| Issue |
Parasite
Volume 33, 2026
|
|
|---|---|---|
| Article Number | 6 | |
| Number of page(s) | 16 | |
| DOI | https://doi.org/10.1051/parasite/2026005 | |
| Published online | 10 February 2026 | |
Research Article
Diversity and abundance of Culicoides on goat and cattle farms in the southern part of the Republic of Korea
Diversité et abondance des Culicoides dans les élevages caprins et bovins du sud de la République de Corée
1
School of Life Sciences, BK21 FOUR KNU Creative BioResearch Group, Kyungpook National University, Daegu 41566, Republic of Korea
2
Department of Biology, College of Natural Sciences, Kyungpook National University, Daegu 41566, Republic of Korea
3
Vector-Borne Disease Laboratory, Foreign Animal Disease Division, Animal and Plant Quarantine Agency, Gimcheon 39660, Republic of Korea
* Corresponding authors: This email address is being protected from spambots. You need JavaScript enabled to view it.
(Kwang Shik Choi); This email address is being protected from spambots. You need JavaScript enabled to view it.
(In-Soon Roh)
Received:
10
June
2025
Accepted:
9
January
2026
Abstract
Biting midges of the genus Culicoides Latreille (Ceratopogonidae) pose a significant threat to veterinary health as vectors of over 60 viruses, most of which affect livestock. In this study, we used light traps to sample Culicoides populations on cattle and goat farms from May to October 2023 at 15 sites in Gyeongsangnam-do, Jeollanam-do, and Jeju Island, South Korea. Diversity and abundance were analysed based on the collection date, environmental conditions, and host species. A total of 124,055 individuals were collected, comprising 14 previously recorded and two newly recorded species: C. asiana and C. palawanensis. The dominant species was C. arakawae, which accounted for 80.60% of the total collected individuals, followed by C. punctatus (10.25%), and C. tainanus (3.36%), while the remaining 13 species constituted 5.80% of the collection. Total Culicoides abundance peaked in August (40.15%), driven largely by fluctuations in C. arakawae abundance, but the seasonal abundances of individual species varied. Culicoides arakawae and C. punctatus were dominant on the mainland, while C. matsuzawai, C. lungchiensis, and C. tainanus were dominant on Jeju Island. The dominant species on cattle farms were C. arakawae and C. punctatus, while C. arakawae dominated in collections from goat farms. The detection of two new species records suggests that the fauna of South Korea is still incompletely understood.
Résumé
Les moucherons piqueurs du genre Culicoides Latreille (Ceratopogonidae) constituent une menace importante pour la santé animale, car ils sont vecteurs de plus de 60 virus, dont la plupart affectent le bétail. Dans cette étude, nous avons utilisé des pièges lumineux pour échantillonner les populations de Culicoides dans des élevages caprins et bovins, de mai à octobre 2023, sur 15 sites répartis dans les provinces de Gyeongsangnam-do, Jeollanam-do et sur l’île de Jeju. La diversité et l’abondance ont été analysées en fonction de la date de collecte, des conditions environnementales et de l’espèce hôte. Au total, 124 055 individus ont été collectés, appartenant à 14 espèces déjà répertoriées et à deux espèces nouvellement répertoriées : C. asiana et C. palawanensis. L’espèce dominante était C. arakawae, représentant 80,60 % du total des individus collectés, suivie de C. punctatus (10,25 %) et C. tainanus (3,36 %), tandis que les 13 autres espèces constituaient 5,80 % des collectes. L’abondance totale des Culicoides a atteint un pic en août (40,15 %), principalement en raison des fluctuations de l’abondance de C. arakawae, mais l’abondance saisonnière des différentes espèces variait. Les espèces C. arakawae et C. punctatus étaient dominantes sur le continent, tandis que C. matsuzawai, C. lungchiensis et C. tainanus l’étaient sur l’île de Jeju. Les espèces dominantes dans les élevages bovins étaient C. arakawae et C. punctatus, tandis que C. arakawae dominait dans les collections provenant des élevages caprins. La détection de deux espèces nouvellement répertoriées suggère que la faune de la Corée du Sud est encore mal connue.
Key words: Biting midges / Culicoides / Species diversity / Distribution / Unrecorded species
Edited by: Jérôme Depaquit
© S.B. An et al., published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The genus Culicoides Latreille is a group of small, haematophagous midges, typically measuring fewer than 3 mm in length. It belongs to the family Ceratopogonidae, along with three other haematophagous genera – Austroconops Wirth and Lee, Leptoconops (Skuse), and Forcipomyia Meigen – and many non-haematophagous genera [8]. Among these genera, Culicoides is the most extensively studied with 1,413 species reported, representing 33 subgenera and many species remaining unplaced into subgenera [11, 12].
Culicoides species feed on a wide range of vertebrates, with some serving as vectors for various viral, protozoan, and parasitic pathogens [43]. Species of Culicoides transmit over 60 viruses, including bluetongue virus (BTV), Schmallenberg virus (SBV), and African horse sickness virus (AHSV), which primarily affect the livestock industry and are considered significant in the veterinary field [16, 43, 44]. In the Republic of Korea (ROK), infection with the arboviruses Akabane virus (AKAV), Chuzan virus (CHUV), Aino virus (AINV), Bovine ephemeral fever virus (BEFV), and Ibaraki virus (IBAV) have been reported in either livestock or midges [2, 3, 31, 38, 48, 51]. Currently, 12 species of Culicoides associated with disease transmission are known to inhabit the ROK. Pathogens have been detected in Korean specimens of four species (Culicoides arakawae (Arakawa), C. oxystoma Kieffer, C. punctatus (Meigen), and C. tainanus Kieffer [47, 65]), while the remaining eight species (C. actoni Smith, C. chiopterus (Meigen), C. circumscriptus Kieffer, C. jacobsoni Macfie, C. lungchiensis Chen & Tsai, C. obsoletus (Meigen), C. sumatrae Macfie, and C. pulicaris (Linnaeus)) are associated with pathogen transmission in other countries [16, 19, 21, 43, 44, 45, 49, 64, 66].
Early studies on Culicoides diversity in the ROK were sporadic [1, 33, 56], until Cho and Chong [14] conducted the first comprehensive nationwide investigation. More recently, the diversity and abundance of Culicoides was investigated in Gyeonggi-do, Gyeongsang-do, and Jeolla-do, and on Jeju Island [26, 27, 28, 30, 65]. Basic morphological descriptions of 16 Culicoides species commonly found in the country were provided by Choi et al. [15], along with their distributions, while Bellis et al. [7] and Jeon et al. [23] each reported three previously unrecorded species; Kim et al. [28] and Lee et al. [35] each added one species. Currently, a total of 38 species are recorded in the National Species List of the ROK and cross-referenced with the species catalogued in Borkent and Dominiak [11] (Table 1). Additionally, C. pulicaris (Linnaeus), 1758 was suggested to be a misidentification of C. punctatus by Kim et al. [26] and Bellis et al. [7]. The list for the ROK appears to be low compared to that of its neighbour Japan, where 83 species have been reported [1, 34, 60]. Furthermore, previous Korean studies have predominantly concentrated on Gyeonggi-do, Gyeongsang-do, and Jeju Island, resulting in insufficient data for other regions.
