| Issue |
Parasite
Volume 33, 2026
|
|
|---|---|---|
| Article Number | 20 | |
| Number of page(s) | 17 | |
| DOI | https://doi.org/10.1051/parasite/2026018 | |
| Published online | 14 April 2026 | |
Research Article
The mosquitoes (Diptera: Culicidae) of Tunisia: updated checklist and new distribution data
Les moustiques (Diptera : Culicidae) de Tunisie : liste actualisée et nouvelles données de répartition
1
Laboratoire des Virus, Vecteurs et Hôtes (LR20IPT02), Institut Pasteur de Tunis, Université Tunis El Manar, Tunis 1002, Tunisia
2
BioSys – EI Schaffner Francis, Steinbach, France
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
2
October
2025
Accepted:
26
February
2026
Abstract
In the context of climate change and rapid environmental changes in the Mediterranean basin, accurate and up-to-date mosquito distribution data are essential to support the control of mosquito-borne diseases, which remain a major public health concern. Here, we report the results of intermittent, nationwide, cross-sectional mosquito sampling missions conducted in Tunisia between 2013 and 2023 to update the Culicidae checklist and document current species distribution of species. A total of 35 species were collected, compared with 49 species previously reported in the Tunisian literature. The primary malaria vectors Anopheles labranchiae and An. claviger sensu stricto were still recorded in the north, while An. multicolor and An. sergentii were found in the south, indicating that vigilance remains necessary following malaria elimination. The most abundant and widespread species were Aedes caspius, Ae. detritus, Culex perexiguus and Cx. pipiens. The latter two are recognised as vectors of pathogens such as West Nile virus and therefore require sustained surveillance. Several mosquito species previously reported in the literature were not detected during our surveys. The invasive species Ae. Albopictus, recently established in Tunisia, requires particular monitoring due to its spread to several regions of the country.
Résumé
Dans un contexte de changement climatique et de modifications environnementales rapides dans le bassin méditerranéen, des données précises et actualisées sur la distribution des moustiques sont essentielles pour lutter contre les maladies transmises par ces insectes, qui constituent un problème majeur de santé publique. Nous présentons ici les résultats de missions d’échantillonnage transversales, menées de manière intermittente et à l’échelle nationale en Tunisie entre 2013 et 2023, afin de mettre à jour la liste des Culicidae et de documenter la distribution actuelle des espèces. Au total, 35 espèces ont été recensées, contre 49 précédemment répertoriées dans la littérature tunisienne. Les principaux vecteurs du paludisme, Anopheles labranchiae et An. claviger sensu stricto, ont été observés dans le nord du pays, tandis qu’An. multicolor et An. sergentii ont été trouvés dans le sud, ce qui indique que la vigilance reste nécessaire après l’élimination du paludisme. Les espèces les plus abondantes et les plus répandues étaient Aedes caspius, Ae. detritus, Culex perexiguus et Cx. pipiens. Ces deux dernières espèces sont reconnues comme vecteurs de pathogènes tels que le virus du Nil occidental et nécessitent donc une surveillance continue. Plusieurs espèces de moustiques précédemment décrites dans la littérature n’ont pas été détectées lors de nos enquêtes. L’espèce invasive Aedes albopictus, récemment implantée en Tunisie, requiert une surveillance particulière en raison de sa propagation dans plusieurs régions du pays.
Key words: Checklist / Culicidae / Tunisia / North Africa / Distribution / Taxonomy / Vector surveillance
Edited by: Jérôme Depaquit
© A. Ouni et al., published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The family Culicidae, known as mosquitoes, of the Diptera order and Nematocera suborder, comprises about 3,583 described species that are globally distributed [57]. Only a small fraction of mosquito species play a role in transmitting pathogens to humans and animals. According to the World Health Organization (WHO), mosquito-borne diseases account for about 17% of all infectious diseases [168]. Mosquito species of the genera Aedes and Culex play a principal role in the transmission of several viruses and Filariae. Anopheles mosquitoes transmit malaria parasites and are responsible for the most serious mosquito-borne disease globally; they cause thousands of deaths, primarily in Africa [169].
In Tunisia, little is known about mosquito fauna. Records of mosquito species collected in the country are found in the few papers published around the mid-20th century [25, 64, 101, 120]. Additional studies are reported in more recent publications [7, 18, 70, 144]. The invasive species Aedes albopictus was reported in Tunisia subsequently [8, 12]. The literature cites 49 mosquito species belonging to 6 genera (Aedes, Anopheles, Culex, Culiseta, Orthopodomyia and Uranotaenia) [12, 17, 108]. Meanwhile the classification of the tribe Aedini underwent a major taxonomic change in 2000 [98] and in several subsequent articles [99, 100, 165], leading to some confusion regarding the identification and use of species names. Moreover, some species may disappear and others emerge as a result of climate change, environmental modifications, demographic growth and urbanisation, which have a significant impact on water bodies, including wastewater.
Earlier publications were particularly interested in the Tunisian Culicidae fauna solely because of the role played by Anopheles in the transmission of malaria parasites [24]. In the early 20th century, several epidemics of malaria, which was endemic in the country, were caused by Plasmodium falciparum [4, 24]. Malaria was officially eradicated in 1979 thanks to the national malaria eradication programme launched in Tunisia in 1966 with the support of WHO [24, 25]. Its eradication lowered interest in mosquito and mosquito-borne disease studies in Tunisia.
Today, its role as an important transit country in North Africa for human travel, livestock trade and migratory birds puts Tunisia at high risk for the (re-) emergence of arboviruses. Importantly, as part of the geographical unit separating sub-Saharan Africa from Mediterranean Europe, Tunisia has a range of environmental zones that can support rich diversity and the occurence of potential mosquito vectors capable of transmitting several pathogens to animals and humans. The precise identification of mosquito species and knowledge of their distributions and bio-ecology is therefore important to support the surveillance and control of mosquito-borne diseases [26, 57].
To update the mosquito fauna and their distribution, we report the results of several cross-sectional mosquito field studies across the country during 2013–2023. Carried out from the north to the south of the country, these surveys sought to investigate as many mosquito habitats as possible.
Materials and methods
Study area
Tunisia is a North African country (33°58′48″ N; 9°32′24″ E) bordered by the Mediterranean Sea to the north and east, Algeria to the west, and Libya to the south-east. The country is mountainous in the northwest; the eastern Mediterranean coast is a large plain, whereas the south is hot and dry, progressively merging into the Sahara. Based on the bioclimatic classification of Emberger (1960) [43] and Gounot’s (1995) [50], Tunisia can be divided from north to south into five main bioclimatic zones (Fig. 1): (i) humid (annual rainfall ranging from 800 to 1 200 mm/year), subhumid (600–800 mm/year), semi-arid (300–600 mm/year), arid (100–300 mm/year) and Saharan or desert zones (up to/less than 100 mm/year).
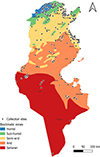 |
Figure 1 Bioclimatic map of Tunisia showing mosquito sampling locations (2013–2023), produced using QGIS Software (Lima 3.32.3). |
Mosquito collection and identification
Mosquito collection was carried out over 10 years (2013–2023). Several cross-sectional investigations of randomly selected potential breeding sites (stagnant water) were conducted in the five bioclimatic zones of the country and focused mainly on aquatic immature stages of mosquitoes (larvae and pupae). These surveys were conducted across different regions during several field missions, with variable sampling intensity (Fig. 1). Then, following the West Nile fever outbreak in Tunisia in 2012, field missions were intensified to identify the mosquito vectors involved. In 2020, field activities were reduced due to the COVID-19 pandemic, resulting in fewer surveys. An additional cross-sectional survey along a north-south transect was carried out in 2017 as part of the VectorNet project (contracted by the European Centre for Disease Prevention and Control [ECDC] and the European Food Safety Authority [EFSA]) which supports the collection of distribution data on vectors of pathogens related to both human and animal health. Sampling sites were selected to cover the main Tunisian bioclimatic zones and the majority of larval habitats (Fig. 1), whether natural (wadis, sebkhas, ponds, streams, swamps, tree holes, etc.) or artificial/man-made (tyres, channel, puddles, etc.). These were chosen based on the national hydrological map and on the information provided by the environmental health officers (Ministry of Health) of each locality. Humid regions receive high levels of rainfall and consequently have more stagnant water, which provides potential breeding sites; therefore, these regions were surveyed more frequently. A subset of key sites was revisited over multiple years to confirm the presence of mosquito species.
We applied a standardised procedure for sampling mosquito larvae in stagnant water, as described in WHO technical manuals [167] and commonly used in entomology, public health, and vector surveillance. Using a standard long-handled 500 mL dipper, mosquito immatures were collected from different breeding sites (natural and human-made water collections), including wadi banks, swamps, pools, drainage sites, catch basins and ditches. The standard dipper was immersed approximately ten times at the edge of large breeding sites. For small habitats, such as tree holes and water vessels, we used a 50 mL pipette with a rubber bulb to aspirate the water. Only 3rd or 4th larvae stages were preserved in ethanol (70%) for morphological identification. First and second stage larvae and pupae were kept alive until obtaining 4th larval stages or adults for accurate species identification.
In addition to collecting immature stages, we trapped adult mosquitoes during the warm season (May–November) using two types of traps: (i) CDC miniature light traps (John W. Hock Co., Gainesville, FL, USA) with dry ice as a source of CO2, and (ii) BG-Sentinel and BG-Lure traps (Biogents, Regensburg, Germany). CDC light traps were installed near animal shelters, dwellings and larval habitats and generally operated from approximately 6 pm to 6 am to cover nocturnal activity. BG-Sentinel traps were placed close to human habitations to collect adult mosquitoes active during the day, mainly Ae. albopictus, usually between 7 am and 5 pm. Depending on mission objectives and site accessibility, up to eight CDC light traps per site per night and up to three BG-Sentinel traps per site per day were deployed for one to three consecutive nights, with placement adapted to local conditions (near dwellings, animal shelters and/or productive larval habitats).
The captured larvae and adults were observed under a binocular microscope. Specimens were identified to the species level using standard dichotomic keys [123, 124] and a computer-aided key [18]. Field observations were considered for biting behaviour of adults. While biting, mosquitoes were caught in tubes (crackers) and kept alive for identification.
Larval habitats characterisation
During sampling, breeding habitats were classified as natural or artificial and characterised according to the depth and the vegetation, and geographic coordinates were recorded using a GPS (Garmin-eTrex 30). Physicochemical parameters (salinity, pH and temperature) were measured in situ using portable meters (Starter Pen Meters ST20S and ST10; OHAUS, Parsippany, NJ, USA) when possible. However, measurements were not systematically performed at all breeding sites due to logistical constraints and site accessibility.
Results and discussion
Characterisation of larval breeding sites
We collected and identified approximately 30,000 mosquito specimens during these decade-long successive field studies throughout the Tunisia’s five bioclimatic zones. Specimens included around 26,300 immature stages collected from breeding sites and 3,700 adults caught by CDC and BG-Sentinel traps and those obtained from pupae. Mosquito larvae were found in a variety of water collections. Among the 2,425 water bodies surveyed for the presence of mosquito larvae, 1,800 were found to be positive for mosquito larvae. Negative stagnant waters were often highly eutrophic (polluted, covered by duckweed, etc.), contaminated by industrial effluents in some wadis, or colonised by Gambusia spp. (introduced in the past for the control of mosquito larvae). Some larval breeding sites, mainly in urban areas, were treated with insecticides by health authorities.
Our surveys highlight a variety of mosquito species occupying diverse larval habitats. Mosquito breeding sites differed from one bioclimatic zone to another. Wadis and tree holes were the most common natural breeding sites in the north, while in the center and the south, sabkhas were the main habitat. In urban environments, open-air canals (which frequently contain greywater) often serve as the primary larval breeding grounds. The prospected larval habitats included artificial (canals, pools) and natural (wadi, sabkhas, swamps) sites with temporary (pools, unused tyres), permanent (sabkha) or semi-permanent (seasonal ponds), depending on their stability (see Supplementary Table 1 for details).
In the investigated habitats, 12 of all identified mosquito species were found in all bioclimatic zones and thus considered to be common, such as Cx. pipiens (see Supplementary Table 1 for details). These species exhibit a wide geographical distribution, indicating their adaptability to diverse environmental conditions. Conversely, other mosquito species were restricted to specific bioclimatic zones such as An. plumbeus which occurs exclusively in the humid northern regions, while other species such as An. sergentii were found exclusively in the southern region where they were collected mainly in the watercourses of the oases (see Supplementary Table 1 for details). Specific habitats such as tree holes and rock pools were occupied by specialised mosquito species.
In the investigated habitats, water temperature ranged from 15 to 35 °C, salinity from 0.1 to 66.2 g/L (NaCl), and pH from 7.08 to 10.8, reflecting a gradient from freshwaters to hypersaline sebkhas and salt marshes. Depths ranged from 5 cm to 2 m.
