklair A. 1988. Abnormalities in the spermatheca and genital ducts in Siphonaptera (Insecta) – Bibliographic review and personal cases. Annales de Parasitologie Humaine et Comparée, 63, 64–75. [Google Scholar]
- Beaucournu J-C, Degeilh B, Mergey T, Muñoz-Leal S, González-Acuña D. 2012. Le genre Tunga Jarocki, 1838 (Siphonaptera: Tungidae). I – Taxonomie, phylogénie, écologie, role pathogéne. Parasite, 19, 297–308. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Beaucournu J-C, Mergey T, Muñoz-Leal S, González-Acuña D. 2012. Description de Tunga bonneti n. sp. du Chili (Siphonaptera: Tungidae) et notes sur sa spécificité, sa chorologie, son dermecos et sa phénologie. Parasite, 19, 207–216. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Bell A, Neely CL, Peeples J. 1979. Tungiasis in Tennessee. Southern Medical Journal, 72, 141–143. [CrossRef] [PubMed] [Google Scholar]
- Bonnet G. 1867. Mémoire sur la puce penetrante ou chique (Pulex penetrans L.). Paris: J.B. Baillière & Fils. [Google Scholar]
- Chen CS, Ku HD. 1958. The discovery of a male specimen of the sandflea, Tunga caecigena Jordan & Rothschild, 1921 with a morphological description [in Chinese]. Acta Entomologica Sinica, 8, 179–184. [Google Scholar]
- De Avelar DM. 2010. Sistemática e análise cladística das espécies neotropicais do gênero Tunga Jarocki, 1838 (Siphonaptera, Tungidae). Tese de Doutoramento. Belo Horizonte, Brasil: Universidade Federal de Minas Gerais. [Google Scholar]
- De Avelar DM, Facury Filho EJ, Linardi PM. 2013. A new species of Tunga (Siphonaptera: Tungidae) parasiting cattle from Brazil. Journal of Medical Entomology, 50, 679–684. [CrossRef] [PubMed] [Google Scholar]
- De Avelar DM, Linardi PM. 2010. Use of multiple displacement amplification as pre-polymerase chain reaction (Pre-PCR) to amplify genomic DNA of siphonapterids preserved for long periods in scientific collections. Parasites & Vectors, 3, 86–91. [CrossRef] [PubMed] [Google Scholar]
- De Avelar DM, Linhares AX, Linardi PM. 2012. A new species of Tunga (Siphonaptera: Tungidae) from Brazil with a key to the adult species and neosomes. Journal of Medical Entomology, 49, 23–28. [CrossRef] [PubMed] [Google Scholar]
- Eisele MJ, Heukelbach J, Van Mardk E, Melhorn H, Meckes O, Franck S, Feldmeier H. 2003. Investigations on the biology, epidemiology, pathology and control of Tunga penetrans in Brazil. I. Natural history of tungiasis in man. Parasitology Research, 90, 87–99. [PubMed] [Google Scholar]
- Enderlein G. 1901. Zur Kenntnis der Flöhe und Sandflöhe. Neue und wenig bekannte Pulliciden und Sarcopsyllidae. Zoologische Jabrbücher, 14, 549–557. [Google Scholar]
- Ezquiaga MC, Lareschi M, Avelar DM, Linardi PM. 2011. Distribución geográfica y hospedatoria de la asociación entre dasipódidos (Xenarthra: Dasypodidae) y species del género Tunga Jarocki (Siphonaptera: Tungidae) en la Argentina, in Resumos, XXIV Jornadas Argentinas de Mastozoología, 8–11 November, La Plata. Argentina: Sociedade Argentina para el Estudio de los Mamíferos. p. 92. [Google Scholar]
- Fonseca FOR. 1936. Sobre o macho de Tunga travassosi Pinto et Dreyfus, 1927, e o parasitismo de Euphractes sexcinctus L. por Tunga penetrans (L., 1758). Revista de Entomologia Rio de Janeiro, 6, 421–424. [Google Scholar]
- Geigy R, Herbig A. 1949. Die hypertrophie der Organe beim Weibchen von Tunga penetrans. Acta Tropica, 6, 246–262. [PubMed] [Google Scholar]
- Hastriter MW. 1997. Establishment of the tungid flea, Tunga monositus (Siphonaptera: Pulicidae), in the United States. Great Basin Naturalist, 57, 281–282. [Google Scholar]
- Hopkins GHE, Rothschild M. 1953. An illustrated catalogue of the Rothschild collection of fleas (Siphonaptera) in the British Museum (Natural History). Vol. 1. Tungidae and Pulicidae, London, United Kingdom: British Museum (Natural History). [Google Scholar]