List of Culicoides species reported in the ROK.
With the continuous progression of global warming, the ROK is expected to gradually shift towards a subtropical climate [17]. This is expected to increase the activity, affect the habitat, and influence the overwintering of disease vectors such as Culicoides [50]. Thus, this study aimed to survey the diversity and abundance of Culicoides in the southern part of the ROK, where the effects of climate change are expected to be the greatest [32].
Materials and methods
A total of 15 collection sites were selected in the southern regions of the ROK: five in Gyeongsangnam-do (three cattle sheds and two goat sheds), five in Jeollanam-do (three cattle sheds and two goat sheds), and five on Jeju Island (four cattle sheds and one goat shed) (Table 2, Fig. 1). Collections were made over a single night every two weeks from May to September, and once in October 2023. Collection dates on the mainland (Gyeongsangnam-do and Jeollanam-do) were the same for all sites, while collection dates for Jeju Island sites differed from those of the mainland by a few days. Blacklight traps equipped with removable fine mesh bags for the collection of Culicoides species, and a CO2 release agent as an auxiliary attractant (model: “ultra trap”, BT Global Co., Ltd., Seongnam, Republic of Korea) were used for collections. Traps were installed inside sheds near host species, and fixed over 1.5 m from land during collection periods. Traps were retrieved one day after installation and transferred to the laboratory. All collected samples were stored in a −70 °C freezer until species identification.
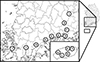 |
Figure 1 Collection sites for the study. A. Hapcheon, B. Sancheong, C. Jinju, D. Sacheon, E. Hadong, F. Gwangyang, G. Suncheon, H. Boseong, I. Gangjin, J. Haenam, K. Jeju1, L. Jeju2, M. Jeju3, N. Jeju4. O. Jeju5; Cow shed (A, B, C, F, I, J, K, L, N, O), Goat shed (D, E, G, H, M). |
Information on Culicoides species collection sites.
Species occurrences were sorted according to the collection region and date. The Statistical Package for the Social Sciences (SPSS) [53] was used to conduct Pearson and Spearman correlation analyses assessing the seasonal occurrence in relation to climate parameters, including temperature and precipitation, obtained from the Korea Meteorological Administration (https://data.kma.go.kr/cmmn/main.do) for the collection locations. Climate parameters used for analyses were based on a mean of the two-week period before each collection.
Preliminary identification was conducted using a stereoscopic microscope (SZ61, Olympus Corporation, Tokyo, Japan). Genus identification was based on morphological keys from Borkent [10], McAlpine et al. [41], and Swanson [52]. Species identification was based on several previous studies [1, 6, 7, 8, 15, 18, 34, 55, 56, 57, 60, 61, 67]. Before DNA extraction, non-destructive tissue digestion was performed. Specimens were digested at 55 °C for 2 h in 350 μL of DLD buffer, 3.5 μL of 2-mercaptoethanol, and 20 μL of proteinase (Invirustech, Gwangju, ROK). Following digestion, specimens were stored in 70% ethanol. Later, cadavers were temporarily placed onto glass slides with 70% ethanol to measure the head, wings, and abdomen using a camera (BUC5H-2000C, Beijing Bestscope Technology Co., Ltd., Beijing, PR China) connected to an optical microscope (CX43, Olympus). For the head, the contiguous or separated compound eyes, presence of pubescence between the ommatidia (facets), the shape of the third palpal segment, presence and shape of the sensory pit, palpal ratio (PR), distribution of sensilla coeloconica (SCo) on the flagellomeres of the antennae, antennal ratio (AR), and the length of the proboscis/head height (P/H) ratio were measured. The length and width of the wings were examined alongside the costal ratio (CR), and wing patterns were characterised. The position and presence of pale bands on the femora and tibiae, the number of hind tibial combs, and the order of the longest comb from the spur were measured for the legs. In the female genitalia, the number, shape, and size of the spermathecae were measured, and the presence and size of the sclerotised neck, rudimentary spermatheca, and sclerotised ring were measured. After completing the measurements, all samples were preserved at –20 °C in a freezer, in 70% ethanol.
To confirm the identification of unrecorded species, the mitochondrial cytochrome oxidase subunit 1 (COI) region was sequenced, and a phylogenetic analysis was performed. First, polymerase chain reactions (PCRs) were performed using the BC1culicFm and JerR2m primer pair [5]. The thermocycler protocol included an initial denaturation step at 94 °C for 2 min; 40 cycles of denaturation at 94 °C for 30 s, annealing at 48 °C for 30 s, and extension at 72 °C for 1 min; and a final 5-minute elongation step at 72 °C. Amplified PCR products were then submitted to Macrogen (Daejeon, ROK) for Sanger sequencing, obtaining bidirectional sequences using the primers M13F and M13R-pUC, the sequences of which are included in the sequences of BC1culicFm and JerR2m. The acquired sequences were aligned using BioEdit v.7.2.6.1 [22], registered in GenBank (accession numbers PQ643289–PQ643290 for C. asiana Bellis; PQ643291 and PV111011–PV111017 for C. palawanensis Delfinado), and then the sequences of each species were compared to those in GenBank using BLAST. Phylogenetic trees were produced using the neighbour-joining (NJ) method and pairwise distance analyses were performed for the unrecorded species using MEGA 11 [54]. Publicly available GenBank-registered sequences related to the two unrecorded species, C. asiana (MW496167, MW496170, MW496171, KJ162955, KJ162956, KT352310, KT352321, and KT352360) and C. palawanensis (KY441765, KY441793, KY441798, KY441807, KY441809, and ON002365) were compared to those of the collected samples, and Alluaudomyia quadripunctata (KT278187) was applied as an outgroup in both phylogenetic analyses.
Results
A total of 124,055 Culicoides samples comprising 14 known domestic species and two newly recorded species: C. asiana and C. palawanensis were collected (Table 3, Fig. 2). The predominant species was C. arakawae (80.60%, 99,985), followed by C. punctatus (10.25%, 12,712), C. tainanus (3.36%, 4,167), C. matsuzawai (2.79%, 3,465) and C. oxystoma (1.30%, 1,617). The remaining 11 species, 2,108 individuals collectively, accounted for 1.70% of the total collection.
 |
Figure 2 Wings of 16 collected Culicoides species including 2 newly recorded species (O and P): (A) C. arakawae, (B) C. homotomus, (C) C. jacobsoni, (D) C. japonicus, (E) C. kibunensis, (F) C. lungchiensis, (G) C. matsuzawai, (H) C. morisitai, (I) C. nipponensis, (J) C. oxystoma, (K) C. pictimargo, (L) C. punctatus, (M) C. sinanoensis, (N) C. tainanus, (O) C. asiana, (P) C. palawanensis. Scale bars = 0.1 mm. |
Seasonal distribution of 16 Culicoides species collected in southern Korea, 2023.