The vegetation in mosquito breeding sites varied widely. In wadis and hill lakes, the Characeae and Potamogeton were the dominant vegetation, submerged, while salt marshes and sabkhas were covered with halophyte plants, such as Salicornia. Sabkhas subjected to flooding with rainwater were covered by Phragmites, Juncus and Typha.
Mosquito checklist
During our surveys we collected and identified around 30,000 mosquito specimens from 35 species belonging to 6 genera: Anopheles (10 species), Aedes (12 species), Culex (9 species), Culiseta (2 species), Orthopodomyia (1 species) and Uranotaenia (1 species) (see Supplementary Table 2 for details). The Culex genus was most represented in our collection (66%), followed by Aedes (17%), Culiseta (9%), Anopheles (6%) and Uranotaenia (2%), and only a few specimens of Orthopodomyia.
Species identified during the current investigation
(See Supplementary Tables 1 and 2 for details)
Subfamily Anophelinae Grassi, 1900
Genus Anopheles Meigen, 1818
Of the 13 Anopheles (An.) species mentioned in the literature, only 10 were identified during our survey: An. algeriensis, An. cinereus, An. claviger, An. labranchiae, An. marteri, An. multicolor, An. petragnani, An. plumbeus, An. sergentii and An. ziemanni. These species were all reported in neighbouring Algeria [146] and Morocco [149]. While Anopheles coustani, An. dthali and An. superpictus have been reported in Tunisia, we did not find them during our surveys.
Anopheles (Anopheles) algeriensis Theobald, 1903
In both humid and subhumid zones, we collected An. algeriensis alongside An. labranchiae; in arid areas, it was associated with An. sergentii (Fig. S1). We found larvae in swamps, slow-moving streams, marshes, wadi banks, ponds and hill lakes overgrown with vegetation. The breeding sites were typically shaded and the water in them was consistently fresh, although it was occasionally saline.
The species was observed in Algeria [52], Morocco [72, 150] and Libya [17] as well. It is also present in Europe and elsewhere, including Afghanistan [59]. The information on the biting behaviour of An. algeriensis adults is contradictory, probably because of the rarity of observations. However, during field visits at dusk in Kebili (southern region), intense host-seeking activity of An. algeriensis was observed in the immediate vicinity of breeding sites.
Anopheles (Cellia) cinereus Theobald, 1901
The results of our survey indicate that An. cinereus is present in all bioclimatic zones of Tunisia (Fig. S2). The species has a wide distribution, and immatures were found in wadi banks, swamps, ponds, pools, drainages and catch basins, and can be collected on the surface of Spirogyra mats. It is typically found in association with An. labranchiae in the north and An. sergentii in the south. Previously identified as An. hispaniola, this species was cited as Anopheles (Cellia) cinereus hispaniola by Theobald 1903 [18]. Several authors have reported the presence of An. cinereus in Algeria and in Morocco [9, 152].
Anopheles (Anopheles) claviger (Meigen, 1804)
Anopheles claviger sensu stricto was identified exclusively in the Tunisia’s northwest humid zone, where the cool climate is typical of oak forests [36°52′0.85″ N; 8°42′51.66″ E]; it is considered to be rare in Tunisia. Larvae were collected with very low density from ponds that were densely vegetated with aquatic plants. Anopheles claviger is also found in mountainous regions of the Maghreb, including Morocco and Algeria, but its presence in Libya requires confirmation [108, 123, 152]. According to Robert et al. (2019) [108], An. claviger is present from Afghanistan to England and in the northernmost areas of Europe, including Sweden and Norway. Although it was considered to be a malaria vector in the Middle East, its vectorial role is considered to be neglected in North Africa due to its rarity [88]. Anopheles claviger sensu stricto is morphologically similar to its sibling species An. petragnani, making differentiation difficult. Some earlier records of An. claviger might therefore belong to An. petragnani.
Anopheles (Anopheles) labranchiae Falleroni, 1926
Anopheles labranchiae is the only species in Tunisia of the Maculipennis Complex. Anopheles labranchiae can be found in areas up to 1,900 m in elevation, with a distribution extending from coastal areas to the Sahara in western Palaearctic regions. During our survey, we collected this species in several habitats (wadis, hill lakes, swamps, ponds and abandoned quarries) from the north of the country towards the center, up to Sidi Bouzid (in Oued Leben) (Fig. S3). The species is frequently encountered in humid, sub-humid, and semi-arid areas, although it is rare in arid regions. The larvae of this mosquito prefer stagnant, fresh, unpolluted waters exposed to sunlight, with vegetation dominated by Ranunculus and Ceratophyllum. The submerged vegetation typical of its habitats is mainly composed of Potamogeton and members of the Characeae. The habitats are often bordered by tamarisk (Tamarix spp.) trees. The current distribution of this species suggests that this mosquito has disappeared from several areas, probably due to environmental changes leading to the loss of suitable habitats. Indeed, this species, sometimes reported as An. maculipennis, was also found in coastal areas where urbanisation has modified the environment. During our field investigations, biting of humans was observed, notably in northern areas near low-water wadis or hill lakes.
Anopheles labranchiae was considered to be a major malaria vector in the north of Tunisia [13, 14, 25]. Several studies have confirmed that this species can transmit Plasmodium falciparum [110, 164]. The regions where this species is present can therefore be considered high-risk sites for the re-emergence of malaria [2].
Anopheles (Anopheles) marteri Sénévet & Prunnelle, 1927
Anopheles marteri is a zoophilic species that we observed in the forested regions of northwest Tunisia, frequently associated with An. petragnani. Its larvae were typically found in freshwater springs and streams in Sidi Mechreg [37°9′36.35″ N; 9°7′1.45″ E]. Anopheles marteri is considered to be rare in Tunisia and given its strong preference for animal hosts, it does not play a role in the transmission of human disease pathogens [32]. First described in Algeria, this species is also present in several Mediterranean countries, particularly in the mountainous areas of North Africa [48, 63, 118].
Anopheles (Cellia) multicolor Cambouliu, 1902
Anopheles multicolor is typically a halophilic species that we rarely observed outside hyper-salty environments in Tunisia. We collected the larvae mostly in the brackish waters of arid, Saharan areas and around oases, where it is associated with An. sergentii (Fig. S4). The species tolerates high salt concentrations but is never found in puddles near the sea and sometimes breeds in organically rich water. It is also typically found in arid and semi-arid areas throughout North Africa from Morocco to Egypt, and as far east as Pakistan [57]. Adults are abundant at the end of summer and the beginning of autumn and have a wide range of dispersion. Females are mainly exophilic and zoophagic, but they also bite humans. The species was incriminated in the transmission of malaria in the south of Tunisia [25] and also suspected as a vector of P. falciparum in Egypt [42, 65].
Anopheles (Anopheles) petragnani Del Vecchio, 1939
During our field investigations in northern Tunisia, we found An. petragnani specifically in the oak forests [36°47′59.87″ N; 8°40′46.22″ E]. This species exhibits a marked preference for bodies of cool, clear water with abundant floating vegetation, often cohabiting with other forest-dwelling mosquito species. Previous reports documented An. petragnani in coastal regions [18, 84]. Its presence in the north suggests that its suitable habitats are restricted to the interior mountainous areas of Tunisia. Reports from northeastern Algeria (Souk Ahras) support the affinity of this mosquito for similar mountainous zones [52].
Anopheles (Anopheles) plumbeus Stephens, 1828
Anopheles plumbeus is a rare species that we found in the forests of northwestern Tunisia. Its larvae breed in water that accumulates in the holes of cork oak trees (Quercus suber) (Fig. S2). This species was reported in Algeria [119] and is abundant throughout Europe and can be found as far east as Iran [112]. During our prospections, numbers of aggressive females were observed near larval breeding sites. The species is known to be aggressive towards humans, mammals, birds and reptiles [19]. Some authors suggest that An. plumbeus can act as a vector of Plasmodium falciparum [113, 145]. Experimentally, it can transmit Plasmodiidae, Flaviviridae and Filariae [79, 81, 113].
Anopheles (Cellia) sergentii (Theobald, 1907)
During our surveys, Anopheles sergentii, an anthropophilic species, was identified in clear fresh or slightly brackish waters, including wadi beds and oasis stream channels, along with other species of the genus Anopheles (Fig. S2). Although historical records attest the presence of this species in semi-arid and arid areas of Tunisia, our results suggest a reduction in its habitat towards arid and pre-Saharan zones. In North Africa, An. sergentii was listed in Libya [66], Algeria [53] and Morocco [151]. It has a wide distribution from Africa to Asia [138] (Sinka et al., 2010) especially in arid zones. This mosquito is considered to be the main vector of malaria in southern Tunisia [25, 144]. Its vector role is also confirmed in Morocco [45], Algeria [97] and Egypt [78].
Anopheles (Anopheles) ziemanni Grünberg, 1902
Anopheles ziemanni is a rare species that we collected in northern Tunisia, where it coexists with An. labranchiae. Anopheles ziemanni larvae typically develop in large, vegetation-littered wet fields like marshes and the grassy edges of large ponds [36°57′ N; 9°0′ E]. Initially classified as a subspecies of An. coustani due to the indistinguishable larvae, it was subsequently elevated to full species status [63]. This change explains why older records may incorrectly refer to An. coustani in the Maghreb, where it is present only in Algeria [122]. The presence of An. ziemanni in Libya needs confirmation [96, 108]. In North Africa, An. ziemanni does not appear to play a significant role in the transmission of human pathogens, although it may serve as a vector for Filariae in animals [67].
Subfamily Culicinae Meigen, 1818
Genus Aedes Meigen, 1818 [Tribe Aedini Neveu-Lemaire, 1902]
Sixteen species of the genus Aedes were cited in Tunisia. Of these, only 12 were identified during the present survey: Ae. albopictus, Ae. berlandi, Ae. caspius, Ae. coluzzii, Ae. detritus, Ae. dorsalis, Ae. echinus, Ae. geniculatus, Ae. mariae, Ae. pulcritarsis, Ae. vexans and Ae. vittatus. Among the Aedes species mentioned in the literature, we did not find Ae. aegypti, Ae. albineus, Ae. cinereus or Ae. zammiti.
Aedes (Stegomyia) albopictus (Skuse, 1894)
Aedes albopictus (Asian tiger mosquito) was recently identified for the first time in Tunisia [12] around Carthage, a city 20 km north of the Tunisian capital, Tunis. Following this initial discovery, the distribution area of the species proved to encompass several districts of Tunis. We subsequently noted its presence in the northern region of Bizerte (60 km northwest of Tunis) and later in the southern region of Sousse (120 km south of Tunis) in 2023 (Fig. S5).
This species has been collected from urban areas, particularly gardens, cemeteries and rubbish tips. Known for its resilience and adaptability, Ae. albopictus colonises artificial habitats such as used tyres and small water containers with fresh water.
It is a proven vector of several arboviruses, including chikungunya, dengue and Zika viruses, and has been experimentally proven to be capable of transmitting many other arboviruses, including Japanese encephalitis, Rift Valley fever and West Nile viruses [11, 68, 81, 95, 140, 143]. In Tunisia, it has not yet been implicated in disease transmission. The rapid spread of Ae. albopictus in the Americas, Europe and Africa in recent decades highlights its invasive potential [80].
Aedes (Ochlerotatus) berlandi Séguy, 1921
Aedes berlandi is a rare species of phytotelma mosquito found in association with Ae. echinus and An. plumbeus in northern Tunisia’s humid, forested regions. Its larvae were collected from the water-filled cavities, predominantly of oak trees (Fig. S6); the water is typically alkaline and rich in organic matter and tannins. The distribution of Ae. berlandi is concentrated primarily in the western Mediterranean region, with additional reports from Algeria, France, Italy, Morocco, Portugal and Spain [86, 106, 108].
Aedes (Ochlerotatus) caspius (Pallas, 1771)
Aedes caspius is one of the most widespread and abundant mosquito species in Tunisia. It was collected during this study, usually at high densities, in all bioclimatic zones (Fig. S7). This halophilic species is commonly found in coastal areas in sabkhas, salt marshes and ponds formed by rainwater or water discharged from food factories, as well as in artificial habitats such as pools and ditches. It is considered to be highly adaptable to saline and brackish water environments, one factor that has contributed to its wide distribution. This mosquito was found in water bodies ranging from freshwater to brackish environments, such as coastal sebkhas (1–59.5 g/L NaCl; pH ~7–8.9), illustrating its broad salinity tolerance.
A comparison of earlier data with our monitoring findings showed a reduction in the number of larval habitats of this species due to increasing urbanisation, particularly in coastal areas. The species has been extensively documented in North Africa, with records extending from Algeria [119], Morocco [48] and Libya [108]. Globally, the species is distributed throughout Europe and the Middle East. From a public health perspective, Ae. caspius is a significant vector for various viruses, including Rift Valley fever virus, and has been proposed as a vector for Francisella tularensis [37, 38, 61].
