| Issue |
Parasite
Volume 33, 2026
|
|
|---|---|---|
| Article Number | 9 | |
| Number of page(s) | 13 | |
| DOI | https://doi.org/10.1051/parasite/2026010 | |
| Published online | 24 February 2026 | |
Research Article
Phenotypic changes in natural populations of Anopheles gambiae s.l. at the onset of the long dry season in tropical savannahs of Burkina Faso, West Africa
Évolution phénotypique des populations naturelles d’Anopheles gambiae s.l. au début de la longue saison sèche dans les savanes tropicales du Burkina Faso, en Afrique de l’Ouest
1
MIVEGEC, University of Montpellier, IRD, CNRS, 911 Avenue Agropolis, 34090, Montpellier, France
2
Institut de Recherche en Sciences de la Santé, Direction Régionale de l’Ouest, 01 P.O. Box 545, Bobo Dioulasso, Burkina Faso
3
Institut de Recherche Agricole pour le Développement (IRAD), PO Box 2123, Yaoundé, Cameroun
4
UMR 6553, University Rennes, CNRS, ECOBIO (Ecosystèmes, Biodiversité, Évolution), 263 Avenue du Général Leclerc, 35042 Rennes, France
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
; This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
18
November
2025
Accepted:
31
January
2026
Abstract
In the tropical savannahs with long dry seasons, malaria mosquito populations virtually disappear after the drying up of breeding sites to reappear in large numbers at the onset of next rainy season. While aestivation and long-distance migration are proposed as key strategies enabling these vectors to persist through the dry-season, the physiological, biochemical, and morphological traits underpinning these mechanisms remain insufficiently explored, particularly under natural field conditions. This study explored seasonal changes in Anopheles coluzzii, An. gambiae, and An. arabiensis at the onset of the dry season in the harsh savannahs of Burkina Faso, West Africa. Late-instar immature specimens were collected from two ecologically distinct sites, one with permanent and the other with only temporary breeding habitats, during the rainy season and the transitional period into the dry season. Larvae were reared to adulthood under natural conditions and several traits were analysed including ovarian development, sub-cuticular fat body hypertrophy, body size, and energy reserves. Gonotrophic dissociation was significantly more frequent in An. coluzzii at the onset of the dry season, indicating a shift toward reproductive arrest. All three species exhibited increased body size and cuticular fat deposits during the transitional period, though with species-specific differences. Notably, only An. coluzzii showed significant increases in energy reserves (proteins, lipids, and carbohydrates) during the transition period. These adaptive responses differed between the study sites, suggesting the influence of breeding habitats. The findings highlight that species within the An. gambiae complex engage in distinct phenotypic trajectories at the onset of the dry season, suggesting divergent adaptations and trade-offs in energy acquisition and allocation to survive during the dry season.
Résumé
Dans les savanes tropicales à longue saison sèche, les populations de moustiques vecteurs du paludisme disparaissent virtuellement après l’asséchement des gîtes larvaires, pour réapparaître en grand nombre au début de la saison des pluies suivante. Si l’estivation et les migrations sur de longues distances sont considérées comme des stratégies clés permettant à ces vecteurs de survivre durant la saison sèche, les caractéristiques physiologiques, biochimiques et morphologiques sous-jacentes à ces mécanismes restent insuffisamment étudiées, notamment en conditions naturelles. Cette étude explore les variations saisonnières d’Anopheles coluzzii, d’An. gambiae et d’An. arabiensis au début de la saison sèche dans les savanes arides du Burkina Faso, en Afrique de l’Ouest. Des larves en fin de stade larvaire ont été collectées sur deux sites écologiquement distincts, l’un présentant des gîtes larvaires permanents et l’autre uniquement des gîtes temporaires, durant la saison des pluies et la période de transition vers la saison sèche. Les larves ont été élevées jusqu’à l’âge adulte en conditions naturelles et plusieurs caractéristiques ont été analysées, notamment le développement ovarien, l’hypertrophie du corps gras sous-cuticulaire, la taille corporelle et les réserves énergétiques. La dissociation gonotrophique était significativement plus fréquente chez An. coluzzii au début de la saison sèche, indiquant un arrêt de la reproduction. Les trois espèces ont présenté une augmentation de leur taille corporelle et de leurs dépôts de graisse cuticulaire durant la période de transition, avec toutefois des différences spécifiques à chaque espèce. Notamment, seule An. coluzzii a montré une augmentation significative de ses réserves énergétiques (protéines, lipides et glucides) durant cette période. Ces réponses adaptatives différaient selon les sites d’étude, suggérant l’influence des habitats de reproduction. Ces résultats soulignent que les espèces du complexe An. gambiae adoptent des trajectoires phénotypiques distinctes au début de la saison sèche, suggérant des adaptations divergentes et des compromis dans l’acquisition et l’allocation d’énergie pour survivre durant cette période.
Key words: Malaria vectors / Aestivation / Long-distance migration / Cuticular fat deposit / Body size / Ovarian development / Energy reserves
Edited by Jean-Lou Justine
© W. Mamai et al., published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Despite intensive efforts to curb the disease, malaria remains a major global health burden. In 2023, 263 million human cases and 597,000 deaths were reported worldwide [81]. The majority of this burden is concentrated in African regions, where about 94% of cases and 90% of total deaths are reported. The primary malaria vectors in Africa are mosquitoes from the Anopheles gambiae s.l. complex and from the Anopheles funestus species group [76, 85]. Anopheles coluzzii Coetzee and Wilkerson 2013, (formerly M-form An. gambiae), An. gambiae Giles (formerly S-form An. gambiae), and An. arabiensis Patton are the most widespread and efficient vectors across Africa [9, 10]. These sibling species coexist in sympatry, but display key differences in traits such as host preference and larval ecology [9, 57, 84]. For instance, An. gambiae and An. arabiensis are typically associated with areas that have temporary breeding sites [11, 74], whereas An. coluzzii favours more stable, permanent larval habitats [4, 36].
In the West African savannah, the dry season can last for up to nine months, during which most standing water bodies and other surface water sources suitable for oviposition and larval development may completely dry up. This period is characterised by extreme environmental conditions with temperatures frequently exceeding 35 °C and relative humidity dropping below 10% [60, 61]. Ongoing climate change, with rising global temperatures, is likely to influence the distribution and (re-)emergence of vector-borne diseases such as malaria, West Nile fever, and dengue [29, 67, 71]. Understanding the mechanisms by which malaria vectors survive and persist through such a harsh dry season is crucial for guiding the development of alternative and more effective vector control strategies.
Several insects face harsh and variable environmental conditions and have thus evolved a wide range of survival mechanisms to cope with desiccation, heat stress, or food deprivation. These strategies include entering dormancy states such as diapause or quiescence [16, 17, 24, 56, 62, 79] and migration to areas with more favourable conditions [1, 8, 14, 33]. Several studies conducted on Anopheles mosquitoes [1, 15, 46–50, 56, 61, 65, 66, 75] support the hypothesis that population persistence during the dry season occurs primarily via aestivation in the form of adult females, particularly in An. coluzzii, as opposed to long-distance migration to and from areas with suitable breeding sites in An. gambiae and An. arabiensis. Under unfavourable conditions, the energetic cost of survival is generally higher, leading to potential trade-offs with other life-history components [72, 77]. As a consequence, organisms may initiate preparatory adjustments, including morphological and physiological adaptations in anticipation of these challenging conditions [2, 42–45, 54, 60]. Alterations in reproductive physiology, accumulation of fat reserves, changes in wing morphology, and reallocation of energy resources are among the most commonly documented traits associated with diapause [14, 18, 38, 59, 79].