Regarding seasonal distribution, Culicoides individuals were most abundant in late August, when 25.29% (31,371) of the total collection was acquired, but this high proportion was mainly comprised of C. arakawae (30,445) (Table 3). Temporal distribution patterns differed among Culicoides species. The predominant species, C. arakawae was mainly collected during late July and early September, exhibiting its highest abundance in late August (30.45%, 30,445). Culicoides punctatus exhibited two peaks in early June (33.22%, 4,223) and late July (23.10%, 2,936), while C. tainanus was concentrated in late May (19.89%, 829), early June (21.31%, 888), and late July (26.35%, 1,098). Culicoides matsuzawai was mainly found in the late July (84.01%, 2,911) collection, and C. oxystoma was primarily collected in late July (26.59%, 430) and in both collection periods in September (26.41%, 427; 22.02%, 356).
Geographic occurrence patterns also differed among species. Culicoides arakawae was collected from 12 locations, excluding Jeju2, Jeju4, and Jeju5, and was more abundant in goat sheds than in cattle sheds (cattle:goat = 7,222:92,763) (Tables 4 and 5, Fig. 3). Individual collection sites in which C. arakawae represented high proportions included cow sheds in Hapcheon (80.62%, 4,501), Sancheong (36.07%, 154), Gangjin (55.65%, 1,004), and Haenam (81.98%, 1,078), and goat sheds in Sacheon (67.47%, 4,933), Hadong (97.31%, 17,734), Suncheon (88.49%, 23,179), and Boseong (99.70%, 46,881) (Table 4). In contrast, C. punctatus was collected from 14 sites, excluding only Jeju5, and was more abundant in cow sheds (cattle:goat = 8,286:4,426). Sites at which C. punctatus represented high proportions included cow sheds in Sancheong (59.02%, 252), Jinju (66.22%, 5,671), Gwangyang (57.68%, 1,761), and Jeju2 (21.36%, 135), and considerable numbers were collected in goat sheds in Sacheon (22.01%, 1,609) and Suncheon (9.39%, 2,460). Culicoides tainanus was collected at all sites, representing high proportions in cow sheds at Jinju (25.04%, 2,144), Jeju2 (48.89%, 309), and Jeju4 (19.77%, 34), C. matsuzawai was collected at seven sites but was dominant only at Jeju3 (87.56%, 2,901), and C. oxystoma was collected from seven locations but exhibited a notable proportion only at Gwangyang (29.84%, 911). Among other species collected at relatively high abundance at one or more sites, C. nipponensis Tokunaga was exclusively collected at Gangjin (42.24%, 762), and C. lungchiensis was dominant at Jeju1 (72.92%, 280), Jeju2 (23.42%, 148), Jeju4 (65.12%, 112), and Jeju5 (79.66%, 47). For the newly recorded species, C. asiana was collected at Jeju1 (32), Jeju2 (13), and Jeju4 (1), while C. palawanensis was exclusively collected in Haenam (82).
 |
Figure 3 Seasonal distribution of Culicoides species at collection sites with climate parameters. |
Total number (%*) of Culicoides species collected at 15 sites in southern Korea, 2023.
Comparison of Culicoides abundances by farm type and environment.
The species compositions of the Culicoides collected at each of the 15 collection sites was compared between environments (mainland vs island) and farm types (cattle vs goats) (Table 5). The ten collection sites located in Gyeongsangnam-do and Jeollanam-do were classified as mainland environments, while the five sites on Jeju Island were classified as island environments. In the mainland environment, a total of 119,495 individuals, representing 15 Culicoides species, were collected, whereas in the island environment, a total of 4,560 individuals from 7 Culicoides species were collected. This indicates comparatively lower species richness and diversity on the island. In mainland environments, the dominant species were C. arakawae (83.64%) and C. punctatus (10.49%). In contrast, the island collections exhibited markedly different compositions, with C. matsuzawai (64.36%), C. lungchiensis (14.93%), and C. tainanus (14.80%) being predominant.
To assess species compositions at the two farm types, the collections from the 10 cattle sheds and were compared to those from the five goat sheds. A total of 21,993 individuals, representing 15 Culicoides species, were collected from cattle sheds, while 102,062 individuals, comprising 12 Culicoides species were collected from goat sheds. In cattle sheds, C. punctatus (37.68%), C. arakawae (32.84%), and C. tainanus (12.34%) were collected in high proportions, while C. arakawae (90.89%) was dominant in goat sheds. Among the 16 collected species, 11 were identified at both types of farm, with four species (C. nipponensis, C. sinanoensis, C. asiana, and C. palawanensis) exclusively found in cattle sheds, and C. pictimargo restricted to goat sheds.
Correlation analyses were conducted for the five dominant species, C. arakawae, C. punctatus, C. tainanus, C. matsuzawai, and C. oxystoma, to examine correlations between occurrence and temperature and precipitation (Table 6, Fig. 4). Culicoides arakawae (r = 0.205, p = 0.018; Rs = 0.303, p < 0.001) and C. oxystoma (r = 0.242, p = 0.034; Rs = 0.382, p < 0.001) exhibited weak Pearson and Spearman correlation coefficients (r and Rs, respectively) in correlation with temperature. Additionally, C. punctatus, C. tainanus, and C. matsuzawai showed no statistically significant correlation with temperature; no species demonstrated a statistically significant correlation with precipitation.
 |
Figure 4 Seasonal distribution and scatter plot of five predominant species in relation to temperature and precipitation. |
Correlation analysis about five predominant Culicoides species with temperature and precipitation.
Taxonomy of unrecorded Culicoides species
Genus Culicoides Latreille, 1809: 251
Type species: Culicoides punctatus Latreille (= Ceratopogon punctatus Meigen), by monotypy.
Subgenus Avaritia Fox 1955: 218
Type species: Ceratopogon obsoletus Meigen, by original designation.
Culicoides asiana Bellis, 2015: 29 (formerly Culicoides asiatica Bellis, in Bellis et al. 2014: 407)
Non-type material examined: ROK, Jeju-do, 33°21′54″N 126°20′17″E, 23.09.27 (three females), accession number PQ643289; 33°17′07″N, 126°28′09″E, 23.09.16 (two females), accession number PQ643290; 33°27′56″N 126°47′16″E 23.08.22 (1 female).
Diagnosis: According to Bellis et al. [6] (as C. asiatica), this species is the only one in the Imicola complex that exhibits two specific characteristics: a pale spot at the apex of the m1 cell of the wing that extends distally but does not narrow or reach the M2 vein, and a dark spot on the base of the costa that is significantly longer than the dark spot on the stigma.
 |
Figure 5 Culicoides asiana (A) lateral view, (B) wing, (C) head, (D) abdomen. Scale bars = 0.5 mm [A]; 0.1 mm [B, C, and D]). |
Body length 1.1 mm (1.01–1.25, n = 6). Head: Compound eyes contiguous, 26 μm or 2 facets, bare. Antennae with SCo on segments 1, 9–13; AR 1.26. Third palpal segment slightly swollen, with a shallow round sensory pit present. PR 2.2 (2.02–2.50, n = 5). P/H 0.71 (0.63–0.79, n = 5). Wings: 0.81 mm in length (0.77–0.88, n = 6) and 0.40 mm in width (0.38–0.44), CR 0.56 (0.54–0.58). Wing pattern matches those of previous records, as shown in Figure 2O. Abdomen: Two spermathecae present, oval-shaped and unequal in size, with developed neck. Rudimentary spermatheca and sclerotised ring present. Larger spermatheca 42 (39–46, n = 6) × 35 (32–41) μm, smaller one 33 (28–37) × 30 (26–36) μm, rudimentary spermatheca 12 (10–15) × 5 (4–5) μm, and sclerotised ring 8 (6–9) × 4 (3–5) μm.