Aedes (Ochlerotatus) coluzzii Rioux, Guilvard & Pasteur, 1998
Aedes coluzzii is closely related to Ae. detritus, from which it cannot be distinguished morphologically [104] (Rioux et al., 1998). We recently confirmed its presence in Tunisia using primers based on ITS2 rDNA sequences (data in preparation), based on specimens collected from sebkhat Korba (northeastern Tunisia) [36°35′2.04″ N; 10°52′10.52″ E], associated with Ae. detritus. This confirms the observations of [104] Rioux et al. (1998) of this species in certain sites in Tunisia. It is in fact genetically isolated and shows a preference for hyperhaline environments, making it particularly well adapted to arid zones. Aedes coluzzii has also been reported in Algeria [73] and Morocco [151].
Aedes (Ochlerotatus) detritus (Haliday, 1833)
Aedes detritus is a halophilic species that is frequent and abundant in Tunisia and often develops in the same habitat as Ae. caspius. We collected it in all bioclimatic zones in Tunisia, where it is most commonly found in sabkhas and coastal salt marshes (Fig. S8). It was also observed to breed in habitats such as wadi banks and swamps, and in artificial water sources such as pools, unused wells, drainages, catch basins, and ditches, with higher population densities in stagnant water with high salinity, particularly along coastal and arid zones. Our physicochemical data showed that Ae. detritus larvae were recorded across a wide salinity gradient mainly in coastal sebkhas and salt marshes (13–66.2 g/L NaCl), with pH ranging from 7.10 to 8.55 and a temperature from 25 to 32 °C, confirming their strong halophily and tolerance to highly mineralised and organically enriched waters. Aedes detritus is a Palearctic species that is also commonly found in northern Europe, particularly in mesohaline coastal habitats [85]. Its southern range spreads out in North Africa where it is found in various saline environments [16].
It is suggested as a vector of myxomatosis virus [62] and shown to transmit chikungunya virus [154], Rift Valley fever virus [87] and West Nile virus [10] under laboratory conditions.
Aedes (Ochlerotatus) dorsalis (Meigen, 1830)
Aedes dorsalis was recorded in northern and eastern Tunisia in 2019 (Nabeul, Bizerte, Sousse, Tunis, Ben Arous). Only a few larvae were detected, mainly in coastal and peri-urban saline/brackish habitats (sebkha margins and shallow pools <0.5 m), with salinity ranging from 0.1 to 11.3 g/L NaCl and near-neutral pH (7.0–7.3). These sites were associated with halophytic vegetation (e.g. Sarcocornia fruticosa, Juncus maritimus) and larvae were sometimes found alongside Ae. caspius and Ae. detritus. Morphologically it is challenging to distinguish Aedes dorsalis from Ae. caspius in the larval stage, but more conclusive at the adult stage. Aedes dorsalis is present throughout northern Europe and has also been reported in Morocco [16] and Egypt [116].
Aedes (Dahliana) echinus (Edwards, 1920) and Aedes (Dahliana) geniculatus (Olivier, 1791)
Aedes echinus and Ae. geniculatus are both phytotelmic species found mainly in tree holes in the cork oak forests of northwestern Tunisia [36°47′ N; 8°40′ E]. Aedes geniculatus larvae have also been collected in some containers holding organic and tannin-rich water [37°12′39.20″ N; 9°33′32.98″ E]. Both species are considered to be rare in Tunisia, where they are usually restricted to forested areas with sufficient tree cover. These species have a wide Palearctic distribution from Norway to Morocco and from Portugal to Kazakhstan [33].
Aedes (Ochlerotatus) mariae (Sergent & Sergent, 1903)
This species was found in rock pools on the rocky northern Tunisian coast where it emerges after seawater floods rock pools. Immatures develop in these salty rock holes (we observed a salinity of 39 g/L NaCl), filled by sea spray [37°17′59.33″ N; 9°33′48.25″ E]. These sites are generally enriched with organic matter (dead algae, phytoplankton, various organic debris, etc.). The larvae can often tolerate wide variations in temperature. While it remains relatively rare, Aedes mariae is nonetheless found around the western Mediterranean basin, especially from Morocco [5, 151] to its northeastern limit in Tunisia [84].
Aedes (Ochlerotatus) pulcritarsis (Rondani, 1872)
Larvae of Aedes pulcritarsis were collected in both humid [37°20′10.19″ N; 9°40′6.99″ E] and subhumid zones [37°4′2.95″ N; 11°1′35.75″ E]. This rare species breeds primarily in the tree holes of the cork oak forests of northern Tunisia. It is frequently found in association with other arboreal species, such as Orthopodomyia pulcripalpis, Ae. echinus, Ae. berlandi and Ae. geniculatus. Aedes pulcritarsis, originally described from specimens collected in Italy, is distributed throughout the Palearctic region and has also been reported in Algeria [119] and Morocco [48]. The species is not known to play a role in disease transmission and therefore poses no public health threat [90].
Aedes (Aedimorphus) vexans (Meigen, 1830)
During our field investigations, the multivoltine mosquito species, Aedes vexans, was mainly observed along the banks of richly vegetated wadis and shallow temporary pools that form after seasonal rains. These habitats are located in central Tunisia (Sousse and Kairouan governorates), and provide a favourable local environment for this species (Fig. S6). Aedes vexans has been reported in Morocco [48] and Libya [124]; its range extends to the Palaearctic, Nearctic, Eastern and Australian regions [34]. The development cycle of Ae. vexans is rapid, and adults are capable of long-distance travel, making them a notable nuisance. During our study, this species was mainly found in springs. The females can live up to two months and feed on a wide range of hosts, biting humans, livestock, and birds throughout the day, especially at sunset. Aedes vexans is of medical interest because it has demonstrated laboratory vector competence for West Nile virus and its long-standing association with Tahyna virus and Rift Valley Fever virus circulation, supporting its potential role as a bridge vector where it occurs [34, 91, 170],
Aedes (Fredwardsius) vittatus (Bigot, 1861)
During our survey, Aedes vittatus was recorded in central Tunisia, particularly in the wadis of the arid Sidi Bouzid and the oasis streams of Tozeur. In contrast, a single earlier record documented this species in northeastern Tunisia (Kelibia) [155] (Fig. S6). Our result suggests that the range for Ae. vittatus in Tunisia could be broader, extending from the semi-arid central regions to oasis habitats in the south. In North Africa, Ae. vittatus has been collected from Morocco [48]. It is present in the Mediterranean basin, the Ethiopian region and the eastern region [112, 142]. This species is a competent vector for several viruses, including chikungunya virus, dengue virus, West Nile virus, yellow fever virus, and Zika virus. Its ability to transmit these viruses has been demonstrated primarily in laboratory settings [35, 89, 91, 92, 141].
Genus Culex Linnaeus, 1758 [Tribe Culicini Meigen, 1818]
In the present study, we identified 9 of the 12 documented species of the genus Culex found in Tunisia: Cx. deserticola, Cx. hortensis, Cx. impudicus, Cx. laticinctus, Cx. mimeticus, Cx. perexiguus, Cx. pipiens, Cx. pusillus and Cx. theileri. All of these species have been recorded in neighbouring countries Algeria [82] and Morocco [151]. Of the species cited in much earlier literature, Cx. antennatus, Cx. territans and Cx. univittatus were not found during our surveys.
Culex (Maillotia) deserticola Kirkpatrick, 1925
Culex deserticola was collected predominantly in central Tunisia, particularly in pre-Saharan regions (Fig. S9). Culex deserticola is highly adapted to arid environments and typically breeds in wadi beds and pools with sandy bottoms, where the water is fresh and clear and vegetation is limited. The larvae are found most commonly during the winter and spring seasons. The species continues to be considered rare overall with a distribution that extends from northern Africa to Iran [56].
Culex (Maillotia) hortensis Ficalbi, 1889
During our investigation, we collected Cx. hortensis in the north during the spring and autumn, seasons during which the species is active. Larvae were found only once in a habitat [36°46′25.03″ N; 8°41′5.09″ E] with cool, fresh water and without significant vegetation cover. Culex hortensis is considered to be rare in Tunisia, with populations restricted mainly to higher elevations and cooler environments. The species is widely distributed throughout the North African countries, including Algeria [124] and Morocco [48]. As females predominantly feed on amphibians and reptiles, the species has no significant public health implications [109].
Culex (Neoculex) impudicus Ficalbi, 1890
During our surveys, we collected Cx. impudicus in humid and sub-humid regions of Tunisia where they were found in small, shaded ponds with cool, clear fresh water (Fig. S9). It has distinct seasonal activity; its larvae are typically found from March to October. Culex impudicus is considered to be common in northern Tunisia where most populations are concentrated in forested areas, where suitable habitats remain relatively undisturbed. This mosquito is known for being batracophilic, feeding mainly on amphibians and other cold-blooded vertebrates, and poses no significant public health risk to humans [6]. In North Africa, it has been documented in Algeria and Morocco [77, 88]. It is also widely distributed across the Mediterranean region, extending as far east as Iran [108].
Culex (Culex) laticinctus Edwards, 1913
During our investigations, Cx. laticinctus was collected mainly in Tunisia’s arid and Saharan regions (Fig. S9). We also detected the species further north (e.g. near Hammamet), suggesting a broader distribution than previously reported. It was found at high densities along wadi banks and streams with slightly saline water. Where physicochemical data were available, Cx. laticinctus was detected in low-salinity inland habitats (≈2.0–2.8 g/L NaCl; pH ~7.6 where measured), such as wadi-bed depressions. Although previously considered to be relatively common, particularly in arid regions, our data suggest more restricted current distribution. This species is not considered a primary vector of human pathogens. The distribution of Cx. laticinctus, primarily in the Mediterranean region, ranges from the Canary Islands to the Middle East [105].
Culex (Culex) mimeticus Noè, 1899
Our findings show that Cx. mimeticus is present in localities in northern Tunisia (Fig. S9). It was previously reported as being restricted to the northwest [18]. Its distribution is more closely associated with lowland areas rather than specific bioclimatic zones. The larvae develop in wadis and shallow temporary pools where the water is generally fresh and cool, and commonly colonised by filamentous algae. They are usually found in low numbers, making this species rare in Tunisia. Culex mimeticus is a montaneous species in the Mediterranean subregion and eastern region [139]. It has also been reported in North Africa, including Algeria and Morocco [56, 82, 151]. Adult females may seek refuge in houses, but bite neither humans nor other mammals.
Culex (Culex) perexiguus Theobald, 1903
Culex perexiguus was one of the key species identified during our survey, with collections spanning all bioclimatic zones of Tunisia (Fig. S10). This primarily ornithophilic species breeds in both natural and artificial habitats. While it feeds mostly on birds, records exist of it entering homes and biting humans at night. In habitats where physico-chemical measurements were taken, Cx. perexiguus was primarily associated with waters having salinity ranging from 0.5 to 2.6 g/L NaCl and pH values of 7.08–8.9, including sites fed by springs and canals connected to sebkhas.
The species is considered to be common in Tunisia, where we found it to coexist with Cx. pipiens during the summer and autumn months. Culex perexiguus is an important vector of arboviruses, including West Nile virus and Sindbis virus, both of which have been detected in this species in Tunisia [83]. Its role in the transmission of these viruses has been established in other North African countries, including Egypt [46]. First described in 1903, Cx. perexiguus has been confused with Cx. decens, Cx. pallidocephalus and especially with Cx. univittatus [55]. Harbach (1988) proposed criteria to clearly distinguish these different taxa. The distribution of Cx. perexiguus extends from Morocco to India [3] and it has been recorded in all countries of North Africa [17].
Culex (Culex) pipiens Linnaeus, 1758
This species is widespread and abundant in Tunisia. It was collected in all bioclimatic zones from various water bodies, with populations observed in both urban and rural areas. Its ecological plasticity explains its adaptability to breeding in various environments under different environmental conditions, making it one of the most widespread mosquito species in the country (Fig. S11). Importantly, Cx. pipiens larvae have been found in a variety of natural habitats, including wadi banks, swamps, shallow temporary pools, hill lakes, ponds, irrigation channels and streams. It also breeds readily in man-made water areas such as canals, pools, unused wells, ditches, catch basins, unused tyres, open-air wastewater and rainwater canals, ditches, water troughs, crawlspaces and drainage canals. In the subset of characterised larval habitats, Cx. pipiens was recorded mainly in low-salinity inland wadis (0.5–2.6 g/L NaCl; pH 7.08–7.7) but also occurred in brackish coastal sebkha/lagoon environments (up to 28–32 g/L NaCl; pH ~7.47–8.5), supporting its broad ecological plasticity. Its distribution Maghreb-wide is well documented, with records in all countries [17, 18, 108, 151]. Globally, Cx. pipiens is widely distributed across temperate and tropical regions, including Europe, Africa, Asia and North America. Culex pipiens is a well-known vector of West Nile virus and thus particularly important from a public health perspective. This species was implicated in the 1997 and 2003 outbreaks of West Nile fever in Tunisia, during which 20 deaths were reported [47, 153]. Additionally, Cx. pipiens is a potential vector of other arboviruses, including Rift Valley fever virus, especially during periods of increased rainfall and standing water [40, 127].