- Johnson PT. 1957. A classification of Siphonaptera of South America with descriptions of new species. Entomological Society of Washington: Washington. [Google Scholar]
- Jordan K. 1937. Two new fleas from South America (Siphonaptera). Novitates Zoologicae, 40, 307–310. [Google Scholar]
- Jordan K. 1962. Notes on Tunga caecigena (Siphonaptera: Tungidae). Bulletin of the British Museum (Natural History) Entomology, 12, 353–364. [Google Scholar]
- Jordan K, Rothschild NC. 1921. A new species of Sarcopsyllidae. Ectoparasites, 1, 131–132. [Google Scholar]
- Karsten H. 1865. XXXIII. Contribution towards the knowledge of the Rhynchoprion penetrans. Annals and Magazine of Natural History, 15, 293–312. [Google Scholar]
- Lavoipierre MMJ, Radovsky F, Budwiser PD. 1979. The feeding process of a tungid flea, Tunga monositus (Siphonaptera: Tungidae), and its relationship to the host inflammatory and repair response. Journal of Medical Entomology, 15, 187–217. [Google Scholar]
- Lewis RE. 1998. Résumé of the Siphonaptera (Insecta) of the world. Journal of Medical Entomology, 35, 377–389. [PubMed] [Google Scholar]
- Lewis RE. 2009. Siphonaptera. Part I – Supraspecific classification. Part II – Alphabetical genus and species list. Part III – Alphabetical species/subspecies list, 16th ed. Published by the author. [Google Scholar]
- Li KC, Chin TH. 1957. Tunga callida sp. nov., a new species of sand-flea from Yunnan. Acta Entomologica Sinica, 7, 113–120. [Google Scholar]
- Lima AC, Hathaway CR. 1943. Pulgas. Bibliografia, catálogo e hospedadores. Rio de Janeiro, Brasil: Instituto Oswaldo Cruz. [Google Scholar]
- Linardi PM, Botelho JR. 1985. Algumas observações sobre Tunga do grupo “caecata” parasitando roedores silvestres, in Resumos, IX Congresso da Sociedade Brasileira de Parasitologia, 30 July-02 August, Fortaleza, CE. Fortaleza, CE: Sociedade Brasileira de Parasitologia. p. 162. [Google Scholar]
- Linardi PM, De Avelar DM. 2014. Neosomes of tungid fleas on wild and domestic animals. Parasitology Research, 113, 3517–3533. [CrossRef] [PubMed] [Google Scholar]
- Linardi PM, De Avelar DM, Facury Filho EJ. 2013. Establishment of Tunga trimamillata (Siphonaptera: Tungidae) in Brazil. Parasitology Research, 112, 3239–3242. [CrossRef] [PubMed] [Google Scholar]
- Linardi PM, Guimarães LR. 2000. Sifonápteros do Brasil. Museu de Zoologia USP/FAPESP: São Paulo, Brasil. [Google Scholar]
- Linné CVon. 1758. Systema Naturae. Stockholmiae. [Google Scholar]
- Liu Z-Y, Li K-C, Wu H-Y, Li G-Z. 1986. Fauna Sinica Insecta Siphonaptera. Science Press: Beijing, China. [Google Scholar]
- Meira JA. 1934. Contribuição parasitológica para a epidemiologia da peste bubônica na cidade de São Paulo. Sobre as pulgas de rato da mesma cidade. Annaes Pauista de Medicina e Cirurgia, 28, 143–193. [Google Scholar]
- Pampiglione SM, Fioravanti ML, Gustinelli A, Onore G, Mantovani B, Luchetti A, Trentini M. 2009. Sand flea (Tunga spp.) infections in humans and domestic animals: state or the art. Medical and Veterinary Entomology, 23, 172–186. [CrossRef] [PubMed] [Google Scholar]
- Pampiglione S, Trentini M, Fioravanti ML, Gustinelli A. 2004. Differential diagnosis between Tunga penetrans (L., 1758) and T. trimamillata Pampiglione et al., 2002 (Insecta, Siphonaptera), the two species of the genus Tunga parasitic in man. Parasite, 11, 51–57. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Pampiglione S, Trentini M, Fioravanti ML, Onore G, Rivasi F. 2000. A new species of Tunga (Insecta, Siphonaptera) in Ecuador. Parassitologia, 44 Suppl. 1, 127. [Google Scholar]
- Pampiglione S, Trentini M, Fioravanti ML, Onore G, Rivasi F. 2003. Additional description of a new species of Tunga (Siphonaptera) from Ecuador. Parasite, 10, 9–15. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Pilgrim RLC. 1993. An instance of tungiasis in New Zealand. New Zealand Medical Journal, 106, 180. [Google Scholar]