Distribution: Japan, China, Taiwan, Malaysia, Indonesia, Thailand, Bangladesh, East Timor, Vietnam, Laos, and the ROK.
Remarks: Culicoides asiana is similar to the domestic species C. tainanus; the anal corner of C. tainanus’s wing is dark, and the pale spots in cells m1 and m2 are connected. In contrast, in C. asiana, the anal corner is pale, and the basal pale spot in cell m1 is distinctly separated from the spot in cell m2. In C. asiana, sclerotised plates surrounding the gonopore are simple and square-like plates, while those of C. tainanus have forefinger and thumb-like projections that partially encircle the gonopore opening as seen in the Orientalis complex [42].
Subgenus unplaced, Ornatus species group
Culicoides palawanensis Delfinado, 1961
Non-type material examined. ROK, Haenam, 34°38′15″N 126°19′55″E 23.06.16 (16 females), accession number PV111013–PV111017; 23.08.25 (one female), accession number PV111011; 23.09.27 (two females), accession number PV111012.
Diagnosis: According to Li et al. [36], the only species in the Ornatus group with a suite of characters including hairy eyes, SCo distributed on the flagellar segments 1–12, and spermathecae with a neck and lacking a sclerotised ring.
 |
Figure 6 Culicoides palawanensis female (A) lateral view, (B) wing, (C) head, (D) abdomen. Scale bars = 0.5 mm [A]; 0.1 mm [B, C, and D]; 0.05 mm [C-1]). |
Body length 2.03 (1.47–2.45, n = 19) mm. Head: Eyes separated by 18 (17–19, n = 8) μm, or about 1.2 facets; hairy eyes, with three short pubescence located on each hexagonal edge of the ommatidium. Antennae with SCo on segments 1–12. AR 1.51 (1.38–1.58, n = 5); third palpal segment swollen distally, moderately deep, round sensory pit present. PR 2.31 (2.07–2.47, n = 6). P/H 0.72 (0.64–0.81, n = 8). Wings: 1.21 (0.89–1.33, n = 18) mm in length, 0.48 (0.43–0.61) mm in width, CR 0.65 (0.63–0.67). Wing pattern matches that of previous records, as seen in Figure 2P. Abdomen: Two spermathecae, oval-shaped and subequal in size, with developed necks; rudimentary spermatheca present; sclerotised ring absent. The larger spermatheca 59 (55–68, n = 8) × 50 (46–66) μm, smaller one 55 (52–57) × 49 (44–56) μm, and rudimentary spermatheca 20 (0.18–24) × 3 (3–4) μm.
Distribution: China, Thailand, Philippines, Indonesia, Malaysia, and the ROK.
Remarks: C. palawanensis’s wing pattern is similar to those of the domestic species C. pallidulus and C. dendrophilus. However, C. pallidulus has larger pale spots on the wings and only one spermatheca, and C. dendrophilus has bare eyes.
Differences in body and wing length were observed between the individuals collected in June and those collected in August and September. The individuals collected in June had body lengths of 2.11 (1.83–2.25, n = 16) mm, wing lengths of 1.25 (1.18–1.33) mm, and wing widths of 0.56 (0.53–0.61) mm, while the individuals collected in August and September had body lengths of 1.57 (1.47–1.73, n = 3) mm, wing lengths of 0.99 (0.89–1.10) mm, and wing widths of 0.48 (0.43–0.54) mm. The COI sequences, however, did not differ between these individuals.
Cytochrome c oxidase subunit 1 (COI) analysis
Phylogenetic analyses based on the COI region were performed to confirm the identification of the two newly recorded species based on the sequences of related species downloaded from GenBank (Table 7, Figs. 7 and 8). The COI sequences of C. asiana were divided into 2 clades in the phylogenetic tree, one representing populations from Japan and the ROK, and the other representing populations from Southeast Asia. The sequences obtained in the ROK demonstrated genetic distances of 0–0.98% when compared with those from Japan (KJ162955, KJ162956, KT352310, KT352321, and KT352360) and 0.98–1.84% when compared with those from Southeast Asia (MW496167, MW496170, and MW496171) (Fig. 8B).
 |
Figure 7 Phylogenetic trees featuring the Culicoides species previously unrecorded in the Republic of Korea, created using the neighbour-joining method based on COI sequences. Bootstrap values < 70 are not displayed. Sequences collected in this study are marked with an asterisk (*). |
 |
Figure 8 Genetic distance of newly recorded species: total pairwise distance (A), C. asiana (B), and C. palawanensis (C). |
Pairwise distance (%) of newly recorded species: C. asiana, C. palawanensis.
The COI sequences of C. palawanensis were divided into two clades, one representing collections from China and the other collections from Thailand. The newly obtained sequences from the ROK exhibited genetic distances of 0–0.70% when compared with the sequences from China (KY441765, KY441793, KY441798, and KY441809); however, they exhibited distances of 2.72–3.45% with the sequences recorded from Thailand (KY441807 and ON002365) (Fig. 8C).
Discussion
Ecological surveys elucidating the seasonality, geographical distributions, and host preferences of Culicoides species are essential for identifying potential disease hotspots and for assessing the vector potential of individual species. In this study, we investigated the distribution of Culicoides at 15 locations throughout the southern part of the ROK at regular intervals, identifying 14 previously recorded species and 2 newly recorded species.
Previous research has shown that C. arakawae or C. punctatus were dominant nationwide [14, 26, 30, 65] exception on Jeju Island, while C. tainanus [28], C. circumscriptus [30], C. nipponensis [14, 25, 27], and C. erairai [27] exhibited dominance in specific studies. In the current study, C. arakawae or C. punctatus were dominant on the mainland, while C. matsuzawai was dominant on Jeju Island (Tables 4 and 5).
We collected C. arakawae throughout the survey period, with the highest abundances seen in July and August in this study. This is consistent with previous studies reporting high abundances from June to August [14] and July to August [26, 61], but Kim et al. [27] reported high numbers of C. arakawae in September to October. For C. punctatus, abundance was high in June and July in the current study, and Cho and Chong [14] also reported high abundances in June and July. However, occurrences of the species were concentrated later in the year in several recent studies, in which high populations were seen in July to August [26, 30] or October [27]. Culicoides tainanus populations were highest from May to July in our study, they were also high in Cho and Chong [14] and Kim et al. [28], but Kim et al. [26] reported high abundance in August.