Culex (Barraudius) pusillus Macquart, 1850
Our findings indicate that Cx. pusillus is a halophilic species, found only in saline, vegetation-rich environments, where its larvae develop. This species was previously considered to be rare in Tunisia and recorded only in the south, but it is now also present in the centre of the country, suggesting a shift in its distribution (Fig. S9). This change is likely driven by habitat changes resulting from increased drought, which may be creating new ecological niches northward.
In North Africa, it has also been documented in Algeria [118] and Egypt [1], and extends to the southern-most Palearctic region [108].
During the course of our research, we noted that females of this species were not aggressive towards humans. Of note, Cx. pusillus is not known to bite humans and is thus not considered a significant public health threat.
Culex (Culex) theileri Theobald, 1903
Culex theileri is another mosquito species that we identified in all bioclimatic zones during this investigation, particularly in a wide range of breeding habitats (Fig. S12). Like Cx. pipiens, Cx. theileri displays great ecological flexibility, inhabiting both natural and artificial bodies of water (wadi banks, swamps, shallow pools, ponds, streams and irrigation channels). It can tolerate varying levels of salinity, even breeding in brackish water, which expands its distribution across arid and semi-arid regions of the country. Culex theileri is considered to be common in Tunisia and has a broad geographic distribution range; we observed significant populations across rural and agricultural areas. In the characterised sites, Cx. theileri occurred from low-salinity inland habitats (0.5–2.6 g/L NaCl; pH 7.08–7.7) to more mineralised/brackish waters (e.g., 4.3 g/L NaCl, pH 8.8; and up to 25 g/L NaCl, pH 8.5), consistent with its tolerance to salinity gradients. Historically, Cx. theileri was noted for its wide distribution in North Africa, with reports in Algeria [136], Morocco [151] and Libya [66]. Globally, it is found from southern Europe to the Middle East, and as far east as Afghanistan. The distribution of Cx. theileri has remained relatively stable, although it has been found more frequently in semi-arid regions where brackish water habitats have been expanded by agricultural practices. Although this species has been recorded as a potential vector of West Nile virus, its public health importance in Tunisia relative to other vector species like Cx. pipiens remains unclear [162].
Genus Culiseta Felt, 1904 [Tribe Culisetini Belkin, 1962]
Among the five species of the genus Culiseta (Cs.) mentioned in Tunisia, we collected only two during our survey, namely Cs. longiareolata and Cs. subochrea. Brunhes et al. (2000) [17] mentioned the presence of Cs. annulata and Cs. fumipennis in Tunisia, and Robert et al. (2019) [108] listed Cs. morsitans in the country. However, we found none of these species during our investigations.
Culiseta (Allotheobaldia) longiareolata (Macquart, 1838)
During our surveys, the larvae of Cs. longiareolata were collected in a variety of habitats spanning both natural breeding sites like wadi banks and swamps and artificial sites such as canals, unused wells, catch basins, discarded tyres and irrigation ditches. In the subset of characterised habitats, Cs. longiareolata was also detected in brackish sites (e.g., 32 g/L NaCl; pH 8.5), co-occurring with Cx. pipiens and Ae. detritus. This adaptability to diverse breeding environments contributes to the widespread distribution and notable abundance of Cs. longiareolata across all bioclimatic zones in Tunisia (Fig. S13). Widely distributed throughout the southern Palearctic, as well as the Eastern and Afro-tropical regions, Cs. longiareolata has a particularly high prevalence in North Africa [18]. In Tunisia, it often shares habitats with Cx. pipiens [20, 105, 119, 137, 158, 159]. Compared with previous surveys, our study suggests an expanded range and confirms the species’ abundance across various Tunisian habitats where it feeds primarily on birds, serving as a vector for avian blood parasites. It rarely targets humans [6].
Culiseta (Culiseta) subochrea (Edwards, 1921)
The larvae of Cs. subochrea develop in fresh water pools with low organic matter; however, they can also tolerate brackish water, as they are found in irrigation ditches lined with Salicornia, in association with Ae. detritus and Ae. caspius. During our investigations, we identified Culiseta subochrea exclusively in the oases of Tozeur [33°54′54.71″ N; 8°7′49.38″ E]. Although currently rare, this species was once more broadly distributed according to historical records [18]. Culiseta subochrea has been reported in North Africa, except in Libya, and penetrates deep into desert regions [88]. It is a widely distributed species found throughout Europe, but appears to be more abundant in the southern Palearctic region [93].
Genus Orthopodomyia Theobald, 1904 [Tribe Orthopodomyiini Belkin, Heinemann & Page, 1970]
Orthopodomyia pulcripalpis (Rondani, 1872)
Orthopodomyia pulcripalpis is a rare arboreal species found in northern forests in humid and sub-humid zones (Fig. S14). We found larvae in cork oak tree holes. This species is present throughout Western Europe and the Mediterranean Palearctic sub-region [6].
Genus Uranotaenia Lynch Arribálzaga, 1891 [Tribe Uranotaeniini Lahille, 1904]
Uranotaenia (Pseudoficalbia) unguiculata Edwards, 1913
Uranotaenia unguiculata larvae were observed from sub-humid to sub-Saharan zones (Fig. S14). The larvae breed primarily in shallow temporary pools with abundant upright vegetation such as poorly maintained canals, ponds and irrigation ditches. The water in these pools is fresh to slightly salty. Adults are frequently collected in early autumn. Females of Ur. unguiculata transmit parasites to amphibians and reptiles. In Tunisia, several authors have reported this common species [124, 156]. Uranotaenia unguiculata has been reported in North Africa from Morocco to Egypt [58]. In addition to the Mediterranean region, it is also found in Central Europe and the Middle East [111].
Species documented in the literature but not collected in our investigations (see Supplementary Tables 1–3 for details)
Anopheles (Anopheles) coustani Laveran, 1900
Despite historical citations of its existence in Tunisia [20, 132], An. coustani was not recorded in our investigations. Anopheles coustani belongs to the Coustani group, which includes morphologically similar species such as An. (Ano.) ziemanni Grünberg, 1902. Robert et al. (2019) [108] included An. coustani in their regional checklist based on outdated data, without recent confirmation. These records are now considered to be questionable. Several studies suggest that earlier identifications of An. coustani in North Africa, including Tunisia, may have resulted from misidentifications with An. ziemanni, a species with overlapping morphological characteristics [151].
Anopheles (Cellia) dthali Patton, 1905
Anopheles dthali has been historically reported in the Maghreb, particularly in the southern regions of Tunisia and Morocco [124]. This species prefers desert or semi-desert environments, and typically breeds in temporary water bodies [54]. In Tunisia, An. dthali has been recorded mainly in the southern part of the country, and is considered rare, with previous records from arid zones [14, 36]. Anopheles dthali was not detected during our surveys. This may reflect a decline in local populations linked to prolonged drought and habitat loss, but its persistence at low densities or in unsampled microhabitats cannot be ruled out. Although not considered a primary vector, An. dthali has been implicated in the transmission of Plasmodium in limited reports from North Africa, and its vectorial capacity remains poorly documented [23].
Anopheles (Cellia) superpictus Grassi, 1899
Anopheles superpictus was reported in eastern Tunisia, particularly on the banks of wadis [63, 161], but we did not detect it during our investigations. This apparent absence may reflect a decline in local populations, due probably to environmental changes that may have severely limited species-suitable breeding sites. Larvae usually develop in river bed pools [112]. Anopheles superpictus is widely distributed across the northern Mediterranean region, from North Africa to the Middle East [108], where it is well known as a malaria vector [138].
Aedes (Stegomyia) aegypti (Linnaeus, 1762)
Ae. aegypti was reported in Tunisia in several cities [20, 114, 166], but it disappeared in the early 1960s. Despite its absence in our surveys, this species must remain under surveillance because it can thrive in urban environments and plays a role as a vector of several arboviruses such as chikungunya, dengue, yellow fever and Zika viruses [44, 95, 147].
Aedes (Ochlerotatus) albineus Séguy, 1923
Aedes albineus was first described by Séguy (1923) [115] from adult specimens collected in southern Algeria. However, its taxonomic status remains uncertain due to the loss of type material and limited original descriptions. While it resembles Ae. caspius, this species is paler and lacks the hook-shaped setae on the male coxite. Some authors (e.g., Knight and Stone, 1977) [69] considered it a synonym of Ae. caspius, whereas others [17, 74] suggested it may correspond to pale forms of Ae. caspius observed in desert regions. Its absence in our current surveys could be due to habitat changes or past misidentifications. Further molecular studies are needed to clarify its status and potential occurrence in Tunisia.
Aedes (Aedes) cinereus Meigen, 1818
Robert et al. reported (2019) [108] Aedes cinereus from Tunisia but several authoritative reviews, such as Moussiegt, 1983 [84] and Brunhes et al., 2000 [18], did not include this species in the Tunisian mosquito fauna. Callot (1938) [20] noted that Ae. cinereus larvae are very similar morphologically to those of Ae. vexans, which may lead to misidentification. The presence of Ae. cinereus in Tunisia therefore remains doubtful, as no detailed descriptions of its morphology or breeding habitat have been documented to date. Under favorable climatic conditions, we cannot exclude its future occurrence in the country.
Aedes (Acartomyia) zammitii (Theobald, 1903)
Among the three species composing the Mariae Complex, two are found in North Africa (Ae. mariae and Ae. zammitii) [29]. Although Ae. zammitii was previously reported in Tunisia [17], we did not find this species during our surveys. Morphologically, Ae. zammitii is very similar to Ae. mariae, and both species share the same habitat niches of rock pools along the seashore. Ae. zammitii is found primarily on the eastern coasts of Italy, Sicily and Malta [28], and it is possible that previous records of Ae. zammitii in Tunisia were misidentifications of Ae. mariae. Alternatively, Ae. zammitii may have lost its range due to climatic changes, urbanisation, or other environmental factors. The presence of this species and of Ae. cinereus has not been confirmed in Morocco or Algeria.
Culex (Culex) antennatus (Becker, 1903)
Culex antennatus is a species widely distributed throughout the Afrotropical region and as far south as Madagascar. It has been recorded in North Africa, including Tunisia, under its synonym Culex laurenti Newstead, 1907 [55]. Culex antennatus has been documented in neighbouring countries such as Algeria and Morocco [17], but was not found during our surveys. This could reflect habitat degradation, particularly in freshwater sources impacted by drying and increased salinity, a consequence of climate change. This species may also have become rarer in northern Africa due to competition from other mosquito species better adapted to altered environments.
Culex (Culex) territans Walker, 1856
Culex territans has been documented in North Africa, especially in Tunisia. It usually breeds in small, clear ponds and has a marked preference for cooler, shaded habitats [18]. It often shares the same habitat as Culex impudicus. The absence of this species in our study could be indicative of the evolution of these specific microhabitats, which have been affected by habitat loss and urban development. One hypothesis is that environmental changes, such as the drying up of small ponds or the reduction in the availability of suitable shaded breeding sites have contributed to the apparent absence of the species [41].
Culex (Culex) univittatus Theobald, 1901
Culex univittatus was first reported and described in Tunisia by Callot (1938) [20], but its current presence remains uncertain. Robert et al. (2019) [108] suggested that earlier reports of Cx. univittatus in Tunisia may have resulted from misidentifications of Cx. perexiguus, due to their close morphological similarities. We found no Cx. univittatus in our current surveys, which could support the hypothesis of past misidentifications.
Culiseta (Culiseta) annulata (Schrank, 1776)
Culiseta annulata is a cold-adapted species with a broad distribution across Europe, parts of Asia, and North Africa [108]. In Tunisia, it was reported by Husson (1907) [60] and later by Colas-Belcour (1931) [27] in the oasis of Tozeur, and also noted in nearby regions such as Biskra in Algeria [20]. This species is easily recognisable by its wing spots and white leg bands. We found no Culiseta annulata in our investigations, possibly due to ecological changes such as desertification and salinisation affecting freshwater habitats, which may contribute to a shift in the composition of the mosquito community in Tunisia’s semi-arid zones.
Culiseta (Culicella) fumipennis (Stephens, 1825)
Culiseta fumipennis is a Euro-Mediterranean mosquito historically recorded in the humid and forested areas of northwestern Tunisia, notably in Ghardimaou and Aïn Draham, based on larval identifications and determinations by Senevet and Prunnelle (1928) [125]. It has also been reported from Algeria [52] and Morocco [151], confirming its presence in the humid bioclimatic zones of North Africa. Despite these earlier records, Cs. fumipennis was not detected during our investigations. This may reflect the degradation of its preferred habitats.