- Pinto C, Dreyfus A. 1927. Tunga travassosi n. sp. parasite de Tatusia novemcinctus do Brasil. Boletim Biológico São Paulo, 9, 174–178. [Google Scholar]
- Rothschild M. 1992. Neosomy in fleas, and the sessile life-style. Journal of Zoology, 226, 613–629. [CrossRef] [Google Scholar]
- Sanusi ID, Brown EB, Shepard TG, Grafton WD. 1989. Tungiasis: report of one case and review of the 14 reported cases in the United States. Journal of the American Academy of Dermatology, 20, 941–944. [CrossRef] [PubMed] [Google Scholar]
- Smit FGAM. 1962. A new sand-flea from Ecuador. Entomologist, 95, 89–93. [Google Scholar]
- Traub R. 1980. The zoogeography and evolution of some fleas, lice and mammals, in Fleas. Starcke H, Traub R, Editors. A. A. Balkema: Roterdam. p. 93–172. [Google Scholar]
- Traub R. 1985. Coevolution of fleas and mammals, in Coevolution of parasitic arthropods and mammals. Kim KC, Editor. Wiley: New York. p. 295–437. [Google Scholar]
- Veraldi S, Camozzi S, Scarabelli G. 1996. Tungiasis presenting with sterile pustular lesions on the hand. Acta Dermato-venereologica, 76, 495. [PubMed] [Google Scholar]
- Wagner J. 1932. Tunga bondari eine neue Art der Sandflöhe. Novitates Zoologicae, 38, 248–249. [Google Scholar]
- Wang DC. 1976. The Chinese Tunga (Siphonaptera: Tungidae). Acta Entomologica Sinica, 19, 117–118. [Google Scholar]
- Whiting MF, Whiting AS, Hastriter MW, Ditmar KA. 2008. A molecular phylogeny of fleas (Insecta: Siphonaptera): origins and host associations. Cladistics, 24, 677–707. [Google Scholar]
- Witt LH, Linardi PM, Meckes O, Schwalfenberg S, Ribeiro RA, Feldmeier H, Heukelbach J. 2004. Blood-feeding of Tunga penetrans males. Medical and Veterinary Entomology, 18, 439–441. [CrossRef] [PubMed] [Google Scholar]
Cite this article as: Linardi PM, Beaucournu J-C, de Avelar DM & Belaz S: Notes on the genus Tunga (Siphonaptera: Tungidae) II – neosomes, morphology, classification, and other taxonomic notes. Parasite, 2014, 21, 68.
All Tables
Tunga species: Geographic distribution, sexes, stages, neosomes, and sites of attachment to their principal true hosts.
All Figures
 |
Figure 1. A neosome of the caecata group of species: (A) embedded on the ear of Nectomys squamipes (×3); (B) frontal view after extraction (×12). |
| In the text | |
 |
Figure 2. A neosome of Tunga penetrans – posterior view. Scale bar = 2 mm. |
| In the text | |
 |
Figure 3. The heads of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. callida; (H) T. libis; (I) T. monositus; (J) T. trimamillata; (K) T. bossii; (L) T. bonneti; (M) T. hexalobulata. |
| In the text | |
 |
Figure 4. The maxillary palps of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. callida; (H) T. monositus; (I) T. trimamillata; (J) T. bossii; (K) T. bonneti; (L) T. hexalobulata. |
| In the text | |
 |
Figure 5. The metacoxae of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. caecigena; (D) T. travassosi; (E) T. bondari; (F) T. terasma; (G) T. monositus; (H) T. trimamillata; (I) T. bossii; (J) T. bonneti; (K) T. hexalobulata. |
| In the text | |
 |
Figure 6. The metatarsi of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. callida; (D) T. libis; (E) T. monositus; (F) T. bonneti. |
| In the text | |
 |
Figure 7. The spermathecae of some species of Tunga: (A) T. penetrans; (B) T. caecata; (C) T. travassosi; (D) T. bondari; (E) T. terasma; (F) T. callida; (G) T. libis; (H) T. monositus; (I) T. trimamillata; (J) T. bossii; (K) T. hexalobulata. |
| In the text | |
 |
Figure 8. Segment IX of the males of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. terasma; (D) T. callida; (E) T. libis; (F) T. monositus; (G) T. trimamillata; (H) T. bonneti. |
| In the text | |
 |
Figure 9. Male phallosomes of some species of Tunga: (A) T. penetrans; (B) T. caecigena; (C) T. callida; (D) T. monositus; (E) T. trimamillata; (F) T. bonneti. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