Another species, C. nipponensis was highly abundant in Gangjin, in late July (37.14%, 283) and in both collections in September (19.16%, 146; 28.08%, 214) (Tables 3 and 4). This pattern was similar to the high relative abundances noted from June to August by Cho and Chong [14] and in August by Kim et al. [30]. However, subsequent research by Kim et al. [28] identified a new domestic species C. lungchiensis, that morphologically resembles C. nipponensis, and suggested that C. lungchiensis may have been misidentified as C. nipponensis on Jeju Island. In the current study, C. nipponensis was only collected in Gangjin, and all specimens from Jeju Island that resembled C. nipponensis were identified as C. lungchiensis, suggesting that C. nipponensis may indeed not inhabit Jeju Island and the distribution record of C. nipponensis in the mainland should be reconsidered.
We recorded significant numbers of C. matsuzawai in July, and the species was highly dominant in goat sheds on Jeju Island (Tables 3 and 4). This temporal pattern contrasts with the those seen in previous studies, which reported low numbers of specimens from July to September [14] in Gyeonggi Province or from June to September [28] on Jeju Island, respectively. In the current study, this species emerged as a dominant presence on Jeju Island, and a considerable number were also collected in Jinju and Sacheon (Table 4). The COI sequences of several C. matsuzawai specimens from Jinju, Sacheon, and Jeju Island (PQ623695–704, PQ263828, PQ263829, PQ489391) were registered in GenBank [23] and coincided with other sequences in the database. Therefore, further investigation is required to understand why we encountered unusually large numbers of C. matsuzawai.
The composition of collected Culicoides populations appeared to vary depending on farm types and environmental conditions. Both C. arakawae (34.8%) and C. punctatus (39.1%) were dominant in the collection from cattle farms on the mainland, whereas C. arakawae (93.9%) exhibited the highest abundance on mainland goat farms. On Jeju Island, C. tainanus (32.24%) and C. lungchiensis (47.07%) were the dominant species on cattle farms, while C. matsuzawai (87.56%) were dominant on goat farms (Table 5). This finding reflects a significant environmental divergence between Jeju Island and the mainland. Regardless of farm types, the composition and relative abundances of Culicoides species varied according to the collection site and environment (Table 4, Fig. 3). This indicates that Culicoides populations may be influenced by numerous unknown factors. Therefore, future studies accounting for multiple environmental conditions, such as temperature, humidity, and the local habitat surrounding collection sites, will be essential for a comprehensive analysis of species distributions.
This study reported two species previously unrecorded in the ROK. Culicoides asiana was collected only on Jeju Island in August and September. The presence of this species has been confirmed in neighbouring countries, including Japan and China, as well as regions of Southeast Asia [6, 19]. Given the specimens collection location and phylogenetic placement (Table 4, Figs. 7 and 8B), the populations of C. asiana on Jeju Island appear to be more closely related to species in Japan than to those found elsewhere, which is not surprising given the geographical proximity of Japan to southern Korea. Additionally, the morphological similarities between C. asiana and C. tainanus suggest that C. asiana may have been overlooked in prior studies and may indeed be more widespread in Korea.
Culicoides palawanensis was collected in Haenam in June, August, and September. This species has been reported in southern China, Thailand, the Philippines, and Indonesia [18, 36, 61], so our records from Korea represent a significant northerly extension of the range of this species. Among publicly available COI sequences, the C. palawanensis sequences from Haenam exhibited greater similarity to sequences obtained from populations in Hainan, China (Figs. 7 and 8C). Considering this, along with the collection locations in China and the ROK, it is likely that C. palawanensis is more widespread in China than currently believed. Additionally, the notable variations in body size observed between specimens collected during different collection periods indicate the possibility of seasonal phenotypic variation, as has been observed in other species [39].
The current data are insufficient to determine whether the two species are native to the ROK or were introduced from external sources. Therefore, further research, for example, monitoring Culicoides along with seasonal airflow changes, is warranted to investigate introduction routes. Furthermore, these two species are primarily reported in tropical to subtropical climates [6, 61]. This suggests that, due to climate change, the ROK is experiencing influxes of new subtropical species from abroad. Thus, thorough epidemiological investigations, for instance analysing vector distributions and identifying potential disease candidates, are imperative to prevent and control disease transmission via Culicoides vectors.
This study identified six recorded species and one previously unrecorded species that have been identified as confirmed or potential disease transmission vectors in the ROK. Specifically, C. arakawae, C. punctatus, C. oxystoma, and C. tainanus have been shown to carry viruses domestically [47, 65], while C. jacobsoni [20] and C. lungchiensis [63] are potential vectors in the country. Additionally, C. asiana is a newly recorded species known to be capable of transmitting AKAV [6] and may transmit BTV (like C. brevitarsis) [6, 64]. Consequently, these seven species warrant more attention than the others. However, as pathogen isolation experiments were not performed in this study, leaving the current status of pathogens in these species and the identification of risk areas undetermined, further investigation is needed.
To implement effective control measures, it is necessary to accurately identify both the hosts and pathogens associated with Culicoides. There is, however, little information on the host range of Korean species of Culicoides. Among the 16 species collected in this study, firm host records exist for only six species. Culicoides homotomus and C. nipponensis are known to feed on cattle and chickens [24], while C. kibunensis and C. punctatus exhibit broader host ranges, targeting multiple mammal and bird species, although C. punctatus is known to prefer cattle and horses [40, 46, 58]. Culicoides oxystoma is known to feed on cattle, goats, sheep, and pigs, with a preference for cattle [24, 25, 37]. These previous studies help explain the species distribution seen in this study; species were collected in proximity to their respective hosts. Notably, C. arakawae is known to feed on cattle, chickens, goats, and pigs [24, 37], and is thought to prefer avian species [66]. However, it has consistently shown high abundances around mammal farms in the ROK [26, 30, 65]. In the current survey, C. arakawae exhibited high collection rates on both cattle and goat farms, being consistently abundant at all the tested goat sheds on the mainland. Thus, it seems possible that host preference of C. arakawae is not perfectly understood. However, there are limitations in the current study: the influence of nearby wild bird or poultry populations, the dispersal ability of Culicoides species, and larval habitat were not measured. Further research, such as direct blood analyses from engorged Culicoides and the measurement of host-specific blood-feeding rates, will therefore be necessary to achieve a comprehensive understanding of host preferences.
In conclusion, this study provided updated information about the distributions of Culicoides species in the southern region of the ROK, identifying two previously unrecorded species, C. asiana and C. palawanensis. As a comprehensive survey on goat farms has not been conducted to date, the data obtained from this survey are expected to serve as a valuable foundational resource for the monitoring and analysis of Culicoides vectors associated with livestock diseases in this country. Until now, only four species have been directly associated with viruses within the ROK. Thus, the identification of a new potential disease vector, C. asiana, raises concerns regarding the introduction of new diseases and disease vectors from abroad. Therefore, the ongoing monitoring of Culicoides vectors and potential foreign incursions will be essential for ensuring the health and hygiene of domestic livestock in the future.