Ovarian development is a physiological process by which female mosquitoes produce and mature eggs. It comprises two distinct phases (previtellogenic and vitellogenic stages), with ovarian development being triggered by the ingestion of a blood meal. In the absence of a blood meal, there is no progression beyond the pre-vitellogenic stage. When the ovaries or follicles of female mosquitoes that have taken a full blood meal fail to develop, the arrested ovarian development is termed gonotrophic dissociation [30, 82, 83].
During unfavourable periods, the fat body plays a vital role in energy storage and metabolic regulation, thus driving mosquito survival [3, 68]. Seasonal changes in fat body hypertrophy characterised by increased fat deposits and enlarged lipid droplets have been reported in numerous insects, including mosquitoes [3, 13, 41, 78]. In addition to the fat body, the accumulation of nutritional energy reserves, in particular glycogen and sugars [3], enables mosquitoes to withstand extended periods of environmental stress. As large body reserves increase survival probability, body size also serves as an indicator of adaptive responses in anticipation of diapause, with larger sizes often reflecting enhanced survival capacity [5]. Approaches such as geometric morphometrics, which assess shape, size, and allometric relationships, provide valuable insights into phenotypic variability under various environmental settings [6, 26, 28, 37].
In the present work, we investigated whether physiological and morphological changes occur in An. coluzzii, An. gambiae, and An. arabiensis in anticipation of the dry season, potentially as adaptive strategies to enhance survival during the prolonged and harsh environmental period. We hypothesize that i) the simultaneous occurrence of elevated temperatures, reduced relative humidity (RH), and increased larval crowding consequent to the progressive desiccation of aquatic breeding sites at the onset of the dry season, act as a set of token stimuli (predictive environmental cues) for mosquitoes, signalling upcoming inhospitable environmental conditions; ii) these stimuli trigger a cascade of physiological, behavioural, and morphological responses in mosquitoes, as a means for their adaptive survival strategies; and iii) these responses include gonotrophic dissociation, development of sub-cuticular fat deposits, increased teneral reserves, and larger body size, and may vary across species and populations sampled from different geographic locations. Beyond its ecological relevance, a better understanding of these seasonal survival mechanisms could support the development of novel malaria vector control interventions.
Material and methods
Study sites
Two ecologically distinct sites, which differ in terms of mosquito population dynamics and the seasonal availability of breeding sites were selected for insect collections. The first site, Bama (11° 23′ N, 4° 24′ W), is a rice cultivation area situated approximately 30 km north of Bobo-Dioulasso (Southwestern Burkina Faso, West Africa) (Figure S1). The rice paddies are constantly irrigated with water from the nearby perennial Kou River, ensuring the presence of stable aquatic habitats throughout the year. These environmental conditions provide permanent habitats for mosquito breeding and are thus characterized by endemic malaria transmission [12, 35]. In this locality, An. coluzzii is the predominant malaria vector. The second site, Soumousso (11° 04′ N, 4° 03′ W), is a savannah area located 30 km east of Bobo-Dioulasso (Figure S1). In contrast to Bama, Soumousso experiences strong seasonal fluctuations in the availability of breeding sites, which directly influence vector abundance: An. gambiae and An. arabiensis are abundant in the rainy season when breeding sites are also abundant, but are very rarely found during the dry season due to the disappearance of breeding sites [20, 21, 70]. Following mosquito sampling from both study sites, larvae and pupae were transferred to Bama for transplantation into a large larval habitat present year-round and where a Vantage Pro2 weather station (Weatherlink; Davis Instruments, Hayward, CA, USA) was installed to continuously monitor environmental conditions. Temperature, relative humidity, and rainfall were recorded at hourly intervals.
Experimental design
The experimental design was divided into two distinct parts. The first part of the study (Experiment 1) focused on assessing ovarian development (gonotrophic dissociation), fat body hypertrophy (including sub-cuticular fat deposit and lipid droplets), insemination status, and geometric morphometrics (centroid size). The second part of the study (Experiment 2) was designed to investigate the dynamics of energy reserve allocation in adult females during the larval developmental stage. The overall experimental setup is illustrated in Figure 1.
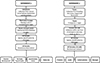 |
Figure 1 Experimental study design: Experiment 1 (left) and Experiment 2 (right). |
Experiment 1: Assessment of ovarian development, fat body hypertrophy, insemination status, and geometric morphometrics
The objective of this experiment was to investigate seasonal phenotypic variation in ovarian development, fat body hypertrophy, and centroid size.
Immature specimen sampling and transplantation
Immature mosquitoes were collected from natural larval habitats at the aforementioned study sites in 2011, during two key periods: (i) September, hereafter referred to as the rainy season, and (ii) November to December, hereafter referred to as the dry season. Aquatic habitats including small rain pools, cattle hoofprints, footprints, mud holes, and stagnant puddles near bridges or lowland rivers were surveyed for the presence of immature mosquitoes. Larvae were collected at both sites (Bama and Soumousso) using a 350 mL soup ladle and transferred to large holding containers. Anopheline larvae were visually sorted, and only late instar stages (third instar to pupa) were retained and counted. Late-instars were specifically selected based on the assumption that they had already been exposed to environmental token stimuli, thereby preconditioning the emerging adults to initiate survival-related physiological responses. To ensure natural larval development under identical biotic and climatic conditions until adult emergence, larvae and pupae from both study sites were transferred the same day and transplanted to a large natural breeding site located in Bama using transplant cages as described by Diabaté et al. [22, 23].
Adult emergence and blood feeding
Following adult emergence (between 1 to 3 days after larval transplantation), mosquitoes were collected in the early morning for several consecutive days using Drosophila tubes. Morphological identification was performed using standard taxonomic keys to confirm adults belonging to the An. gambiae s.l complex [34]. Identified adults were subsequently transferred to a large screened greenhouse enclosure (15 × 8 × 4.5 m) constructed with a metal frame and covered by mesh walls and a netted roof supported by metal structures, as described by Mamai et al. [61]. This setup effectively exposed mosquitoes to the prevailing environmental conditions. An equal number of male and female adult mosquitoes (50:50 sex ratio) were placed in a 30 × 30 × 30 cm cage to allow mating. A 10% glucose solution was provided ad libitum as a carbohydrate source. For blood feeding, restrained rabbits were positioned on top of the cage and served as a blood meal source for females on days 2, 4, 6, 8, and 10 post-emergence. Blood feeding occurred during the night from 07:30 to 08:30 pm, after which females were visually observed for the presence of blood in the abdomen.
Females lacking visible blood meals in their abdomens were removed and transferred to smaller cages for an additional one-hour feeding period. Females that successfully fed during either session were returned to the original cage, while individuals that failed to feed were excluded from the experiment. On the fourteenth day, mosquito cages were transported from the greenhouse to the laboratory of the Institut de Recherche en Sciences de la Santé (IRSS) in Bobo-Dioulasso, Burkina Faso. Upon arrival at the IRSS, females were promptly knocked down at −20 °C, after which they were individually isolated and stored at −80 °C for subsequent analysis.
Physiological and morphological assessments
Legs were carefully removed from each specimen and preserved in silica-gel desiccant for species identification using the SINE PCR method, as described by Santolamazza et al. [73]. The remaining carcasses were dissected under a microscope (Leica ICC 50) in a drop of physiological saline for the subsequent investigations. From a total of 39,016 late-stage immature mosquitoes collected, 1,946 blood-fed females were successfully identified across both study sites and subsequently included in the analysis.