Culiseta (Culicella) morsitans (Theobald, 1901)
Culiseta morsitans has also been reported to be present in Tunisian mosquito fauna [17, 108]. In reality, Brunhes et al. (2000b) [17] had already excluded this species from the list of mosquitoes of the Maghreb. Similarly, Moussiegt (1983) [84] made no mention of Cs. morsitans in Tunisia. Historically, Senevet and Prunnelle (1928) [125] reported having examined 18 larvae of Cs. morsitans collected by Gauthier in a marsh located in the former Lake Halloula (Algeria). In Libya, Ghidini (1934) [49] found larvae of this species in Oued Tmini. Later, Gaud (1953) [48] and Trari (1991) [151] noted that they had never encountered this species in Morocco, despite apparently favourable climatic conditions in the country. Taken together, these findings suggest that the presence of Culiseta morsitans in Tunisia remains highly doubtful.
Conclusion
During our investigations, we observed 35 mosquito species belonging to 6 genera, of the 49 reported in the literature. It is certain that some species, such as Ae. aegypti have disappeared completely, while others such as An. dthali and Ae. albineus have become rare, especially since Tunisia has experienced record water shortages and extremely hot summers over the past decade as a result of climate change. The expanding human population has resulted in the construction of buildings and infrastructure and the introduction of roads, which have had a detrimental effect on the natural environment. The improper disposal of wastewater, often discharged into waterways without any form of treatment, exacerbates the situation. These environmental changes have a significant impact on the mosquito habitats whose disruption is a key factor in the observed changes in population diversity; the disappearance of many ponds, marshes and lakes, and the drying up of wadis and certain water sources. Consequently, several larval breeding sites have disappeared. We found that some stagnant water points have become very polluted and are suitable only for Cx. pipiens.
Our work provides an overview of the mosquito species present in Tunisia, their current distribution and their favourable habitats, thus facilitating investigations should arboviroses circulate. From a taxonomic and nomenclatural point of view, it remains important to update classifications and identifications. The implementation of new identification techniques using genetic/molecular approaches and the use of new techniques such as matrix-assisted laser desorption/ionisation time-of-flight MALDI-ToF mass spectrometry will allow for rapid and accurate identification of specimens and potentially highlight cryptic species, such as those of the Detritus or Mariae complexes or others with a similar morphology that create identification problems. It is also notable that compared to previous records, the number of species has decreased, potentially due to misidentifications such as An. coustani, An. ziemanni and Cx. univittatus. Additionally, some species have changed their distribution areas, such as An. petragnani, which has recently been found in central Tunisia, and Ae. mariae, which now occupies more habitats along the northern coasts. Finally, monitoring the spread of the recently introduced and invasive Ae. albopictus is necessary given its role as a vector for several arboviruses. The introduction of other species such as An. stephensi should also be surveyed.
Acknowledgments
We thank all individuals and institutions whose previous entomological fieldwork and published data contributed to the updated mosquito checklist presented in this manuscript. We also acknowledge the support of the VectorNet project, funded by the European Centre for Disease Prevention and Control (ECDC) and the European Food Safety Authority (EFSA), which contributed to vector distribution data collection during the 2017 cross-sectional survey in Tunisia. The authors would also like to thank Deborah Glassman for English editing and for her constructive comments on the manuscript.
Funding
This work was supported by the research laboratory Viruses, Vectors and Hosts (LR20IPT02), funded by the Ministry of Higher Education and Scientific Research of Tunisia.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
Author contribution statement
Ahmed Ouni: Writing – original draft, Conceptualisation, Methodology, Formal analysis, Data curation, Investigation, Visualisation, Validation. Adel Rhim: Investigation, Methodology, Software, Validation, Visualisation. Youmna M’Ghirbi: Conceptualisation, Validation, Writing – review & editing, Project administration. Francis Schaffner: Visualisation, Validation, Writing – review & editing. Ali Bouattour: Supervision, Validation, Writing – original draft, Conceptualisation, Visualisation, Writing – review & editing, Funding acquisition, Resources. All authors have read and approved the final version of the manuscript.
Ethics approval
No formal ethics approval was required.
Supplementary material
Table S1. Natural and artificial breeding sites of mosquito species in Tunisia across bioclimatic zones. Access Supplementary Material
Table S2. Checklist of mosquito species (Diptera: Culicidae) in Tunisia based on field collections (2013–2023) and literature records. Access Supplementary Material
Table S3. Historical and bibliographic records of mosquito species in Tunisia. Table S3 includes many references, including [15, 21, 22, 30, 31, 39, 51, 71, 75, 76, 94, 102, 103, 107, 117, 121, 126, 128, 129, 130, 131, 133, 134, 135, 148, 157, 160]. Access Supplementary Material
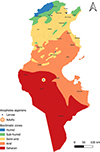 |
Figure S1. Geographical distribution of Anopheles algeriensis in Tunisia (2013–2023) across the different bioclimatic zones. |
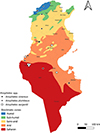 |
Figure S2. Geographical distribution of Anopheles spp. [Anopheles algeriensis, Anopheles cinereus, Anopheles multicolor, Anopheles plumbeus and Anopheles sergentii] in Tunisia (2013–2023) across the different bioclimatic zones. |
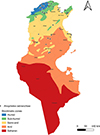 |
Figure S3. Geographical distribution of Anopheles labranchiae in Tunisia (2013–2023) across the different bioclimatic zones. |
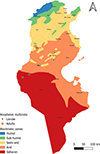 |
Figure S4. Geographical distribution of Anopheles multicolor in Tunisia (2013–2023) across the different bioclimatic zones. |
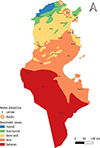 |
Figure S5. Geographical distribution of Aedes albopictus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
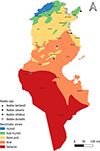 |
Figure S6. Geographical distribution of selected Aedes spp. [Aedes berlandi, Aedes dorsalis, Aedes vexans and Aedes vitatus] recorded in Tunisia (2013–2023) across the different bioclimatic zones. |
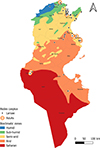 |
Figure S7. Geographical distribution of Aedes caspius in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
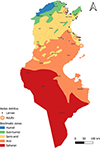 |
Figure S8. Geographical distribution of Aedes detritus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
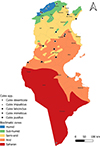 |
Figure S9. Geographical distribution of different Culex species [Culex deserticola, Culex impudicus, Culex laticinctus, Culex mimeticus and Culex pusillus] in Tunisia (2013–2023) across the different bioclimatic zones. |
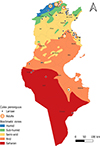 |
Figure S10. Geographical distribution of Culex perexiguus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
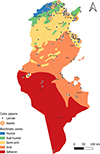 |
Figure S11. Geographical distribution of Culex pipiens in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
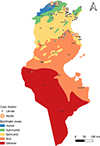 |
Figure S12. Geographical distribution of Culex theileri in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
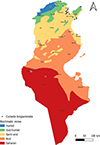 |
Figure S13. Geographical distribution of Culiseta longiareolata in Tunisia (2013–2023) across the different bioclimatic zones. |
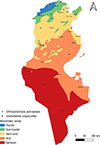 |
Figure S14. Geographical distribution of Orthopodomyia pulcripalpis and Uranotaenia unguiculata in Tunisia (2013–2023) across the different bioclimatic zones. |
References
- Abdel-Malek A. 1960. The culicine mosquitoes of the northern region of the United Arab Republic. Bulletin de la Société Entomologique d’Égypte, 44, 111–128. [Google Scholar]
- Adlaoui E, Faraj C, El Bouhmi M, El Aboudi A, Ouahabi S, Tran A, Fontenille D, El Aouad R. 2011. Mapping malaria transmission risk in Northern Morocco using entomological and environmental data. Malaria Research and Treatment, 2011, 1–9. [Google Scholar]
- Alves J, Gomes B, Rodrigues R, Silva J, Arez AP, Pinto J, Sousa CA. 2010. Mosquito fauna on the Cape Verde Islands (West Africa): an update on species distribution and a new finding. Journal of Vector Ecology, 35, 307–312. [Google Scholar]
- Ayadi A, Makni F, Sellami H, Cheikh-Rouhou F, Ben Hamed S. 2000. Imported malaria in Sfax (Tunisia). Médecine Tropicale, 60, 249–252. [Google Scholar]
- Bailly-Choumara H. 1968. Contribution à l’étude des moustiques du Maroc (Diptera, Culicidae). Six espèces nouvelles pour le pays. Cahiers de l’ORSTOM, Série Entomologie Médicale et Parasitologie, 6, 139–144. [Google Scholar]
- Becker N, Petric D, Zgomba M, Boase C, Madon M, Dahl C, Kaiser A. 2010. Mosquitoes and their control. 2nd edn. Heidelberg: Springer. [Google Scholar]
- Beji M, Rhim A, Roiz D, Bouattour A. 2017. Ecophysiological characterization and molecular differentiation of Culex pipiens forms (Diptera: Culicidae) in Tunisia. Parasites & Vectors, 10, 327. [Google Scholar]
- Ben Ayed W, Amraoui F, M’ghirbi Y, Schaffner F, Rhaim A, Failloux A-B, Bouattour A. 2019. A survey of Aedes (Diptera: Culicidae) mosquitoes in Tunisia and the potential role of Aedes detritus and Aedes caspius in the transmission of Zika Virus. Journal of Medical Entomology, 56, 1377–1383. [Google Scholar]
- Benhissen S, Habbachi W, Ouakid ML. 2017. Biodiversité et répartition des moustiques (Diptera: Culicidae) dans les oasis de la region de Biskra (sud-est algérien). Algerian Journal of Arid Environment, 7, 96–101. [Google Scholar]
- Blagrove MSC, Sherlock K, Chapman GE, Impoinvil DE, McCall PJ, Medlock JM, Lycett G, Solomon T, Baylis M. 2016. Evaluation of the vector competence of a native UK mosquito Ochlerotatus detritus (Aedes detritus) for dengue, chikungunya and West Nile viruses. Parasites & Vectors, 9, 452. [Google Scholar]
- Bohers C, Mousson L, Madec Y, Vazeille M, Rhim A, M’ghirbi Y, Bouattour A, Failloux A-B. 2020. The recently introduced Aedes albopictus in Tunisia has the potential to transmit chikungunya, dengue and Zika viruses. PLoS Neglected Tropical Diseases, 14, e0008475. [Google Scholar]
- Bouattour A, Khrouf F, Rhim A, M’ghirbi Y. 2019. First detection of the Asian Tiger Mosquito, Aedes (Stegomyia) albopictus (Diptera: Culicidae), in Tunisia. Journal of Medical Entomology, 56, 1112–1115. [Google Scholar]
- Bouattour A, Rhaiem A, BachHamba D. 1990. Study of the vectorial capacity of Anopheles labranchiae in the Nefza region. Report of the Research Program of the Pasteur Institute of Tunis, French Cooperation and the National Agency of Environmental Protection. pp. 54–60. [Google Scholar]
- Bouattour A, Rhaim A, BachHamba D. 1993. Étude de la capacité vectorielle d’Anopheles labranchiae dans la région de Nefza. Rapport technique multigrade disponible à l’Institut Pasteur de Tunis et l’Agence Nationale de Protection de l’Environnement. [Google Scholar]