Acknowledgments
This project was supported by grants from the Animal and Plant Quarantine Agency (Grant No. Z-1543085-2023-25-01) from the Republic of Korea.
Conflicts of interest
The authors declare that they have no competing interests.
Author contribution statement
SBA, JJ, JR, and KSC conceived the study. SBA, JJ, JR, and JUJ collected the specimens. SBA and JJ conducted the experiments, analysed the data, and drafted the manuscript. JR, JUJ, ISR, and KSC helped draft the manuscript and analysed the data. All authors read and approved the final manuscript.
References
- Arnaud P. 1956. The heleid genus Culicoides in Japan, Korea and Ryukyu Islands (Insecta: Diptera). Microentomology, 21, 84–207. [Google Scholar]
- Bak U-B, Cheong C-K, Choi H-I, Lee C-W, Oh H-S, Rhee Y-O, Cho M-R, Lim Y-I. 1983. An outbreak of Ibaraki disease in Korea. Korean Journal of Veterinary Research, 23(1), 81–89. [Google Scholar]
- Bak U-B, Lim CH, Cheong CK, Hwang WS, Cho MR. 1980. Outbreaks of Akabane disease of cattle in Korea. Korean Journal of Veterinary Research, 20(1), 65–78. [Google Scholar]
- Barnett HC, Toshioka S. 1951. The bloodsucking insects, mites and ticks of Korea and their relation to disease transmission. San Francisco, California: 406th Medical General Laboratory. p. 1–25. [Google Scholar]
- Bellis G, Dyce A, Gopurenko D, Mitchell A. 2013. Revision of the Immaculatus group of Culicoides Latreille (Diptera: Ceratopogonidae) from the Australasian region with description of two new species. Zootaxa, 3680(1), 15–37. [CrossRef] [Google Scholar]
- Bellis G, Dyce A, Gopurenko D, Yanase T, Garros C, Labuschagne K, Mitchell A. 2014. Revision of the Culicoides (Avaritia) Imicola complex Khamala & Kettle (Diptera: Ceratopogonidae) from the Australasian region. Zootaxa, 3768(4), 401–427. [Google Scholar]
- Bellis G, Kim H-C, Kim M-S, Klein TA, Lee D-K, Gopurenko D. 2013. Three species of Culicoides Latreille (Diptera: Ceratopogonidae) newly recorded from the Republic of Korea. Zootaxa, 3718(2), 171–182. [Google Scholar]
- Bellis GA, Halling L, Anderson SJ. 2015. Pictorial key to adult female Culicoides Latreille, 1809 (Diptera: Ceratopogonidae) from the Northern Territory, Western Australia and South Australia. Austral Entomology, 54(1), 28–59. [Google Scholar]
- Borkent A. 2004. Insecta: Diptera, Ceratopogonidae, in Freshwater invertebrates of the Malaysian region. Kuala Lumpur, Malaysia: Academy of Sciences Malaysia. pp. 683–710. [Google Scholar]
- Borkent A. 2024. The phylogeny of the genera of biting midges (Diptera: Ceratopogonidae) of the World. Zootaxa, 5438(1), 1–274. [Google Scholar]
- Borkent A, Dominiak P. 2020. Catalog of the biting midges of the world (Diptera: Ceratopogonidae). Zootaxa, 4787(1), 1–377. [CrossRef] [Google Scholar]
- Borkent A, Dominiak P, Díaz F. 2022. An update and errata for the catalog of the biting midges of the world (Diptera: Ceratopogonidae). Zootaxa, 5120(1), 56–64. [Google Scholar]
- Bullock HR, Akiyama J. 1959. A new biting midge from Japan and Korea (Diptera, Heleidae). Medical Entomology and Zoology, 10(1), 23–26. [Google Scholar]
- Cho HC, Chong CS. 1974. Notes on biting midges of the genus Culicoides from South Korea-with special reference to unrecorded species and distribution. Korean Journal of Parasitology, 12(1), 45–75. [Google Scholar]
- Choi J-G, Kim HJ, Ha HH, Kang H-E. 2018. Atlas of the Culicoides in Korea (Diptera: Ceratopogonidae). Available from https://ebook.qia.go.kr/20190109_172028/ (in Korean). [Google Scholar]
- Collins ÁB, Doherty ML, Barrett DJ, Mee JF. 2019. Schmallenberg virus: a systematic international literature review (2011–2019) from an Irish perspective. Irish Veterinary Journal, 72(1), 9. [Google Scholar]
- Chung YS, Yoon MB, Kim HS. 2004. On climate variations and changes observed in South Korea. Climatic Change, 66(1), 151–161. [Google Scholar]
- Delfinado MD. 1961. The Philippine biting midges of the genus Culicoides (Diptera: Ceratopogonidae). Fieldiana Zoology, 33, 627–675. [Google Scholar]
- Duan YL, Li L, Bellis G, Yang ZX, Li HC. 2021. Detection of bluetongue virus in Culicoides spp. in southern Yunnan Province, China. Parasites & Vectors, 14(1), 68. [CrossRef] [PubMed] [Google Scholar]
- Duan Y-L, Li Z-H, Bellis GA, Li L, Liu B-G, Wang J-P, Liu J-M, Liao D-F, Zhu JB. 2024. Culicoides and midge-associated arboviruses on cattle farms in Yunnan Province, China. Parasite, 31, 72. [Google Scholar]
- Foxi C, Delrio G, Falchi G, Marche MG, Satta G, Ruiu L. 2016. Role of different Culicoides vectors (Diptera: Ceratopogonidae) in bluetongue virus transmission and overwintering in Sardinia (Italy). Parasites & Vectors, 9(1), 440. [Google Scholar]
- Hall TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series, 41(41), 95–98. [Google Scholar]
- Jeon J, Lee DY, An SB, Ryu J, Jeong JU, Roh IS, Choi KS. 2025. Hiding in plain sight: Uncovering the hidden diversity of Culicoides spp. (Diptera: Ceratopogonidae) in the Republic of Korea using DNA barcoding data. Acta Tropica, 270, 107821. [Google Scholar]
- Kang C, Yu H. 1991. Seasonal abundance and host blood meal sources of the genus Culicoides (Diptera: Ceratopogonidae) from cattle and poultry farms in Kyonggi Province, Korea. Korean Journal of Entomology (Korea Republic), 21(1), 29–36. [Google Scholar]
- Kar S, Mondal B, Ghosh J, Mazumdar SM, Mazumdar A. 2022. Host preference of bluetongue virus vectors, Culicoides species associated with livestock in West Bengal, India: Potential relevance on bluetongue epidemiology. Acta Tropica, 235, 106648. [CrossRef] [PubMed] [Google Scholar]