1) Ovarian development – ovaries were examined to determine the stage of follicular maturation, following the classification system established by Christophers and further refined by Detinova [19]. Ovarian stages 1 to 3 were observed using 100× and 200× magnification, while stages 4 and 5 were determined directly under the binocular microscope (40 × magnification);
2) Fat body hypertrophy (Figure S2) – the presence (Figure S2-A) or absence (Figure S2-C) of sub-cuticular fat deposits, along with associated lipid droplets (Figure S2-B) was assessed directly under a dissecting microscope;
3) Insemination status – a random subsample of 300 females, drawn from the total females collected at each site was analysed using the methodology previously reported in Tripet et al. [80]. The abdomen was excised and immersed in 70% ethanol for 5 days. Subsequently, the spermathecae was then extracted from the 8th segment and opened to determine the presence or absence of a sperm ball (which forms upon contact with ethanol);
4) Geometric morphometrics – the left wings of adult female mosquitoes were gently removed from the thorax using fine forceps, then dry-mounted onto microscope slides and covered with coverslips. The prepared slides were allowed to dry before imaging. Wing photographs were captured using a Leica binocular microscope equipped with a digital camera connected to a computer. The images were processed and analysed using standard geometric morphometric techniques. For each wing, 12 landmarks (vein crosses) were identified and digitised (Fig. 2) using the COO package of Collecting Landmarks for Characterization and Identification (CLIC) software [26]. Wing size was quantified using the isometric estimator known as “centroid size”, defined as the square root of the sum of the squared distances of each landmark from the centroid of the configuration [25]. Centroid size has been widely recognised as a robust and informative proxy of body size in insects [27, 52, 53].
 |
Figure 2 A left wing belonging to a member of the Anopheles gambiae species complex indicating the location of 12 landmarks used in geometric morphometrics analysis using CLIC software. |
Experiment 2: Monitoring the dynamics of energy reserve allocation in newly emerged adult females
The goal of this experiment was to assess whether environmental conditions during larval development across distinct seasonal phases lead to differential allocation of energy reserves in newly emerged adult females (within the first hour of their adult life), prior to any post-emergence feeding or metabolic activity.
Immature specimen sampling
Immature specimens were collected at the aforementioned study sites from breeding habitats in 2011 during three distinct seasonal periods: (i) the onset of the rainy season (June), (ii) the peak of the rainy season (August), and (iii) the onset of the dry season (November). Only collected pupae were retained and placed in trays inside emergence cages (30 × 30 × 30 cm), which were checked regularly until emergence. Newly emerged female mosquitoes were collected at hourly intervals and immediately flash-frozen in liquid nitrogen to ensure minimal post-emergence metabolic transformation of larval-derived reserves. Upon return to the laboratory, the tubes containing the mosquitoes were stored at −80 °C for subsequent analysis of energy reserves.
Nutrient reserves
Legs were carefully removed from each specimen and preserved in silica-gel desiccant for species identification using the SINE PCR method [73]. Following successful molecular identification, 30 individuals per species, site, and collection period were used for nutrient quantification. From each mosquito specimen, the amount of body proteins, lipids, carbohydrates, and glycogen were quantified using colorimetry-based biochemical assays, following the protocol developed by Rivero et al. [69] and adapted by Mouline et al. [64].
Briefly, individual mosquitoes were homogenised in 70 μL of methanol, followed by the addition of 120 μL of methanol. The resulting homogenate was split into two equal aliquots of 70 μL each, one of which was used for protein quantification. To the second aliquot, 680 μL of chloroform: methanol (1:2) and 100 μL of sodium sulfate were added and homogenised. The final supernatant was divided equally for lipid and carbohydrate quantification, while the precipitate was used for glycogen determination. Protein concentrations were determined using the Bradford assay, and lipids using the vanillin reagent assay, while both carbohydrate and glycogen content were determined using an anthrone-based assay. Metabolite concentrations were calculated using standard curves specific to each biochemical compound.
Statistical analysis
All statistical analyses were conducted using R software, version 4.3.2 (R Development Core Team 2008) along with the RStudio environment, version 2024.10.31. (RStudio, Inc. Boston, MA, USA, 2016). For ovarian development, females were pooled for pre-vitellogenic (i.e. Christophers’ stage I + II) and vitellogenic (III–V) stages, and Chi-square tests were used to compare their distribution across species, sites, and collection periods. Fisher’s exact tests were used to explore differences in the distribution of the three phenotypic classes of fat body hypertrophy across species, sites, and collection periods. Geometric morphometric analyses were based on landmark coordinate data, which were subjected to generalised procrustes superimposition and standard geometric morphometric procedures for multivariate analysis [26–28]. The centroide size was analysed using a Gaussian linear mixed-effects model, with season and species as fixed effects.
For nutrient reserves data analysis, one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was performed using GraphPad Prism, version 5.00. Graphical representations were produced using Microsoft Excel 2019. Normality and homoscedasticity of data were verified by Kolmogorov-Smirnov and Bartlett tests, respectively.
Results
Environmental conditions at the Bama study site
Climatic variables, including monthly average temperatures (°C), relative humidity (%), and total yearly rainfall (mm), measured at the study site in Bama are presented in Figure S3. As expected, the area exhibited pronounced fluctuations in temperature and humidity regimes, allowing for the definition of three distinct phases:
Rainy season (from mid-June to mid-October). This phase accounts for the majority of annual precipitation (about 740 mm) and is characterized by moderately low and stable temperatures (e.g. range 22–30 °C), along with high (>50%) relative humidity levels;
Transitional period (November–December). This phase marks the onset of the dry season and is characterized by substantial diurnal variations, with extreme values for both temperature (e.g. range 12–35 °C) and relative humidity (10–93%). The broad thermal amplitude is attributed to marked night-time cooling, with temperatures occasionally dropping to as low as 10 °C;
Hot dry season (January to mid-June). This period is characterised by extreme daytime temperatures (up to 38.4 °C), persistently low relative humidity (~10%), and a narrower diurnal thermal range compared to the transitional phase, largely due to elevated night-time temperatures. Notably, during the transitional period between rainy and dry seasons (i.e. onset of dry season), most larval breeding sites dried up at our study sites. This phase potentially corresponds to the preparatory stage of aestivation in Anopheles mosquitoes, during which key physiological adaptations for survival are likely initiated.
Species composition
A total of 39,016 late-stage immature specimens (L3 to pupa) belonging to An. gambiae complex were collected across the two study sites in Experiment 1. PCR analysis successfully identified 1,946 females 14 days after successful blood feeding. Species-specific distribution by location revealed that only An. coluzzii was found in Bama, accounting for 100% (n = 803) of the females identified at this site. In contrast, among the 1,143 specimens identified in Soumousso, An. gambiae was the most abundant, representing 48.47% (n = 554), followed by An. arabiensis (29.66%, n = 339), and An. coluzzii (21.87%, n = 250).
Ovarian development
Dissection of the spermathecae from 300 females from both sites revealed an insemination rate of 98%, indicating that the observed ovarian development reflects normal reproductive biology and is not attributable to insemination failure.
Regardless of the season, An. coluzzii was the species that most efficiently developed eggs beyond ovarian stage 2 (ranging from 65.15 to 80.77%) compared to both An. arabiensis (51.02–59.91%) and An. gambiae (31.36–31.88%) (Fig. 3, Table 1). Despite multiple blood meals being provided, 19.23 to 68.64% of female mosquitoes collected at both localities failed to develop their ovaries beyond Christopher’s stage 2, indicating a physiological state of gonotrophic dissociation. Interestingly, An. coluzzii from both sites showed significantly higher proportions of females in gonotrophic dissociation at the onset of the dry season than during the rainy season (χ2 = 3.953, df = 1, p = 0.004 and χ2 = 5.584, df = 1, p = 0.018 for Soumousso and Bama populations, respectively). By contrast, no significant seasonal variation in the proportion of females in gonotrophic dissociation was evidenced in An. gambiae (χ2 = 0.00066, df = 1, p = 0.979) and An. arabiensis females (χ2 = 1.846, df = 1, p = 0.17) from Soumousso (Table 1).
 |
Figure 3 Ovarian developmental stages in female Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis during the rainy and at the onset of the dry seasons, and across two study sites in Burkina Faso. A = Bama (permanent breeding sites); B = Soumousso (temporary breeding sites). |
Statistical analyses comparing the effects of season, locality, and species on phenotypic traits in Anopheles mosquitoes. Results present probabilities from Chi-square (χ2) tests for gonotrophic dissociation, Fisher’s exact tests for fat body hypertrophy (lipid droplets and fat deposits), and linear mixed model (LMM) analyses for wing centroid size. Significant differences (p < 0.05) indicate the influence of season (rainy vs. dry), study locality (Bama vs. Soumousso), and interspecific variation (Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis) on these traits.