- Boucheté B, Kennou M, Chauvet G. 1991. Ethology and vectorial capacity of Anopheles from Tunisia in the region of Sidi Bouzid. Rapport technique. Geneva: World Health Organization. [Google Scholar]
- Brunhes J. 1999. Culicidae du Maghreb. Description d’Aedes (Ochlerotatus) biskraensis n. sp. d’Algérie (Diptera, Nematocera). Bulletin de la Société Entomologique de France, 104, 45–52. [Google Scholar]
- Brunhes J, Hassaïne K, Rhaiem A, Hervy J-P. 2000. Les Culicides de l’Afrique méditerranéenne: espèces présentes et répartition (Diptera, Nematocera). Bulletin de la Société Entomologique de France, 105, 195–204. [Google Scholar]
- Brunhes J, Rhaim A, Geoffroy B, Angel G, Hervy J-P. 2000. Les moustiques de l’Afrique méditerranéenne : logiciel d’identification et d’enseignement- fdi:010021400- Horizon. [Google Scholar]
- Bueno-Marí R, Jiménez-Peydró R. 2011. Anopheles plumbeus Stephens, 1828: a neglected malaria vector in Europe. Malaria Reports, 1, e2-e2. [Google Scholar]
- Callot J. 1938. Contribution à l’étude des moustiques de Tunisie et en particulier du sud de la régence. Archives de l’Institut Pasteur de Tunis, 27, 133–183. [Google Scholar]
- Callot J. 1947. Sur les larves de Culex hortensis Fic. et de C. deserticola K. Annales de Parasitologie. 22, 480–481. [Google Scholar]
- Callot J, Ristorcelli A. 1938. Localité nouvelle pour Anopheles (Myzomyia) sergenti Theobald 1907. Archives de l’Institut Pasteur deTunis, 15, 105–107. [Google Scholar]
- Carnevale P, Robert V, Molez J-F, Baudon D. 1984. Epidémiologie générale : faciès épidémiologiques des paludismes en Afrique subsaharienne- fdi:15177-Horizon. [Google Scholar]
- Chadli A, Kennou MF, Kooli J. 1985. Malaria in Tunisia: history and present status. Bulletin de la Societé de Pathologie Exotique et de ses Filiales, 78, 844–851. [Google Scholar]
- Chadli A, Kennou MF, Kooli J. 1986. Campaigns for the eradication of malaria in Tunisia: history and current situation. Archives de l’Institut Pasteur de Tunis, 63, 35–50. [Google Scholar]
- Chanda E, Ameneshewa B, Bagayoko M, Govere JM, Macdonald MB. 2017. Harnessing integrated vector management for enhanced disease prevention. Trends in Parasitology, 33, 30–41. [Google Scholar]
- Colas-Belcour J. 1931. Notes sur la faune parasitologique des oasis de Tozeur et Kébili. Archives de l’Institut Pasteur de Tunis 4, 1–25. [Google Scholar]
- Coluzzi M, Bianchi-Bullini, AP, Bullini L. 1976. Speciation in the mariae complex of the genus Aedes (Diptera: Culicidae). Bollettino della Società Italiana di Biologia Sperimentale, 51, 222–223. [Google Scholar]
- Coluzzi M, Sabatini A. 1968. Morphological divergences and sterility barriers in the Aedes mariae complex (Diptera, Culicidae). Bollettino della Società Italiana di Biologia Sperimentale, 44, 49–70. [Google Scholar]
- Coluzzi M, Sabatini A, Bullini L, Ramsdale C. 1974. Nuovi dati sulla distribuzione delle specie del complesso mariae del genere Aedes. Bollettino della Società Italiana di Biologia Sperimentale, 35, 321–339. [Google Scholar]
- Coluzzi M, Sacca G, Feliciangefi D. 1965. Il complesso A. claviger nella sotto-regione mediteranea. Bollettino della Società Italiana di Biologia Sperimentale, 41, 97–102. [Google Scholar]
- Ribeiro H, da Cunha Ramos H, Pires CA, Capela RA. 1988. An annotated checklist of the mosquitoes of continental Portugal (Diptera: Culicidae). Actas III Congresso Ibérico de Entomologia, Granada. pp. 233–254. [Google Scholar]
- Dahl C, Blackmore MS. 2001. The distribution and status of Ochlerotatus geniculatus (Olivier) in Fennoscandia. European Mosquito Bulletin, 9, 12–16. [Google Scholar]
- Danielová V. 1966. Quantitative relationships of Tahyna virus and the mosquito Aëdes vexans. Acta Virologica, 10, 62–65. [Google Scholar]
- Diallo D, Sall AA, Diagne CT, Faye O, Faye O, Ba Y, Hanley KA, Buenemann M, Weaver SC, Diallo M. 2014. Zika virus emergence in mosquitoes in southeastern Senegal, 2011. Plos One, 9, e109442. [Google Scholar]
- Doby JM, Vermeil C. 1952. Gîtes tunisiens à Anopheles multicolor et prophylaxie antipaludique. Bulletin de la Société de Pathologie Exotique, 27, 481–482. [Google Scholar]
- Doosti S, Yaghoobi-Ershadi MR, Sedaghat MM, Moosa-Kazemi SH, Akbarzadeh K, Hashemi-Aghdam SS. 2018. Genetic population diversity of Aedes caspius in southern provinces of Iran. Bulletin de la Societé de Pathologie Exotique, 111, 1–7. [Google Scholar]
- Drouin A, Chevalier V, Durand B, Balenghien T. 2022. Vector competence of Mediterranean mosquitoes for Rift Valley fever virus: A meta-analysis. Pathogens, 11, 503. [Google Scholar]
- Edwards FW. 1921. A revision of the mosquitos of the Palaearctic region. Bulletin of Entomological Research, 12, 263–351. [Google Scholar]
- Eifan S, Hanif A, Nour I, Alqahtani S, Eisa ZM, Dafalla O, Kohl A. 2021. Distribution and molecular iIdentification of Culex pipiens and Culex tritaeniorhynchus as potential vectors of Rift Valley fever virus in Jazan, Saudi Arabia. Pathogens, 10, 1334. [Google Scholar]
- Eisen L, Bolling BG, Blair CD, Beaty BJ, Moore CG. 2008. Mosquito species richness, composition, and abundance along habitat-climate-elevation gradients in the northern Colorado front range. Journal of Medical Entomology, 45, 800–811. [Google Scholar]
- El-Bahnasawy MM, Saleh NMK, Khalil MF, Morsy TA. 2011. The impact of three anopheline mosquito species in Toshka, on the introduction of chloroquine resistant P. falciparum to Egypt. Journal of the Egyptian Society of Parasitology, 41, 573–592. [Google Scholar]
- Emberger L. 1960. L’exploitation traditionnelle du maquis du nord de la Tunisie: Possibilité d’une meilleure utilisation. Tunis: Institut Agronomique et de Recherches Forestières. [Google Scholar]
- Facchinelli L, Badolo A, McCall PJ. 2023. Biology and behaviour of Aedes aegypti in the human environment: Opportunities for vector control of arbovirus transmission. Viruses, 15, 636. [Google Scholar]
- Faraj C, Adlaoui E, Ouahabi S, Rhajaoui M, Fontenille D, Lyagoubi M. 2009. Entomological investigations in the region of the last malaria focus in Morocco. Acta Tropica, 109, 70–73. [Google Scholar]
- Ferraguti M, Heesterbeek H, Martínez-de la Puente J, Jiménez-Clavero MÁ, Vázquez A, Ruiz S, Llorente F, Roiz D, Vernooij H, Soriguer R, Figuerola J. 2021. The role of different Culex mosquito species in the transmission of West Nile virus and avian malaria parasites in Mediterranean areas. Transboundary and Emerging Diseases, 68, 920–930. [Google Scholar]
- Garbouj M, Bejaoui M, Aloui H, Ben Ghorbal M. 2003. La maladie du Nile occidental. Bulletin Épidémiologique, 3, 4–6. [Google Scholar]
- Gaud J. 1953. Notes biogéographiques sur les Culicides du Maroc. Archives de l’Institut Pasteur du Maroc, 21, 443–490. [Google Scholar]
- Ghidini GM. 1934. Contributo alla conescenza dei Culicidi della Libia. Bollettino della Società Entomologica Italiana, 66, 31–34. [Google Scholar]
- Gounot M. 1995. Bioclimatologie de la Tunisie. Essai de synthèse sur la végétation et la phyto-écologie Tunisiennes. Tunis: Institut National de la Recherche Agronomique de Tunisie (Ed. M.A. NABLI). [Google Scholar]
- Guy Y, Salieres A, Boesiger E. 1976. Contribution à l’étude du complexe maculipennis (Diptera-Culicidae-Anophelinae). Revue d’Épidémiologie et de Santé Publique, 15, 227–282. [Google Scholar]
- Hamaidia H, Berchi S. 2018. Étude systématique et écologique des Moustiques (Diptera: Culicidae) dans la région de Souk-Ahras (Algérie). Entomologie faunistique - Faunistic Entomology, 21, 45–56. [Google Scholar]
- Hammadi D, Boubidi S, Chaib SE, Saber A, Khechache Y, Gasmi M, Harrat Z. 2009. Le paludisme au Sahara algérien. Bulletin de la Societé de Pathologie Exotique, 102, 185–192. [Google Scholar]
- Hamon J, Dyemkouma A, Ouedraogo A. 1963. Présence en Afrique occidentale d’Anopheles argenteolobatus et d’A. salbaii et nouvelles localités pour A. dthali et A. rufipes var. Brucechwatti. Bulletin de la Societé de Pathologie Exotique, 56, 407–415. [Google Scholar]
- Harbach R. 1988. The mosquitoes of the subgenus Culex in southwestern Asia and Egypt (Diptera: Culicidae). Contributions of the American Entomological Institute, 24, 1–247. [Google Scholar]
- Harbach R, Harrison B, Gad A-A, Kenawy M, El-Said S. 1988. Records and notes on mosquitoes (Diptera: Culicidae) collected in Egypt. Mosquito Systematics, 20, 317–342. [Google Scholar]
- Harbach RE. 2013. The phylogeny and classification of Anopheles. Chapter: 1, in Anopheles mosquitoes - New insights into malaria vectors. Manguin S, Editor. InTech: Rijeka, Croatia. [Google Scholar]
- Himmi O. 1991. Culicid (Diptera) from Morocco. Updated determination key and study of the dynamics and biological cycles of some populations in the Rabat Kenitra Region. Scientific Research Publishing. [Google Scholar]
- Horsfall WR. 1955. Mosquitoes: Their bionomics and relation to disease. New York: Ronald Press Company. [Google Scholar]
- Husson AD. 1907. Enquête sur le paludisme dans trois centres de colonisation : Mateur, Béja, Le Goubellat (août- novembre 1906). Archives de l’Institut Pasteur de Tunis, 5, 1–35. [Google Scholar]
- Jansen S, Heitmann A, Lühken R, Leggewie M, Helms M, Badusche M, Rossini G, Schmidt-Chanasit J, Tannich E. 2019. Culex torrentium: A potent vector for the transmission of West Nile virus in Central Europe. Viruses, 11, 492. [Google Scholar]
- Joubert L, Oudar J, Mouchet J, Hannoun C. 1967. Transmission de la myxomatose par les moustiques de Camargue. Rôle prééminent de Aedes caspius et des Anopheles du groupe maculipennis. Bulletin de la Société de Pathologie Exotique, 60, 27–33. [Google Scholar]
- Juminer B. 1959. Notes sur l’anophélisme en Tunisie. Archives de l’Institut Pasteur de Tunis, 36, 37–42. [Google Scholar]
- Juminer B, Kchouk M, Rioux JA, Ben Osman F. 1964. À propos des culicidés vulnérants de la littorale de Tunis. Archives de l’Institut Pasteur de Tunis, 41, 23–32. [Google Scholar]
- Kenawy MA, Sowilem MM, Hamed MS, Merdan AI. 1996. The ecological characteristics of Anopheles mosquitoes associated with low malaria transmission in Siwa oasis, Egypt. Journal of the Egyptian Public Health Association, 71, 185–200. [Google Scholar]
- Khalefa AS, Ghana S, Rashid NM, Shaibi T. 2022. Mosquito Fauna (Diptera, Culicidae) in Tarhuna Region, Libya. Zoodiversity, 56, 1–10. [Google Scholar]
- Khalil GM. 1980. A preliminary survey of mosquitoes in Upper Egypt. Journal of the Egyptian Public Health Association, 55, 355–362. [Google Scholar]
- Khongwichit S, Chansaenroj J, Chirathaworn C, Poovorawan Y. 2021. Chikungunya virus infection: molecular biology, clinical characteristics, and epidemiology in Asian countries. Journal of Biomedical Science, 28, 84. [Google Scholar]
- Knight KL, Stone A. 1977. A catalog of the mosquitoes of the world (Diptera: Culicidae). 2nd ed. Berkeley, CA: University of California Press. p. 1–611. [Google Scholar]
- Krida G, Daoud-Bouattour A, Mahmoudi E, Rhim A, Ghrabi-Gammar Z, Chermiti B, Failloux A-B, Bouattour A. 2012. Relation entre facteurs environnementaux et densités larvaires d’Ochlerotatus caspius Pallas 1771 et Ochlerotatus detritus Haliday 1833 (Diptera: Culicidae) en Tunisie. Annales de la Société Entomologique de France, 48, 18–28. [Google Scholar]
- Krida G, Rhaiem A, Jarraya A, Bouattour A. 1998. Morphologie comparée des quatre stades larvaires de Culex (Culex) pipiens Linné récolté en Tunisie (Diptera, Culicidae). Bulletin de la Société Entomologique de France. 103 (1), 5–10. [Google Scholar]
- Laboudi M, Faraj C, Sadak A, Harrat Z, Boubidi SC, Harbach RE, El Aouad R, Linton Y-M. 2011. DNA barcodes confirm the presence of a single member of the Anopheles maculipennis group in Morocco and Algeria: An. sicaulti is conspecific with An. labranchiae. Acta Tropica, 118, 6–13. [Google Scholar]