- Kim HC, Bellis GA, Kim M-S, Chong S-T, Lee D-K, Park J-Y, Yeh J-Y, Klein TA. 2012. Seasonal abundance of biting midges, Culicoides spp. (Diptera: Ceratopogonidae), collected at cowsheds in the southern part of the Republic of Korea. Korean Journal of Parasitology, 50(2), 127–131. [Google Scholar]
- Kim HC, Bellis GA, Kim M-S, Klein TA, Chong S-T, Park J-Y. 2014. Seasonal abundance of Culicoides (Diptera: Ceratopogonidae) collected by mosquito magnet® in Northern Gyeonggi-do (Province), Korea. Korean Journal of Parasitology, 52(1), 57–62. [Google Scholar]
- Kim HC, Bellis GA, Kim M-S, Klein TA, Gopurenko D, Cai D-C, Seo H-J, Cho I-S, Park J-Y. 2015. Species diversity and seasonal distribution of Culicoides spp. (Diptera: Ceratopogonidae) in Jeju-do, Republic of Korea. Korean Journal of Parasitology, 53(4), 501–506. [CrossRef] [PubMed] [Google Scholar]
- Kim JR, Kang JJ, Ri D, Kim HS, Kang SH, Choi SW, O HD, Kim DS, Ri MC, Hong UG, Ri GP. 1993. Baekdu Mountain series (Animals). Science and Technology Publishing House. pp. 183–184 (in Korean). [Google Scholar]
- Kim M-S, Kim HC, Bellis GA, Chong S-T, Kim H-S, Klein TA. 2021. Seasonal abundance of Culicoides at Yongsan US Army Garrison (USAG) and Camp Humphreys USAG, Republic of Korea, 2010–2013 and 2014–2017. Korean Journal of Parasitology, 59(3), 273–280. [CrossRef] [PubMed] [Google Scholar]
- Kim Y-H, Oem J-K, Lee E-Y, Lee K-K, Kim S-H, Lee M-H, Park SC. 2015. Seroprevalence of five arboviruses in sentinel cattle as part of nationwide surveillance in South Korea, 2009–2012. Journal of Veterinary Medical Science, 77(2), 247–250. [Google Scholar]
- Kim Y, Shim KM, Jung MP, Choi IT, Kang KK. 2017. Study on the change of climate zone in South Korea by the climate change scenarios. Korean Journal of Agricultural and Forest Meteorology. 19(2), 37–42. [Google Scholar]
- Kinoshita S. 1918. Chosen-san kyuketsusei Culicoides in tsukite. Dobutsugaku Zasshi, 30, 155–160 (in Japanese). [Google Scholar]
- Kitaoka S. 1984. Japanese Culicoides (Diptera: Ceratopogonidae) and keys for the species. Bulletin of the National Institute of Animal Health, 87, 73–108 (in Japanese). [Google Scholar]
- Lee W, Kim SY, Hur J, Park T, Do MS, Choi HK, Kim J, Lim C, Lee T, Bae YJ. 2023. Culicoides dokdoensis, sp. nov. (Diptera: Ceratopogonidae): A new biting midge species from Dokdo Island, Republic of Korea. Entomological Research, 53(11), 469–478. [Google Scholar]
- Li J-H, Gopurenko D, Cai D, Yang Y-M, Hu R, Thepparat A, Wardhana AH, Kim HC, Klein TA, Kim M-S. 2017. Culicoides Latreille biting midges (Diptera: Ceratopogonidae) of the Dongzhaigang Mangrove Forest, Hainan Province, China. Zootaxa, 4227(1), 49–60. [Google Scholar]
- Li N, Meng J, He Y, Wang W, Wang J. 2024. Potential roles of Culicoides spp. (Culicoides imicola, Culicoides oxystoma) as biological vectors of bluetongue virus in Yuanyang of Yunnan, P.R. China. Frontiers in Cellular and Infection Microbiology, 13, 1283216. [CrossRef] [PubMed] [Google Scholar]
- Lim SI, Kweon CH, Tark DS, Kim SH, Yang DK. 2007. Sero-survey on Aino, Akabane, Chuzan, bovine ephemeral fever and Japanese encephalitis virus of cattle and swine in Korea. Journal of Veterinary Science, 8(1), 45–49. [Google Scholar]
- Linley J, Hinds M. 1976. Seasonal changes in size, female fecundity and male potency in Culicoides melleus (Diptera: Ceratopogonidae). Journal of Medical Entomology, 13(2), 151–156. [Google Scholar]
- Martínez-de la Puente J, Figuerola J, Soriguer R. 2015. Fur or feather? Feeding preferences of species of Culicoides biting midges in Europe. Trends in Parasitology, 31(1), 16–22. [Google Scholar]
- McAlpine JF, Peterson BV, Shewell G, Teskey H, Vockeroth J, Wood D. 1981. Manual of Nearctic Diptera, 1, 393–421. [Google Scholar]
- Meiswinkel R. 2004. Adult characters defining and separating the Imicola and Orientalis species complexes of the subgenus Avaritia Fox, 1955 (Culicoides, Diptera: Ceratopogonidae). Veterinaria Italiana, 40(3), 345–351. [PubMed] [Google Scholar]
- Meiswinkel R, Venter GJ, Nevill EM. 2004. Vectors: Culicoides spp., in Infectious diseases of Livestock (2nd ed., Vol. 1). South Africa: Oxford University Press Cape Town. pp. 93–136. [Google Scholar]
- Mellor P, Boorman J, Baylis M. 2000. Culicoides biting midges: their role as arbovirus vectors. Annual Review of Entomology, 45(1), 307–340. [CrossRef] [PubMed] [Google Scholar]
- Mellor P, Wittmann E. 2002. Bluetongue virus in the Mediterranean Basin 1998–2001. Veterinary Journal, 164(1), 20–37. [Google Scholar]
- Ninio C, Augot D, Delecolle J-C, Dufour B, Depaquit J. 2011. Contribution to the knowledge of Culicoides (Diptera: Ceratopogonidae) host preferences in France. Parasitology Research, 108, 657–663. [Google Scholar]
- Oem J-K, Chung J-Y, Kwon M-S, Kim T-K, Lee T-U, Bae Y-C. 2013. Abundance of biting midge species (Diptera: Ceratopogonidae, Culicoides spp.) on cattle farms in Korea. Journal of Veterinary Science, 14(1), 91–94. [Google Scholar]
- Park BK, Rhee JC, An SH, Moon HK, Kim KS, Son DS, Lee DW. 1993. An outbreak of Chuzan disease in Korea and the immunogenicity of binary ethylenimine-treated chuzan virus vaccine in cattle. Korean Journal of Veterinary Public Health, 17, 301–305. [Google Scholar]
- Purse B, Carpenter S, Venter G, Bellis G, Mullens B. 2015. Bionomics of temperate and tropical Culicoides midges: knowledge gaps and consequences for transmission of Culicoides-borne viruses. Annual Review of Entomology, 60(1), 373–392. [CrossRef] [PubMed] [Google Scholar]
- Sanders CJ, Shortall CR, England M, Harrington R, Purse B, Burgin L, Carpenter S, Gubbins S. 2019. Long‐term shifts in the seasonal abundance of adult Culicoides biting midges and their impact on potential arbovirus outbreaks. Journal of Applied Ecology, 56(7), 1649–1660. [Google Scholar]