Fat body hypertrophy
The proportion of females with subcuticular fat body hypertrophy in the form of fat deposit was significantly higher at the onset of the dry season by comparison to the rainy season across the three Anopheles species, and at both sites for An. coluzzii (p < 0.05, Fig. 4, Table 1), although this trend was more marked in Soumousso (temporary) than in Bama (permanent) (p < 0.05). In turn, the proportion of females with no evidence for fat body hypertrophy (i.e. no fat deposit) was higher during the rainy than at the onset of the dry season in all species, accounting for over 98% of adult An. coluzzii females collected in Bama during the rainy season. At the onset of the dry season, very few female mosquitoes were deprived of lipid reserves in Soumousso, regardless of the species, whereas in Bama, they still represented over 40% of An. coluzzii females. The proportion of females showing subcuticular lipid droplets was highest in Soumousso during the rainy season, in all three species, although with statistically significant differences in their distribution between species. It was also the major phenotypic class observed at the onset of the dry season in An. arabiensis, as well as in An. coluzzii females from both sites. The proportion of females with lipid droplets was significantly higher at the onset of the dry season than in the rainy season in An. coluzzii in Bama (p < 0.05, Table 1), whereas An. arabiensis showed no seasonal difference (p = 0.112). Within An. coluzzii, the proportion of females with lipid droplets was higher at the temporary site (Soumousso) than at the permanent site (Bama), at both time points (p < 0.05, Table 1).
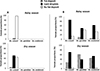 |
Figure 4 Proportion of Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis females with sub-cuticular fat body hypertrophy collected from two different sites of Burkina Faso. A = permanent (Bama); B = temporary (Soumousso). See Supplementary Material S2 for phenotype description. |
Interestingly, at the onset of the dry season in Soumousso, the proportion of females of all three species in a state of gonotrophic dissociation were significantly more likely to possess subcuticular fat deposits and lipid droplets (Fisher test, p < 0.05).
Adult body size
The isometric estimator of the adult body size, measured as wing centroid size, is shown in Figure 5. A total of 107 individuals were analysed: An. coluzzii (n = 47), An. gambiae (n = 32), and An. arabiensis (n = 28). In particular, An. coluzzii exhibited significantly larger body size at the onset of the dry season (LMM, df = 45, t = 3.914, p = 3e-04) compared to the rainy season. However, this difference was not significant in An. gambiae (LMM, df = 30, t = 1.028, p = 0.312) and An. arabiensis (LMM, df = 26, t = 0.783, p = 0.441). Across all seasons, An. arabiensis females were significantly larger than An. coluzzii (LMM, df = 73, t = −4.301, p = 1e–04) and An. gambiae females (LMM, df = 58, t = −5.199, p < 0.005).
 |
Figure 5 Wing centroid size in Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis collected during the rainy season and at the onset of the dry season at two sites in Burkina Faso. Different letters indicate statistically different p-values. |
Nutrient reserves
At each study site, only one species was found (collected) in sufficient numbers for analysis, i.e. An. coluzzii from Bama (permanent breeding sites) and An. gambiae from Soumousso. As a result, analyses were unfortunately restricted to these two mosquito species. The results presented in Figure 6 revealed significant seasonal variation in nutrient reserves (ANOVA, F = 28.39, df = 11, p < 0.001 and F = 19.17, df = 11, p < 0.001, respectively for An. coluzzii and An. gambiae). In An. coluzzii, protein, lipid, and carbohydrate contents were significantly lower during the peak of the rainy season (August) compared to the onset of the rainy season (June) (Fig. 6, Tukey’s post hoc test, p < 0.005), while glycogen levels remained stable. At the onset of the dry season (November), protein, lipid, and carbohydrate reserves significantly increased relative to the levels observed in the peak of the rainy season (August) (Tukey’s post hoc test, p < 0.005). In contrast, An. gambiae exhibited no significant changes in protein, lipid, and carbohydrate contents between the onset (June) and the peak (August) of the rainy season (Tukey’s post hoc test, p > 0.005). At the onset of the dry season, lipid and glycogen levels remained unchanged, whereas carbohydrate levels decreased and protein levels increased significantly (Tukey’s post hoc test, p < 0.005). Overall, An. coluzzii accumulated higher nutrient reserves than An. gambiae, both at the onset of the rainy season and at the onset of the dry season (Tukey’s post hoc test, p < 0.005).
 |
Figure 6 Nutritional reserves (mean ± SE) in newly emerged Anopheles coluzzii and Anopheles gambiae. Different letters indicate significant differences in means (t-test, p < 0.05). |
Discussion
Previous and recent studies have provided compelling evidence that certain members of the An. gambiae s.l. complex can persist over the duration of the dry season in tropical savannahs through mechanisms such as aestivation or long-distance migration [1, 15, 56, 61, 63]. However, the physiological, biochemical, and morphological traits underpinning these survival strategies remain insufficiently explored, particularly under natural field conditions. In this study, we investigated physiological (gonotrophic dissociation, fat-body hypertrophy), biochemical (level of glycogen, sugars, proteins, and lipids), and morphological (body size) traits in mosquito populations from two ecologically distinct sites in Bobo-Dioulasso (Burkina Faso, West Africa), during the rainy season and at the onset of the dry season, with the aim of identifying signatures of adaptations to the harsh period.
Anopheles coluzzii females collected from Soumousso exhibited a significantly higher incidence of gonotrophic dissociation at the onset of the dry season compared to the rainy season. In these females, ovaries remained in the pre-vitellogenic stage of development (up to stage 2 according to Christopher’s classification) despite the ingestion of multiple blood meals, indicating a state of reproductive arrest. These findings are consistent with previous studies reported by Yaro et al. [86], who documented gonotrophic dissociation in An. coluzzii collected indoors during the dry season in an arid region of Mali. In that study, reproductive arrest was not limited to arrested ovarian development, but also involved suppressed oviposition, even in females with fully developed eggs. Similar trends of gonotrophic dissociation had previously been reported in An. arabiensis in the Sudan [65], in hibernating females of An. earlei [32] and in An. punctipennis [58], suggesting broader ecological relevance of this strategy among Anopheles species facing unfavourable environmental conditions.
The results of this study did not show significant seasonal variation in gonotrophic dissociation in either An. gambiae and An. arabiensis. Moreover, a surprisingly high proportion of females from both species exhibited gonotrophic discordance even during the rainy season, when environmental conditions are generally most favourable for reproduction. This has been observed in An. gambiae under insectary conditions, where reduced ovarian maturation was noted compared to An. coluzzii [64]. Anopheles gambiae and An. arabiensis, which typically colonise temporary habitats, may have evolved more plastic reproductive responses than specific dry-season survival strategies. This plasticity is likely shaped by a combination of complex factors, including carryover effects from larval environments such as intense competition for food, exposure to sublethal stressors, or predation pressure. Additionally, certain genotypes within these populations may be inherently more prone to exhibit gonotrophic discordance. The assumption that An. gambiae might be more physiologically sensitive to environmental stressors than An. coluzzii and An. arabiensis cannot be excluded, especially given the well-documented difficulties in establishing stable insectary colonies of the latter species. However, these hypotheses deserve further investigation.