- Lafri I, Bitam I, Beneldjouzi A, Ben Mahdi MH. 2014. An inventory of mosquitoes (diptera: Culicidae) in Algeria. Asian Pacific Journal of Tropical Biomedicine, 4, 430–434. [Google Scholar]
- Lambert MN, Pasteur J, Rioux A, Delalbre-Belmonte A, Balard Y. 1990. Aedes caspius (Pallas, 1771) and Ae. dorsalis (Meigen, 1830) (Diptera: Culicidae). Morphological and genetic analysis of two sympatric populations and proof of reproductive isolation, Journal of Medical Entomology, 26, 381–398. [Google Scholar]
- Langeron M. 1916. Remarques sur les larves de Culex geniculatus et sur les larves de Culicinés pourvus de long siphon. Annales de Parasitologie Humaine et Comparée, 9, 438–442. [Google Scholar]
- Langeron M. 1921. Deuxième mission parasitologique en Tunisie, Tamerza (Septembre-Octobre 1919). Archives De l’Institut Pasteur de Tunis, 1, 347–382. [Google Scholar]
- Lounaci Z, Doumandji S, Doumandji-Mitiche B, Taguemout M. 2016. Bioecology of Culicidae (Diptera; Nematocera) of Taksebt dam of Tizi-Ouzou (Algeria). Journal of Entomology and Zoology Studies , 4, 20–29. [Google Scholar]
- Mahmoud DM, Hussein HM, El Gozamy BMR, Thabet HS, Hassan MA, Meselhey RA-A. 2019. Screening of Plasmodium parasite in vectors and humans in three villages in Aswan Governorate, Egypt. Journal of Parasitic Diseases, 43, 158–163. [Google Scholar]
- Marchant P, Eling W, Gemert GJ van, Leake CJ, Curtis CF. 1998. Could British mosquitoes transmit falciparum malaria? Parasitology Today, 14, 344–345. [Google Scholar]
- Medlock JM, Hansford KM, Versteirt V, Cull B, Kampen H, Fontenille D, Hendrickx G, Zeller H, Van Bortel W, Schaffner F. 2015. An entomological review of invasive mosquitoes in Europe. Bulletin of Entomological Research, 105, 637–663. [Google Scholar]
- Medlock JM, Snow KR, Leach S. 2005. Potential transmission of West Nile virus in the British Isles: an ecological review of candidate mosquito bridge vectors. Medical and Veterinary Entomology, 19, 2–21. [Google Scholar]
- Merabti B, Boumaza M, Ouakid ML, Carvajal TM, Harbach RE. 2021. An updated checklist of the mosquitoes (Diptera: Culicidae) present in Algeria, with assessments of doubtful records and problematic species. Zootaxa, 5027, 515–545. [CrossRef] [PubMed] [Google Scholar]
- M’ghirbi Y, Mousson L, Moutailler S, Lecollinet S, Amaral R, Beck C, Aounallah H, Amara M, Chabchoub A, Rhim A, Failloux A-B, Bouattour A. 2023. West Nile, sindbis and usutu viruses: Evidence of circulation in mosquitoes and horses in Tunisia. Pathogens, 12, 360. [Google Scholar]
- Moussiegt O. 1983. Les moustiques de Tunisie, leur répartition, bibliographie. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 47, 3–29. [Google Scholar]
- Moussiégt O. 1987. Aedes (Ochlerotatus) detritus Haliday, 1833: bibliographie. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 54, 43. [Google Scholar]
- Moussiegt O. 1991. Les espèces françaises de moustiques des creux d’arbres. Bibliographie. Revue Française d’Entomologie (N.S.), 13, 1–15. [Google Scholar]
- Moutailler S, Krida G, Schaffner F, Vazeille M, Failloux A-B. 2008. Potential vectors of Rift Valley fever virus in the Mediterranean region. Vector Borne and Zoonotic Diseases, 8, 749–753. [Google Scholar]
- Nebbak A, Almeras L, Parola P, Bitam I. 2022. Mosquito vectors (Diptera: Culicidae) and mosquito-borne diseases in North Africa. Insects, 13, 962. [Google Scholar]
- Ngoagouni C, Kamgang B, Manirakiza A, Nangouma A, Paupy C, Nakoune E, Kazanji M. 2012. Entomological profile of yellow fever epidemics in the Central African Republic, 2006–2010. Parasites & Vectors, 5, 175. [Google Scholar]
- Nikookar SH, Charkame A, Nezammahalleh A, Moradi-Asl E, Enayati A, Fazeli-Dinan M, Sedaghat MM, Zaim M. 2023. Entomological surveillance of invasive Aedes mosquitoes in Mazandaran Province, northern Iran from 2014 to 2020. Scientific Reports, 13, 8683. [Google Scholar]
- Outammassine A, Zouhair S, Loqman S. 2022. Global potential distribution of three underappreciated arboviruses vectors (Aedes japonicus, Aedes vexans and Aedes vittatus) under current and future climate conditions. Transboundary and Emerging Diseases, 69 (4), e1160–e1171. [Google Scholar]
- Pagac BB, Spring AR, Stawicki JR, Dinh TL, Lura T, Kavanaugh MD, Pecor DB, Justi SA, Linton Y-M. 2021. Incursion and establishment of the Old World arbovirus vector Aedes (Fredwardsius) vittatus (Bigot, 1861) in the Americas. Acta Tropica, 213, 105739. [Google Scholar]
- Paronyan L, Babayan L, Manucharyan A, Manukyan D, Vardanyan H, Melik-Andrasyan G, Schaffner F, Robert V. 2020. The mosquitoes of Armenia: review of knowledge and results of a field survey with first report of Aedes albopictus. Parasite, 27, 42. [Google Scholar]
- Pasteur N, Verdier JM, Rioux JA, Guilvard E, Perieres J. 1978. Le complexe Aedes detritus (Haliday, 1833) : existence de deux espèces jumelles en Afrique du Nord. Annales de Parasitologie humaine et Comparée, 53, 761–763. [Google Scholar]
- Paupy C, Delatte H, Bagny L, Corbel V, Fontenille D. 2009. Aedes albopictus, an arbovirus vector: from the darkness to the light. Microbes and Infection, 11, 1177–1185. [CrossRef] [PubMed] [Google Scholar]
- Ramsdale C. 1990. Anopheles mosquitoes and imported malaria in Libya. Mosquito Systematics, 22, 35–42. [Google Scholar]
- Ramsdale CD, Zulueta J de. 1983. Anophelism in the Algerian Sahara and some implications of the construction of a trans-Saharan highway. Journal of Tropical Medicine and Hygiene, 86, 51–58. [Google Scholar]
- Reinert JF. 2000. New classification for the composite genus Aedes (Diptera: Culicidae: Aedini), elevation of subgenus Ochlerotatus to generic rank, reclassification of the other subgenera, and notes on certain subgenera and species. Journal of the American Mosquito Control Association, 16, 175–188. [Google Scholar]
- Reinert JF, Harbach RE, Kitching IJ. 2006. Phylogeny and classification of Finlaya and allied taxa (Diptera: Culicidae: Aedini) based on morphological data from all life stages. Zoological Journal of the Linnean Society, 148, 1–101. [Google Scholar]
- Reinert JF, Harbach RE, Kitching IJ. 2009. Phylogeny and classification of tribe Aedini (Diptera: Culicidae). Zoological Journal of the Linnean Society, 157, 700–794. [Google Scholar]
- Rioux JA. 1958. Les Culicides du « Midi » méditerranéen. Étude systématique et écologique, in Encyclopédie Entomologique. Paris: Lechevalier. p. 1–320. [Google Scholar]
- Rioux JA, Cousserans J, Croset H, Osman FB, Gabinaud A, Sinegre G, Belmonte A. 1975. The autogenic character of Aedes pullatus (Coquillett, 1904) and new geographic localizations for Aedes caspius (Pallas, 1771), Aedes mariae (Sergent and Sergent, 1903), Aedes detritus (Haliday, 1833) and Culiseta subochrea (Edwards, 1921). Annales de Parasitologie Humaine et Comparée, 50, 131–133. [Google Scholar]
- Rioux JA, Dedet JP, Ben Osman F. 1968. Présence de Culiseta subochrea (Edwards, 1921) et Culex theileri (Theobald, 1903) dans l’extrême sud de la Tunisie. Archives de l’Institut Pasteur de Tunis, 45, 419–421. [Google Scholar]
- Rioux JA, Guilvard E, Pasteur N. 1998. Description d'Aedes (Ochlerotatus) coluzzii n. sp. (Diptera, Culicidae), espèce jumelle a du complexe detritus. Parassitologia, 40, 353–360. [PubMed] [Google Scholar]
- Rioux JA, Juminer B. 1963. Contribution à l'étude des Culicides du Hoggar. Archives de l’Institut Pasteur de Tunis, 40, 217–228. [Google Scholar]
- Rioux JA, Juminer B, Kchouk M. 1964. Anopheles (A.) plumbeus Stephens, 1928, Aedes (O.) berlandi Seguy, 1921 et Aedes (F.) geniculatus (Olivier, 1791) : culicides nouveaux pour la Tunisie. Archives de l’Institut Pasteur de Tunis, 41, 5–22. [Google Scholar]
- Rioux JA, Kchouk M, Croset H, Ben Osman F. 1974. Présence du caractère autogène dans les populations tunisiennes de Culiseta (C.) subochrea (Edw., 1921) (Diptera, Culicidae). Bulletin de la Société de Pathologie Exotique, 49, 373–375. [Google Scholar]
- Robert V, Günay F, Le Goff G, Boussès P, Sulesco T, Khalin A, Medlock J, Kampen H, Petrić D, Schaffner F. 2019. Distribution chart for Euro-Mediterranean mosquitoes (western Palaearctic region). Journal of the European Mosquito Control Association, 37, 1–28. [Google Scholar]
- Roiz D, Vazquez A, Rosà R, Muñoz J, Arnoldi D, Rosso F, Figuerola J, Tenorio A, Rizzoli A. 2012. Blood meal analysis, flavivirus screening, and influence of meteorological variables on the dynamics of potential mosquito vectors of West Nile virus in northern Italy. Journal of Vector Ecology, 37, 20–28. [Google Scholar]
- Romi R, Boccolini D, Vallorani R, Severini F, Toma L, Cocchi M, Tamburro A, Messeri G, Crisci A, Angeli L, Costantini R, Raffaelli I, Pontuale G, Thiéry I, Landier A, Le Goff G, Fausto AM, Di Luca M. 2012. Assessment of the risk of malaria re-introduction in the Maremma plain (Central Italy) using a multi-factorial approach. Malaria Journal, 11, 98. [Google Scholar]
- Rudolf I, Šebesta O, Straková P, Betášová L, Blažejová H, VEnclíková K, Seidel B, Tóth S, Hubálek Z, Schaffner F. 2015. Overwintering of Uranotaenia Unguiculata adult females in Central Europe: A possible way of persistence of the putative new lineage of West Nile virus? Journal of the American Mosquito Control Association, 31, 364–365. [Google Scholar]
- Schaffner F, Angel G, Geoffroy B, Hervy JP, Rhaiem A, Brunhes J. 2001. The mosquitoes of Europe : an identification and training programme. Paris: IRD Editions. [Google Scholar]
- Schaffner F, Thiéry I, Kaufmann C, Zettor A, Lengeler C, Mathis A, Bourgouin C. 2012. Anopheles plumbeus (Diptera: Culicidae) in Europe: a mere nuisance mosquito or potential malaria vector? Malaria Journal, 11, 393. [Google Scholar]
- Séguy E. 1920. Les moustiques de France. Bulletin du Muséum National d’Histoire Naturelle, 26, 51–58. [Google Scholar]
- Séguy E. 1923. Les moustiques de France. Paris: Éditions Lechevalier. [Google Scholar]
- Séguy E, Bouvier EL. 1924. Les Moustiques de l’Afrique mineure, de l’Égypte et de la Syrie. Paris: Edition Paul Lechevalier. [Google Scholar]
- Senevet G. 1947. The genus Culex in North Africa; Larvae. Archives de l’Institut Pasteur d’Algérie, 25, 107–136. [Google Scholar]
- Senevet G, Andarelli L. 1954. Le genre Aedes en Afrique du Nord. I. Les larves. Archives de l’Institut Pasteur d’Algérie. 32, 310–351. [Google Scholar]
- Senevet G, Andarelli L. 1960. Contribution à l’étude de la biologie des moustiques en Algérie et dans le Sahara algérien. Archives de l’Institut Pasteur d’Algérie, 38, 305–326. [Google Scholar]
- Senevet G, Andarelli L. 1963. Les moustiques de l’Afrique du nord et du bassin méditerranéen. 3. les Aedes. Archives de l’Institut Pasteur d’Algérie, 41, 115–141. [Google Scholar]
- Senevet G, Andarelli L. 1964. Les moustiques de l’Afrique du nord et du bassin méditerranéen. III. - Les Aedes. Deuxième partie (suite) : Description des espèces : sous-genre Ochlerotatus, groupe e. Archives de L’Institut Pasteur d’Algérie, 42, 1–79. [Google Scholar]
- Senevet G, Andarelli L, Abonnenc E. 1955. Les soies antépalmées chez les larves d’Anophèles. Leur utilisation taxinomique. Archives de l’Institut Pasteur d’Algérie, 33, 106–127. [Google Scholar]
- Senevet G, Andarelli L, Sergent E. 1956. Les Anophèles de l’Afrique du nord et du Bassin méditerranéen. Paris : Editions, Paul Lechevalier. [Google Scholar]
- Senevet G, Andarelli L, Sergent E. 1959. Les moustiques de l’Afrique du nord et du Bassin méditerranéen. Paris : Editions, Paul Lechevalier. [Google Scholar]