- Shin YK, Oem J-K, Yoon S, Hyun B-H, Cho I-S, Yoon S-S, Song J-Y. 2009. Monitoring of five bovine arboviral diseases transmitted by arthropod vectors in Korea. Journal of Bacteriology and Virology, 39(4), 353–362. [Google Scholar]
- Swanson DA. 2012. Ecology and phylogeny of the biting midge genus Culicoides (Diptera: Ceratopogonidae), PhD thesis, Clemson University, South Carolina. [Google Scholar]
- Sweet SA, Grace-Martin K. 1999. Data analysis with SPSS (Vol. 1), Boston, MA, USA: Allyn & Bacon. [Google Scholar]
- Tamura K, Stecher G, Kumar S. 2021. MEGA11: molecular evolutionary genetics analysis version 11. Molecular Biology and Evolution, 38(7), 3022–3027. [CrossRef] [PubMed] [Google Scholar]
- Tokunaga M. 1941. Biting midges from Manchuria (Ceratopogonidae, Diptera). Insecta Matsumurana, 15(3), 89–102. [Google Scholar]
- Tokunaga M. 1955. Notes on biting midges from Japan and Korea (Heleidae or Ceratopogonidae, Diptera). Scientific Reports of the Saikyo University. Agriculture, 7(7), 1–8. [Google Scholar]
- Tokunaga M. 1962. Biting midges of the Ryukyu Islands (Diptera: Ceratopogonidae). Pacific Insects, 4(1), 153–217. [Google Scholar]
- Tomazatos A, Jöst H, Schulze J, Spînu M, Schmidt-Chanasit J, Cadar D, Lühken R. 2020. Blood-meal analysis of Culicoides (Diptera: Ceratopogonidae) reveals a broad host range and new species records for Romania. Parasites & Vectors, 13, 79. [CrossRef] [PubMed] [Google Scholar]
- Wada Y. 1990. The verbosus group of the genus Culicoides Latreille (Diptera: Ceratopogonidae) in Japan, with descriptions of three new species and one hitherto unknown male. Tropical Medicine, 32, 49–72. [Google Scholar]
- Wada Y. 1999. Culicoides biting midges of Japan (Diptera: Ceratopogonidae). Nagasaki-ken Seibutsu Gakkaishi, 50, 45–70 (in Japanese). [Google Scholar]
- Wirth WW, Hubert AA. 1989. The Culicoides of southeast Asia (Diptera: Ceratopogonidae) (Vol. 44), American Entomological Institute. [Google Scholar]
- Yanase T, Kato T, Hayama Y, Shirafuji H, Yamakawa M, Tanaka S. 2019. Oral susceptibility of Japanese Culicoides (Diptera: Ceratopogonidae) species to Akabane virus. Journal of Medical Entomology, 56(2), 533–539. [Google Scholar]
- Yanase T, Kato T, Katagiri Y, Aizawa M, Nakamura K, Kokuba T, Araki M, Shirafuji H, Yamakawa M, Tsuda T. 2010. Isolation and characterization of Bluetongue virus from Culicoides brevitarsis (Diptera: Ceratopogonidae) in Okinawa. Medical Entomology and Zoology, 61(1), 85–91. [Google Scholar]
- Yanase T, Kato T, Kubo T, Yoshida K, Ohashi S, Yamakawa M, Miura Y, Tsuda T. 2005. Isolation of bovine arboviruses from Culicoides biting midges (Diptera: Ceratopogonidae) in southern Japan: 1985–2002. Journal of Medical Entomology, 42(1), 63–67. [Google Scholar]
- Yang D, Yang M-S, Rhim H, Han J-I, Oem J-K, Kim Y-H, Lee K-K, Lim C-W, Kim B. 2018. Analysis of five arboviruses and Culicoides distribution on cattle farms in Jeollabuk-do, Korea. Korean Journal of Parasitology, 56(5), 477–485. [Google Scholar]
- Yu C-Y, Wang J-S, Yeh C-C. 2000. Culicoides arakawae (Diptera: Ceratopogonidae) population succession in relation to leucocytozoonosis prevalence on a chicken farm in Taiwan. Veterinary Parasitology, 93(2), 113–120. [Google Scholar]
- Yu YX, Liu JH, Liu GP, Liu ZJ, Hao BS, Yan G, Zhao TS. 2005. Ceratopogonidae of China: Insecta, Diptera (Vol. 2). Military Medical Science Press. [Google Scholar]
Cite this article as: An SB, Jeon J, Ryu J, Jeong J-U, Roh I-S & Choi KS. 2026. Diversity and abundance of Culicoides on goat and cattle farms in the southern part of the Republic of Korea. Parasite 33, 6. https://doi.org/10.1051/parasite/2026005.
All Tables
Seasonal distribution of 16 Culicoides species collected in southern Korea, 2023.
Total number (%*) of Culicoides species collected at 15 sites in southern Korea, 2023.
Correlation analysis about five predominant Culicoides species with temperature and precipitation.
All Figures
 |
Figure 1 Collection sites for the study. A. Hapcheon, B. Sancheong, C. Jinju, D. Sacheon, E. Hadong, F. Gwangyang, G. Suncheon, H. Boseong, I. Gangjin, J. Haenam, K. Jeju1, L. Jeju2, M. Jeju3, N. Jeju4. O. Jeju5; Cow shed (A, B, C, F, I, J, K, L, N, O), Goat shed (D, E, G, H, M). |
| In the text | |
 |
Figure 2 Wings of 16 collected Culicoides species including 2 newly recorded species (O and P): (A) C. arakawae, (B) C. homotomus, (C) C. jacobsoni, (D) C. japonicus, (E) C. kibunensis, (F) C. lungchiensis, (G) C. matsuzawai, (H) C. morisitai, (I) C. nipponensis, (J) C. oxystoma, (K) C. pictimargo, (L) C. punctatus, (M) C. sinanoensis, (N) C. tainanus, (O) C. asiana, (P) C. palawanensis. Scale bars = 0.1 mm. |
| In the text | |
 |
Figure 3 Seasonal distribution of Culicoides species at collection sites with climate parameters. |
| In the text | |
 |
Figure 4 Seasonal distribution and scatter plot of five predominant species in relation to temperature and precipitation. |
| In the text | |
 |
Figure 5 Culicoides asiana (A) lateral view, (B) wing, (C) head, (D) abdomen. Scale bars = 0.5 mm [A]; 0.1 mm [B, C, and D]). |
| In the text | |
 |
Figure 6 Culicoides palawanensis female (A) lateral view, (B) wing, (C) head, (D) abdomen. Scale bars = 0.5 mm [A]; 0.1 mm [B, C, and D]; 0.05 mm [C-1]). |
| In the text | |
 |
Figure 7 Phylogenetic trees featuring the Culicoides species previously unrecorded in the Republic of Korea, created using the neighbour-joining method based on COI sequences. Bootstrap values < 70 are not displayed. Sequences collected in this study are marked with an asterisk (*). |
| In the text | |
 |
Figure 8 Genetic distance of newly recorded species: total pairwise distance (A), C. asiana (B), and C. palawanensis (C). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