Fat body hypertrophy (characterised by a subcuticular fat layer) and the presence of lipid droplets were observed in females of the three species, with the highest prevalence in An. gambiae. In holometabolous insects, fat body serves as a key metabolic organ, functioning as the principal site for the storage of lipids, glycogen, and protein. Lipid accumulation leads to hypertrophy of the fat body, a condition known to enhance survival in hibernating Culex pipiens (Diptera: Culicidae) females [3]. Moreover, lipids represent the primary energy reserves utilised during insect diapause [41]. In Anopheles messeae, fat body formation during hibernation has been attributed to nutrients derived from either larval feeding or adult blood meals [51]. The accumulation of such reserves is recognised as a prerequisite for diapause induction in many insects [13, 41]. Ultimately, the survival of an individual during adverse conditions, such as the extended dry season, depends largely on its capacity to acquire and store sufficient energy to compensate for resource deficits. Supporting this, lipid accumulation has been reported in Drosophila under nutrient stress [7] and in An. gambiae under desiccation stress [40].
Interestingly, our study also revealed phenotypic variation between the two selected study sites. Although the two localities display similar trends in climatic conditions, they differ markedly in the temporal availability of aquatic habitats. Soumousso represents a typical wet savannah environment, where breeding sites are exclusively rainfall-dependent, resulting in vector breeding activity that closely follows the rainy season. In contrast, Bama is characterised by irrigated rice cultivation, which ensures the presence of permanent breeding sites throughout the year, allowing for continuous mosquito proliferation. The phenotypic variation observed in An. coluzzii between temporary vs. permanent breeding sites likely reflects its remarkable adaptability to anthropogenic environments and underscores its ecological plasticity. The capacity to colonise permanent water bodies may enable An. coluzzii to maintain reproductive activity even during the dry season. Thus, in areas where permanent breeding sites are present year-round, this species could invest either in survival by accumulating nutrient reserves or continued reproduction. Conversely, in areas where oviposition sites disappear during the dry season, a significant proportion of individuals may shift their physiology towards a state of gonotrophic discordance, as suggested by our results.
Newly emerged An. coluzzii accumulated significantly higher levels of lipids, sugars, glycogen, and protein during the transitional period (i.e. at the onset of the dry season) compared to the rainy season. This substantial accumulation of energy reserve likely reflects the enhanced ability of this species to cope with desiccation stress, thereby promoting greater adult survival, as previously demonstrated by Lee et al. [55]. Such physiological conditioning may contribute to its continuous presence in Bama throughout the year [4, 36] and also suggests a potential capacity for aestivation in response to the temporary nature of breeding habitats in Soumousso [56]. However, it should be noted that nutrient reserves in experiment 2 were expressed in mg/mL without standardisation to mosquito body size or weight, which represents a limitation of this study. Consequently, variations between seasons or species may partly reflect differences in mosquito size rather than true physiological differences. Future studies should include size- or mass adjusted biochemical quantification to more accurately assess seasonal or species-specific metabolic strategies.
As expected in our study, females collected at the onset of the dry season exhibited significantly larger body sizes than those collected during the rainy season, particularly in An. coluzzii. Body size was assessed using centroid size, a geometric morphometric measure considered to be a more informative and efficient estimator of body size compared to traditional metrics, such as wing length [52, 53]. The higher body size observed in An. coluzzii supports the general hypothesis that under adverse environments, natural selection would favour larger phenotypes. An alternative explanation for the observed size variation is diapause preparation for aestivation. Benoit and Denlinger [5] indeed demonstrated that diapausing individuals tend to exhibit greater body sizes compared to their reproductively active counterparts. Additionally, it is known that body size at emergence is closely linked to the quality of larval nutrition, and larger individuals are more resistant to desiccation [31, 39]. Therefore, the increased body size observed in An. coluzzii at the onset of the dry season may indicate a physiological adaptation involving the accumulation of substantial energy reserves in anticipation of the dry period.
Based on these results, we cannot definitively separate the assumptions of aestivation and long-distance migration as alternative survival strategies employed by different Anopheles species. Phenotypes such as fat body hypertrophy, documented here to the best of our knowledge for the first time in Anopheles, and gonotrophic dissociation, while commonly associated with aestivation/diapause, are not exclusive indicators of this state and may also occur in individuals preparing for migratory behaviour. It is plausible that the accumulated lipid reserves serve as an energy source for sustained flight, while the observed subcuticular fat deposition in all three species may reflect preparatory physiological adaptations for migration toward more favourable habitats. Future studies integrating ecological tracking methods such as mark-release-recapture, with the identification of molecular or genetic biomarkers, would enhance our understanding of aestivation and migratory behaviours in An. gambiae s.l.
Conclusion
Several physiological, biochemical, and morphological changes are triggered in An. gambiae s.l. mosquitoes at the onset of the dry season in the tropical savannahs of Burkina Faso, West Africa. The observed inter- and intra-specific variability in the expression of these traits likely reflects differential trade-offs in resources acquisition, allocation and utilisation, enabling populations to cope with environmental stressors and ecological constraints associated with seasonal changes. Unravelling the biological mechanisms underpinning the population dynamics of major human malaria vectors in tropical Africa is key to sustainable disease prevention and control, especially in a context of climate change and global warming.
Acknowledgments
We thank Herve L. Somda, Karim Ouedraogo, Ali Ouari, Ilboudo Seini, and Guel Zila, as well as the late Boubakar Nikiema and the late Yeye Pascal for their valuable help during field sampling, rearing, and molecular identification of mosquitoes. This research was funded by the French Agence Nationale de la Recherche through grant No. ANR-08-MIEN-006 to F. Simard, with additional support from IRD/MIVEGEC.
Conflict of interests
The authors declare that they have no competing interests.
Data availability
All data generated or analysed during this study are included in this published article.
Authors contribution statement
KM, RD, RKD and FS conceived the study. WM, KM, KH, performed the experiments. WM drafted the manuscript, which was critically revised by KM, HK, RD, RKD and FS. KM and FS supervised the entire work. All authors read and approved the final version of the manuscript.