- Senevet G, Prunelle M. 1928. Larves de culicides recueillies en Algérie et en Tunisie par M. H. Gauthier. Archives de l’Institut Pasteur d’Algérie, 19, 94–99. [Google Scholar]
- Sergent E. 1909. Liste des moustiques de l’Afrique du Nord (1re note). Bulletin de la Société de Pathologie Exotique, 2, 440–448. [Google Scholar]
- Seufi AM, Galal FH. 2010. Role of Culex and Anopheles mosquito species as potential vectors of Rift Valley fever virus in Sudan outbreak, 2007. BMC Infectious Diseases, 10, 65. [Google Scholar]
- Seurat L.G. 1942. Faune des eaux continentales de la Tunisie. Eaux souterraines. Archives de l’Institut Pasteur de Tunis, 31, 311–335. [Google Scholar]
- Sicart M. 1940. Notes sur la présence de Culex (Culiciomyia) impudicus Ficalbi en Tunisie. Archives de l’Institut Pasteur de Tunis, 17, 471–474. [Google Scholar]
- Sicart M. 1941. Contribution à l’études des Anophèles de Tunisie. Présence de Anopheles (A.) marteri (Senevet et Prunelles, 1921). Archives de l’Institut Pasteur de Tunis, 30, 132–134. [Google Scholar]
- Sicart M. 1942. Contribution à l’étude des anophèles de Tunisie. Présence de Anopheles (A.) claviger (Meigen, 1884). Archives de l’Institut Pasteur de Tunis, 31, 132–134. [Google Scholar]
- Sicart M. 1949. Clé dichotomique et notes sur les Anopheles de Tunisie. Ministère de la Santé Publique, Service antipaludique. pp. 3–15. [Google Scholar]
- Sicart M. 1951. Note sur des larves d’Anopheles maculipennis présentant un appendice thoracique. Bulletin de la Société de Sciences Naturelles de Tunisie, 4, 45–48. [Google Scholar]
- Sicart M. 1952. Description et étude d’Aedes pulchritarsis (Rondani, 1872) récolté en Tunisie. Bulletin de la Société de Sciences Naturelles de Tunisie, 5, 95–101. [Google Scholar]
- Sicart M. 1952. Sur la présence de Aedes zammittii et Aedes echinus en Tunisie. Bulletin de la Société d’Histoire Naturelle de Tunisie, 5, 109–110. [Google Scholar]
- Sicart M. 1953. Contribution à l’étude des nymphes du genre Culex en Tunisie. Bulletin de la Société des Sciences Naturelles de Tunisie, 7, 27–29. [Google Scholar]
- Sicart M, Sardou R. 1961. Anomalies de gîtes et d’associations larvaires chez des Culicidés. Bulletin de la Société d’Histoire Naturelle de Toulouse, 96(3–4), 273–274. [Google Scholar]
- Sinka ME, Bangs MJ, Manguin S, Coetzee M, Mbogo CM, Hemingway J, Patil AP, Temperley WH, Gething PW, Kabaria CW, Okara RM, Van Boeckel T, Godfray HCJ, Harbach RE, Hay SI. 2010. The dominant Anopheles vectors of human malaria in Africa, Europe and the Middle East: occurrence data, distribution maps and bionomic précis. Parasites & Vectors, 3, 117. [Google Scholar]
- Somboon P, Phanitchakun T, Namgay R, Harbach RE. 2023. Review of the Mimeticus subgroup of Culex (Culex) (Diptera: Culicidae), with an assessment of the specific status of three nominal species described from India. Journal of Medical Entomology, 60, 255–271. [Google Scholar]
- Soni S, Gill VJS, Anusheel Singh J, Chhabra J, Gill GJS, Bakshi R. 2023. Dengue, chikungunya, and Zika: The causes and threats of emerging and re-emerging arboviral diseases. Cureus, 15, e41717. [Google Scholar]
- Sudeep AB, Mohandas S, Bhanarkar SR, Ghodke YS, Sonawane PA. 2020. Vector competence of Aedes vittatus (Bigot) mosquitoes from India for Japanese encephalitis, West Nile, Chandipura and Chittoor viruses. Journal of Vector Borne Diseases, 57, 234–239. [Google Scholar]
- Sudeep AB, Shil P. 2017. Aedes vittatus (Bigot) mosquito: An emerging threat to public health. Journal of Vector Borne Diseases, 54, 295–300. [Google Scholar]
- Tabachnick WJ. 1991. Evolutionary genetics and arthropod-borne disease: The yellow fever mosquito. American Entomologist, 37, 14–26. [Google Scholar]
- Tabbabi A, Boussès P, Rhim A, Brengues C, Daaboub J, Ben-Alaya-Bouafif N, Fontenille D, Bouratbine A, Simard F, Aoun K. 2015. Larval habitats characterization and species composition of Anopheles mosquitoes in Tunisia, with particular attention to Anopheles maculipennis complex. American Journal of Tropical Medicine and Hygiene, 92, 653–659. [Google Scholar]
- Tagliapietra V, Arnoldi D, Di Luca M, Toma L, Rizzoli A. 2019. Investigation on potential malaria vectors (Anopheles spp.) in the province of Trento, Italy, Malaria Journal, 18, 1–5. [CrossRef] [PubMed] [Google Scholar]
- Tchicha B. 2011. Historique de la Lutte Antipaludique en Algérie. Alger: Ministère de la Santé, de la Population et de la Réforme Hospitalière. [Google Scholar]
- Tedjou AN, Kamgang B, Yougang AP, Njiokou F, Wondji CS. 2019. Update on the geographical distribution and prevalence of Aedes aegypti and Aedes albopictus (Diptera: Culicidae), two major arbovirus vectors in Cameroon. PLOS Neglected Tropical Diseases, 13, e0007137. [Google Scholar]
- Theobald FV. 1901. A monograph of the Culicidae, or mosquitoes. Printed by order of the Trustees: London. [Google Scholar]
- Trari B, Carnevale P. 2011. De la préélimination à l’élimination du paludisme au Maroc. Quels risques pour l’avenir ? Bulletin de la Société de Pathologie Exotique, 104, 291–295. [Google Scholar]
- Trari B, Dakki M. 2017. Atlas des Moustiques du Maroc: distribution, bioécologie et rôle vecteur. Rabat: Travaux de l’Institut Scientifique. [Google Scholar]
- Trari B, Dakki M, Harbach RE. 2017. An updated checklist of the Culicidae (Diptera) of Morocco, with notes on species of historical and current medical importance. Journal of Vector Ecology, 42, 94–104. [Google Scholar]
- Trari B, Dakki M, Himmi O, El Agbani MA. 2002. Les moustiques (Diptera Culicidae) du Maroc. Revue bibliographique (1916–2001) et inventaire des espèces. Bulletin de la Société de Pathologie Exotique, 95, 101–120. [Google Scholar]
- Triki H, Murri S, Le Guenno B, Bahri O, Hili K, Sidhom M, Dellagi K. 2001. West Nile viral meningo-encephalitis in Tunisia. Médecine Tropicale, 61, 487–490. [Google Scholar]
- Vazeille M, Jeannin C, Martin E, Schaffner F, Failloux A-B. 2008. Chikungunya: a risk for Mediterranean countries? Acta Tropica, 105, 200–202. [Google Scholar]
- Vermeil C. 1953. Aedes vittatus Bigot, 1861, Culicide nouveau pour la Tunisie. Bulletin de la Société de Pathologie Exotique, 46, 338–339. [Google Scholar]
- Vermeil C. 1953. Nouvelle localité tunisienne pour Uranotaenia unguiculata Edwards, 1913. Bulletin de la Société de Pathologie Exotique, 28, 240. [Google Scholar]
- Vermeil C. 1953. De la reproduction par autogénèse chez Aedes (O.) detritus Haliday. Bulletin de la Société de Pathologie Exotique, 46, 971–973. [Google Scholar]
- Vermeil C. 1954. Faune parasitologique des îles de Zembra et Zembretta. Bulletin de la Société de Pathologie Exotique, 2, 47–56. [Google Scholar]
- Vermeil CF. 1950. Contribution à l’études des culicides de Tunisie, Thèse de médecine. Université de Strasbourg. [Google Scholar]
- Villain G. 1932. Note sur la présence de larves d’anophélines dans les eaux salées du Sahel Tunisien. Bulletin de la Société de Pathologie Exotique, 11, 346–352. [Google Scholar]
- Villain G, Dupoux R, Marini C. 1935. Contribution à l’étude de l’anophélisme tunisien et aperçu de la lutte antianophèlienne dans la Régence. Archives de l’Institut Pasteur de Tunis, 24, 30–42. [Google Scholar]
- Wasfi F, Dowall S, Ghabbari T, Bosworth A, Chakroun M, Varghese A, Tiouiri H, Ben Jemaa M, Znazen A, Hewson R, Zhioua E, Letaief A. 2016. Sero-epidemiological survey of Crimean-Congo hemorrhagic fever virus in Tunisia. Parasite, 23, 10. [Google Scholar]
- Wassilieff A. 1934. Quelques remarques sur les moustiques de Tunisie. Archives de l’Institut Pasteur de Tunis, 23, 369–383. [Google Scholar]
- Wernsdorfer WH, McGregor IA. 1988. Malaria: principles and practice of malariology. Edinburgh: Churchill Livingstone. [Google Scholar]
- Wilkerson RC, Linton Y-M. 2015. Elevation of Pseudoskusea, Rusticoidus and Protomacleaya to valid subgenera in the mosquito genus Aedes based on taxon naming criteria recently applied to other members of the Tribe Aedini (Diptera: Culicidae). Parasites & Vectors, 8, 1–4. [CrossRef] [PubMed] [Google Scholar]
- Wint W, Jones P, Kraemer M, Alexander N, Schaffner F. 2022. Past, present and future distribution of the yellow fever mosquito Aedes aegypti: The European paradox. Science of the Total Environment, 847, 157566. [CrossRef] [Google Scholar]
- World Health Organization. 2013. Larval source management: a supplementary measurefor malaria vector control – An operational manual. Geneva: WHO, ISBN: 9789241505604. [Google Scholar]
- World Health Organization. 2021. Evaluation of genetically modified mosquitoes for the control of vector-borne diseases. Geneva, Switzerland: World Health Organization. Available at: https://www.who.int/publications/i/item/9789240013155. [Google Scholar]
- World Health Organization. 2020. World malaria report 2020. Geneva, Switzerland: World Health Organization. Available at: https://www.who.int/publications/i/item/9789240015791. [Google Scholar]
- Wöhnke E, Vasic A, Raileanu C, Holicki CM, Tews BA, Silaghi C. 2020. Comparison of vector competence of Aedes vexans Green River and Culex pipiens biotype pipiens for West Nile virus lineages 1 and 2. Zoonoses Public Health, 67 (4), 416–424. [Google Scholar]
Cite this article as: Ouni A, Rhim A, M’Ghirbi Y, Schaffner F & Bouattour A. 2026. The mosquitoes (Diptera: Culicidae) of Tunisia: updated checklist and new distribution data. Parasite 33, 20. https://doi.org/10.1051/parasite/2026018.
All Figures
 |
Figure 1 Bioclimatic map of Tunisia showing mosquito sampling locations (2013–2023), produced using QGIS Software (Lima 3.32.3). |
| In the text | |
 |
Figure S1. Geographical distribution of Anopheles algeriensis in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S2. Geographical distribution of Anopheles spp. [Anopheles algeriensis, Anopheles cinereus, Anopheles multicolor, Anopheles plumbeus and Anopheles sergentii] in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S3. Geographical distribution of Anopheles labranchiae in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S4. Geographical distribution of Anopheles multicolor in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S5. Geographical distribution of Aedes albopictus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S6. Geographical distribution of selected Aedes spp. [Aedes berlandi, Aedes dorsalis, Aedes vexans and Aedes vitatus] recorded in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S7. Geographical distribution of Aedes caspius in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S8. Geographical distribution of Aedes detritus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S9. Geographical distribution of different Culex species [Culex deserticola, Culex impudicus, Culex laticinctus, Culex mimeticus and Culex pusillus] in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S10. Geographical distribution of Culex perexiguus in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S11. Geographical distribution of Culex pipiens in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S12. Geographical distribution of Culex theileri in Tunisia (2013–2023), by life stage (larvae and adults) across the different bioclimatic zones. |
| In the text | |
 |
Figure S13. Geographical distribution of Culiseta longiareolata in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
 |
Figure S14. Geographical distribution of Orthopodomyia pulcripalpis and Uranotaenia unguiculata in Tunisia (2013–2023) across the different bioclimatic zones. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