Supplementary material
 |
Figure S1: Geographic location of the two study sites in Burkina Faso (West Africa). |
 |
Figure S2: Phenotypes of subcuticular fat body hypertrophy in female Anopheles gambiae s.l., mosquitoes showing the presence of subcuticular fat deposits (A), lipid droplets (B), and absence of lipid reserves (C). |
 |
Figure S3: Climatic variation in temperature (°C), relative humidity (%), and total monthly rainfall (mm) variations at Bama (Burkina-Faso). Monthly maximum, mean, and minimum temperatures (T°, red lines), relative humidity (RH, blue lines), and rainfall (black vertical bars). |
References
- Adamou A, Dao A, Timbine S, Kassogué Y, Yaro AS, Diallo M, Traoré SF, Huestis DL, Lehmann T. 2011. The contribution of aestivating mosquitoes to the persistence of Anopheles gambiae in the Sahel. Malaria Journal, 10, 151. [Google Scholar]
- Arcaz AC, Huestis DL, Dao A, Yaro AS, Diallo M, Andersen J, Blomquist GJ, Lehmann T. 2016. Desiccation tolerance in Anopheles coluzzii: the effects of spiracle size and cuticular hydrocarbons, Journal of Experimental Biology, 219, 1675–1688. [Google Scholar]
- Arrese EL, Soulages JL. 2010. Insect fat body: energy, metabolism, and regulation. Annual Review of Entomology, 55, 207–225. [Google Scholar]
- Baldet T, Diabaté A, Guiguemdé T. 2003. Étude de la transmission du paludisme en 1999 dans la zone rizicole de la vallée du Kou (Bama), Burkina Faso. Cahiers de Santé, 13, 55–60. [Google Scholar]
- Benoit JB, Denlinger DL. 2007. Suppression of water loss during adult diapause in the northern house mosquito, Culex pipiens. Journal of Experimental Biology, 210, 217–226. [Google Scholar]
- Camara M, Caro-Riaño H, Ravel S, Dujardin J-P, Hervouet J-P, De Meeüs T, Kagbadouno MS, Bouyer J, Solano P. 2006. Genetic and morphometric evidence for population isolation of Glossina palpalis gambiensis (Diptera: Glossinidae) on the Loos islands, Guinea. Journal of Medical Entomology, 43, 853–860. [Google Scholar]
- Chippindale AK, Chu TJF, Rose MR. 1996. Complex trade-offs and the evolution of starvation resistance in Drosophila melanogaster. Evolution, 50, 753–766. [Google Scholar]
- Clark MS, Worland MR. 2008. How insects survive the cold: molecular mechanisms-a review. Journal of Comparative Physiology. B, Biochemical, Systemic, and Environmental Physiology, 178, 917–933. [Google Scholar]
- Coetzee M, Craig M, Sueur D Le. 2000. Distribution of African malaria mosquitoes belonging to the Anopheles gambiae complex. Parasitology Today, 16, 74–77. [Google Scholar]
- Coetzee M, Hunt R, Wilkerson R, Della TA, Coulibaly M, Besansky N. 2013. Anopheles coluzzii and Anopheles amharicus, new members of the Anopheles gambiae complex. Zootaxa, 3619, 246–274. [Google Scholar]
- Costantini C, Ayala D, Guelbeogo WM, Pombi M, Some CY, Bassole IH, Ose K, Fotsing J-M, Sagnon N, Fontenille D, Besansky NJ, Simard F. 2009. Living at the edge: biogeographic patterns of habitat segregation conform to speciation by niche expansion in Anopheles gambiae. BMC Ecology, 9, 16. [Google Scholar]
- Dabiré KR, Diabaté A, Paré-Toé L, Rouamba J, Ouari A, Fontenille D, Baldet T. 2008. Year to year and seasonal variations in vector bionomics and malaria transmission in a humid savannah village in west Burkina Faso. Journal of the Society for Vector Ecology, 33, 70–75. [Google Scholar]
- Danks HV. 2006. Key themes in the study of seasonal adaptations in insects II. Life-cycle patterns. Applied Entomology and Zoology, 41, 1–13. [Google Scholar]
- Danks H. 1987. Insect dormancy: an ecological perspective. Ottawa: Biological Survey of Canada (Terrestrial Arthropods). [Google Scholar]
- Dao A, Yaro AS, Diallo M, Timbiné S, Huestis DL, Kassogué Y, Traoré AI, Sanogo ZL, Samaké D, Lehmann T. 2014. Signatures of aestivation and migration in Sahelian malaria mosquito populations. Nature, 516, 387–390. [Google Scholar]
- Denlinger DL. 1986. Dormancy in tropical insects. Annual Review of Entomology, 31, 239–264. [Google Scholar]
- Denlinger D. 2002. Regulation of diapause. Annual Review of Entomology, 47, 93–122. [Google Scholar]
- Denlinger D, Yocum G, Rinehart J. 2012. Hormonal control of diapause. Insect Endocrinology, 1, 430–463. [Google Scholar]
- Detinova T. 1962. Age grouping methods in diptera of medical importance with special reference to some vectors of malaria. Series No. 47. Geneva: World Health Organization Monograph Series. [Google Scholar]
- Diabate A, Baldet T, Chandre F, Akoobeto M, Guiguemde TR, Darriet F, Brengues C, Guillet P, Hemingway J, Small GJ, Hougard JM. 2002. The role of agricultural use of insecticides in resistance to pyrethroids in Anopheles gambiae s.l. in Burkina Faso. American Journal of Tropical Medicine and Hygiene, 67, 617–622. [Google Scholar]
- Diabate A, Brengues C, Baldet T, Dabiré KR, Hougard JM, Akogbeto M, Kengne P, Simard F, Guillet P, Hemingway J, Chandre F. 2004. The spread of the Leu-Phe kdr mutation through Anopheles gambiae complex in Burkina Faso: genetic introgression and de novo phenomena. Tropical Medicine & International Health, 9, 1267–1273. [Google Scholar]
- Diabaté A, Dabiré RK, Heidenberger K, Crawford J, Lamp WO, Culler LE, Lehmann T. 2008. Evidence for divergent selection between the molecular forms of Anopheles gambiae: role of predation. BMC Evolutionary Biology, 8, 5. [Google Scholar]
- Diabaté A, Dabire RK, Kim EH, Dalton R, Millogo N, Baldet T, Simard F, Gimnig JE, Hawley WA, Lehmann T. 2005. Larval development of the molecular forms of Anopheles gambiae (Diptera: Culicidae) in different habitats: a transplantation experiment. Journal of Medical Entomology, 42, 548–553. [Google Scholar]
- Diniz DFA, de Albuquerque CMR, Oliva LO, de Melo-Santos MAV, Ayres CFJ. 2017. Diapause and quiescence: dormancy mechanisms that contribute to the geographical expansion of mosquitoes and their evolutionary success. Parasites & Vectors, 10, 310. [Google Scholar]
- Dryden IL, Mardia KV. 2016. Statistical shape analysis: with applications in R (Second edition). United Kingdom: John Wiley & Sons. [Google Scholar]
- Dujardin J-PAL, Kaba D, Henry AB. 2010. The exchangeability of shape. BMC Research Notes, 3, 266. [Google Scholar]
- Dujardin J-P. 2008. Morphometrics applied to medical entomology. Infection, Genetics and Evolution, 8, 875–890. [Google Scholar]
- Dujardin JP. 2011. Modern morphometrics of medically important insects. Genetics and Evolution of Infectious Diseases, 1, 473–501. [Google Scholar]
- Ebi KL. 2014. Health in the new scenarios for climate change research. International Journal of Environmental Research and Public Health, 11, 30–46. [Google Scholar]
- Eldridge B. 1966. Environmental control of ovarian development in mosquitoes of the Culex pipiens complex. Science, 151, 826–830. [Google Scholar]
- Fouet C, Gray E, Besansky NJ, Costantini C. 2012. Adaptation to aridity in the malaria mosquito Anopheles gambiae: Chromosomal inversion polymorphism and body size influence resistance to desiccation. PloS One, 7, e34841. [Google Scholar]
- Gallaway WJ, Brust RA. 1982. Blood-feeding and gonotrophic dissociation in overwintering Anopheles earli (Diptera: Culicidae) from southern Manitoba. Canadian Entomologist, 114, 1105–1107. [Google Scholar]
- Gatehouse A. 1997. Behavior and ecological genetics of wind-borne migration by insects. Annual Review of Entomology, 42, 475–502. [Google Scholar]
- Gillies MT, Coetzee M. 1987. Anophelines mosquitoes: A supplement to the Anophelinae of Africa south of the Sahara (Afrotropical region). South African Institute for Medical Research, 55, 143. [Google Scholar]
- Gimonneau G, Bouyer J, Morand S, Besansky NJ, Diabate A, Simard F. 2010. A behavioral mechanism underlying ecological divergence in the malaria mosquito Anopheles gambiae. Behavioral Ecology, 21, 1087–1092. [Google Scholar]
- Gimonneau G, Pombi M, Choisy M, Morand S, Dabiré RK, Simard F. 2012. Larval habitat segregation between the molecular forms of the mosquito Anopheles gambiae in a rice field area of Burkina Faso, West Africa. Medical and Veterinary Entomology, 26, 9–17. [Google Scholar]
- Gómez GF, Márquez EJ, Gutiérrez LA, Conn JE, Correa MM. 2014. Geometric morphometric analysis of Colombian Anopheles albimanus (Diptera: Culicidae) reveals significant effect of environmental factors on wing traits and presence of a metapopulation. Acta Tropica, 135, 75–85. [Google Scholar]
- Govoetchan R, Sovi A, Aïkpon R, Salako A, Agbo FO, Asidi A, Akogbéto M. 2013. The impact of oviposition-site deprivation in gravid females of Anopheles gambiae (Diptera: Culicidae) on fecundity, trophic behaviour and life expectancy. International Journal of Tropical Insect Science, 33, 207–215. [Google Scholar]
- Gray E, Bradley T. 2005. Physiology of desiccation resistance in Anopheles gambiae and Anopheles arabiensis. American Journal of Tropical Medicine and Hygiene, 73, 553–559. [Google Scholar]
- Gray EM, Rocca KAC, Costantini C, Besansky NJ. 2009. Inversion 2La is associated with enhanced desiccation resistance in Anopheles gambiae. Malaria Journal, 8, 215. [Google Scholar]
- Hahn DA, Denlinger DL. 2007. Meeting the energetic demands of insect diapause: Nutrient storage and utilization. Journal of Insect Physiology, 53, 760–773. [Google Scholar]
- Hahn D, Denlinger D. 2011. Energetics of insect diapause. Annual Review of Entomology, 56, 103–121. [Google Scholar]
- Hidalgo K, Dujardin J-P, Mouline K, Dabiré RK, Renault D, Simard F. 2015. Seasonal variation in wing size and shape between geographic populations of the malaria vector, Anopheles coluzzii in Burkina Faso (West Africa). Acta Tropica, 143, 79–88. [Google Scholar]
- Hidalgo K, Montazeau C, Siaussat D, Braman V, Trabalon M, Simard F, Renault D, Dabiré RK, Mouline K. 2018. Distinct physiological, biochemical and morphometric adjustments in the malaria vectors Anopheles gambiae and Anopheles coluzzii as means to survive dry season conditions in Burkina Faso. Journal of Experimental Biology, 221, jeb174433. [Google Scholar]
- Hidalgo K, Mouline K, Mamai W, Foucreau N, Dabiré KR, Bouchereau A, Simard F, Renault D. 2014. Novel insights into the metabolic and biochemical underpinnings assisting dry-season survival in female malaria mosquitoes of the Anopheles gambiae complex. Journal of Insect Physiology, 70, 102–116. [Google Scholar]
- Holstein M. 1954. Biology of Anopheles gambiae. Research in French West Africa. Geneva: World Health Organisation Monograph Series. [Google Scholar]
- Huestis DL, Artis ML, Armbruster PA, Lehmann T. 2017 Photoperiodic responses of Sahelian malaria mosquitoes Anopheles coluzzii and An. arabiensis, Parasites & Vectors, 10, 621. [Google Scholar]
- Huestis DL, Lehmann T. 2014. Ecophysiology of Anopheles gambiae s.l.: persistence in the Sahel. Infection, Genetics and Evolution, 28, 648–661. [Google Scholar]
- Huestis DL, Yaro AS, Traoré AI, Adamou A, Kassogué Y, Diallo M, Timbiné S, Dao A, Lehmann T. 2011. Variation in metabolic rate of Anopheles gambiae and A. arabiensis in a Sahelian village. Journal of Experimental Biology, 214, 2345–2353. [Google Scholar]
- Huestis DL, Yaro AS, Traoré AI, Dieter KL, Nwagbara JI, Bowie AC, Adamou A, Kassogué Y, Diallo M, Timbiné S, Dao A, Lehmann T. 2012. Seasonal variation in metabolic rate, flight activity and body size of Anopheles gambiae in the Sahel. Journal of Experimental Biology, 215, 2013–2021. [Google Scholar]
- Jaenson TG, Ameneshewa B. 1991. Prehibernation diet and reproductive condition of female Anopheles messeae in Sweden. Medical and Veterinary Entomology, 5, 243–252. [Google Scholar]
- Jirakanjanakit N, Dujardin J. 2005. Discrimination of Aedes aegypti (Diptera: Culicidae) laboratory lines based on wing geometry. Southeast Asian Journal of Tropical Medicine and Public Health, 36, 858–861. [Google Scholar]
- Jirakanjanakit N, Leemingsawat S, Thongrungkiat S, Apiwathnasorn C, Singhaniyom S, Bellec C, Dujardin JP. 2007. Influence of larval density or food variation on the geometry of the wing of Aedes (Stegomyia) aegypti. Tropical Medicine & International Health, 12, 1354–1360. [Google Scholar]
- Krajacich BJ, Huestis DL, Dao A, Yaro AS, Diallo M, Krishna A, Xu J, Lehmann T. 2018. Investigation of the seasonal microbiome of Anopheles coluzzii mosquitoes in Mali. PLoS One, 13, 3e0194899. [Google Scholar]
- Lee Y, Meneses CR, Fofana A, Lanzaro GC. 2009. Desiccation resistance among subpopulations of Anopheles gambiae s.s. from Selinkenyi, Mali. Journal of Medical Entomology, 46, 316–320. [Google Scholar]
- Lehmann T, Dao A, Yaro AS, Adamou A, Kassogue Y, Diallo M, Sékou T, Coscaron-Arias C. 2010. Aestivation of the African malaria mosquito, Anopheles gambiae in the Sahel. American Journal of Tropical Medicine and Hygiene, 83, 601–606. [Google Scholar]
- Lindsay SW, Parson L, Thomas CJ. 1998. Mapping the ranges and relative abundance of the two principal African malaria vectors, Anopheles gambiae sensu stricto and An. arabiensis, using climate data. Proceedings of the Royal Society of London. Series B: Biological Sciences, 265(1399), 847–854. [Google Scholar]
- Magnarelli L. 1979. Blood-feeding and gonotrophic dissociation in Anopheles Punctipennis (Diptera: Culicidae) prior to hibernation in Connecticut. Journal of Medical Entomology, 15, 278–281. [Google Scholar]
- Mala A, Irungu L, Mitaki E, Shililu J, Mbogo C, Njagi J, Githure J. 2014. Gonotrophic cycle duration, fecundity and parity of Anopheles gambiae complex mosquitoes during an extended period of dry weather in a semi-arid area in Baringo County, Kenya. International Journal of Mosquito Research, 1, 28–34. [Google Scholar]
- Mamai W, Mouline K, Blais C, Larvor V, Dabiré KR, Ouedraogo GA, Simard F, Renault D. 2014. Metabolomic and ecdysteroid variations in Anopheles gambiae s.l. mosquitoes exposed to the stressful conditions of the dry season in Burkina Faso, West Africa. Physiological and Biochemical Zoology, 87, 486–497. [Google Scholar]
- Mamai W, Simard F, Couret D, Ouedraogo GA, Renault D, Dabiré KR, Mouline K. 2016. Monitoring dry season persistence of Anopheles gambiae s.l. populations in a contained semi-field system in southwestern Burkina Faso, West Africa. Journal of Medical Entomology, 53, 130–138. [Google Scholar]
- Masaki S. 1980. Summer diapause. Annual Review of Entomology, 25, 1–25. [Google Scholar]
- Minakawa N, Githure JI, Beier JC, Yan G. 2001. Anopheline mosquito survival strategies during the dry period in western Kenya. Journal of Medical Entomology, 38, 388–392. [Google Scholar]
- Mouline K, Mamai W, Agnew P, Tchonfienet M, Brengues C, Dabire R, Robert V, Simard F. 2012. Physiology and development of the M and S molecular forms of Anopheles gambiae in Burkina Faso (West Africa). Medical and Veterinary Entomology, 26, 447–454. [Google Scholar]
- Omer SM, Cloudsley-Thompson J. 1970. Survival of female Anopheles gambiae Giles through a 9-month dry season in Sudan. Bulletin of World Health Organisation, 42, 319–330. [Google Scholar]
- Omer SM, Cloudsley-Thompson J. 1968. Dry season biology of Anopheles gambiae Giles in the Sudan. Nature, 217, 879–880. [CrossRef] [Google Scholar]
- Patz JA, Campbell-Lendrum D, Holloway T, Foley JA. 2005. Impact of regional climate change on human health. Nature, 438, 310–317. [Google Scholar]
- Reynolds J, Poelchau MF, Rahman Z, Armbruster PA, Denlinger DL. 2012. Transcript profiling reveals mechanisms for lipid conservation during diapause in the mosquito, Aedes albopictus. Journal of Insect Physiology, 58, 966–973. [Google Scholar]
- Rivero A, Agnew P, Bedhomme S, Sidobre C, Michalakis Y. 2007. Resource depletion in Aedes aegypti mosquitoes infected by the microsporidia Vavraia culicis. Parasitology, 134, 1355–1362. [Google Scholar]
- Robert V, Gazin P, Carnevale P. 1987. Malaria transmission in three sites surrounding the area of Bobo-Dioulasso (Burkina Faso): the savanna, a rice field, and the city. Bulletin of the Society of Vector Ecologists, 12, 541–543. [Google Scholar]
- RocklövJ, Dubrow R. 2020. Climate change: an enduring challenge for vector-borne disease prevention and control. Nature Immunology, 21, 479–483. [Google Scholar]
- Roff D. 2002. Life history evolution. Sunderland, Massachusetts: Sinauer Associates, Inc. [Google Scholar]
- Santolamazza F, Mancini E, Simard F, Qi Y, Tu Z, Torre A. 2008. Insertion polymorphisms of SINE200 retrotransposons within speciation islands of Anopheles gambiae molecular forms. Malaria Journal, 7, 163. [Google Scholar]
- Simard F, Ayala D, Kamdem G, Pombi M, Etouna J, Ose K, Fotsing J-M, Fontenille D, Besansky NJ, Costantini C. 2009. Ecological niche partitioning between Anopheles gambiae molecular forms in Cameroon: the ecological side of speciation. BMC Ecology, 9, 17. [Google Scholar]
- Simard F, Lehmann T, Lemasson JJ, Diatta M, Fontenille D. 2000. Persistence of Anopheles arabiensis during the severe dry season conditions in Senegal: an indirect approach using microsatellite loci. Insect Molecular Biology, 9, 467–479. [Google Scholar]
- Sinka ME. 2013. Global distribution of the dominant vector species of malaria, in Anopheles Mosquitoes – New Insights into Malaria Vectors. London, United Kingdom: InTechOpen. pp. 109–144. [Google Scholar]
- Stearns S. 1989. Trade-offs in life-history evolution. Functional Ecology, 3, 259–268. [Google Scholar]
- Tan Q, Feng L, Liu W, Zhu L, Lei C, Wang X. 2016. Differences in the pre-diapause and pre-oviposition accumulation of critical nutrients in adult females of the beetle Colaphellus bowringi. Entomologia Experimentalis et Applicata, 160, 117–125. [Google Scholar]
- Tauber M, Tauber C, Masaki S. 1986. Seasonal adaptations of insects. New York: Oxford University Press. [Google Scholar]
- Tripet F, Touré YT, Taylor CE, Norris DE, Dolo G, Lanzaro GC. 2001. DNA analysis of transferred sperm reveals significant levels of gene flow between molecular forms of Anopheles gambiae. Molecular Ecology, 10, 1725–1732. [Google Scholar]
- Venkatesan P. 2025. WHO world malaria report 2024. Lancet Microbe, 6, 101073. [Google Scholar]
- Vinogradova EB. 2007. Diapause in aquatic insects, with emphasis on mosquitoes, in Diapause in Aquatic Invertebrates Theory and Human Use, Alekseev VR, De Stasio BT, Gilbert JJ, Editors. Springer, Netherlands: Dordrecht. pp. 83–113. [Google Scholar]
- Washino RK. 1977. The physiological ecology of gonotrophic dissociation and related phenomena in mosquitoes. Journal of Medical Entomology, 13, 381–388. [Google Scholar]
- White GB. 1974. Anopheles gambiae complex and disease transmission in Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene, 68, 278–298. [Google Scholar]
- Wiebe A, Longbottom J, Gleave K, Shearer FM, Sinka ME, Massey NC, Cameron E, Bhatt S, Gething PW, Hemingway J, Smith DL, Coleman M, Moyes CL. 2017. Geographical distributions of African malaria vector sibling species and evidence for insecticide resistance. Malaria Journal, 16, 85. [Google Scholar]
- Yaro AS, Traoré AI, Huestis DL, Adamou A, Timbiné S, Kassogué Y, Diallo M, Dao A, Traoré SF, Lehmann T. 2012. Dry season reproductive depression of Anopheles gambiae in the Sahel. Journal of Insect Physiology, 58, 1050–1059. [Google Scholar]
Cite this article as: Mamai W, Mouline K, Renault D, Hidalgo K, Dabiré KR & Simard F. 2026. Phenotypic changes in natural populations of Anopheles gambiae s.l. at the onset of the long dry season in tropical savannahs of Burkina Faso, West Africa. Parasite 33, 9. https://doi.org/10.1051/parasite/2026010.
All Tables
Statistical analyses comparing the effects of season, locality, and species on phenotypic traits in Anopheles mosquitoes. Results present probabilities from Chi-square (χ2) tests for gonotrophic dissociation, Fisher’s exact tests for fat body hypertrophy (lipid droplets and fat deposits), and linear mixed model (LMM) analyses for wing centroid size. Significant differences (p < 0.05) indicate the influence of season (rainy vs. dry), study locality (Bama vs. Soumousso), and interspecific variation (Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis) on these traits.
All Figures
 |
Figure 1 Experimental study design: Experiment 1 (left) and Experiment 2 (right). |
| In the text | |
 |
Figure 2 A left wing belonging to a member of the Anopheles gambiae species complex indicating the location of 12 landmarks used in geometric morphometrics analysis using CLIC software. |
| In the text | |
 |
Figure 3 Ovarian developmental stages in female Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis during the rainy and at the onset of the dry seasons, and across two study sites in Burkina Faso. A = Bama (permanent breeding sites); B = Soumousso (temporary breeding sites). |
| In the text | |
 |
Figure 4 Proportion of Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis females with sub-cuticular fat body hypertrophy collected from two different sites of Burkina Faso. A = permanent (Bama); B = temporary (Soumousso). See Supplementary Material S2 for phenotype description. |
| In the text | |
 |
Figure 5 Wing centroid size in Anopheles coluzzii, Anopheles gambiae, and Anopheles arabiensis collected during the rainy season and at the onset of the dry season at two sites in Burkina Faso. Different letters indicate statistically different p-values. |
| In the text | |
 |
Figure 6 Nutritional reserves (mean ± SE) in newly emerged Anopheles coluzzii and Anopheles gambiae. Different letters indicate significant differences in means (t-test, p < 0.05). |
| In the text | |
 |
Figure S1: Geographic location of the two study sites in Burkina Faso (West Africa). |
| In the text | |
 |
Figure S2: Phenotypes of subcuticular fat body hypertrophy in female Anopheles gambiae s.l., mosquitoes showing the presence of subcuticular fat deposits (A), lipid droplets (B), and absence of lipid reserves (C). |
| In the text | |
 |
Figure S3: Climatic variation in temperature (°C), relative humidity (%), and total monthly rainfall (mm) variations at Bama (Burkina-Faso). Monthly maximum, mean, and minimum temperatures (T°, red lines), relative humidity (RH, blue lines), and rainfall (black vertical bars). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


