| Issue |
Parasite
Volume 27, 2020
|
|
|---|---|---|
| Article Number | 50 | |
| Number of page(s) | 16 | |
| DOI | https://doi.org/10.1051/parasite/2020046 | |
| Published online | 18 August 2020 | |
urn:lsid:zoobank.org:pub:7589B476-E0EB-4614-8BA1-64F8CD0A1BB2
Research Article
No vagina, one vagina, or multiple vaginae? An integrative study of Pseudaxine trachuri (Monogenea, Gastrocotylidae) leads to a better understanding of the systematics of Pseudaxine and related genera
Pas de vagin, un vagin ou plusieurs vagins ? Une étude intégrative de Pseudaxine trachuri (Monogenea, Gastrocotylidae) mène à une meilleure compréhension de la systématique de Pseudaxine et des genres apparentés
1
Université des Sciences et de la Technologie Houari Boumediene, Faculté des Sciences Biologiques, Laboratoire de Biodiversité et Environnement: Interactions – Génomes, BP 32, El Alia, Bab Ezzouar, 16111 Alger, Algérie
2
Service de Systématique Moléculaire, UMS 2700 CNRS, Muséum National d’Histoire Naturelle, Sorbonne Universités, 43 Rue Cuvier, CP 26, 75231 Paris Cedex 05, France
3
UMR7245 MCAM, Muséum National d’Histoire Naturelle, 61, Rue Buffon, CP52, 75231 Paris Cedex 05, France
4
Institut Systématique Évolution Biodiversité (ISYEB), Muséum National d’Histoire Naturelle, CNRS, Sorbonne Université, EPHE, Université des Antilles, 57 Rue Cuvier, CP 51, 75231 Paris Cedex 05, France
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
27
May
2020
Accepted:
24
July
2020
Abstract
The presence/absence and number of vaginae is a major characteristic for the systematics of the Monogenea. Three gastrocotylid genera share similar morphology and anatomy but are distinguished by this character: Pseudaxine Parona & Perugia, 1890 has no vagina, Allogastrocotyle Nasir & Fuentes Zambrano, 1983 has two vaginae, and Pseudaxinoides Lebedev, 1968 has multiple vaginae. In the course of a study of Pseudaxine trachuri Parona & Perugia 1890, we found specimens with structures resembling “multiple vaginae”; we compared them with specimens without vaginae in terms of both morphology and molecular characterisitics (COI barcode), and found that they belonged to the same species. We also investigated the male copulatory organ (MCO) of this species, the accuracy of the original description of which is known to be a matter of debate. We found that the genital atrium is armed with 12 hooks arranged as a single circle and a central hollow stylet which is probably involved in traumatic insemination. We redescribed Pseudaxine trachuri based on newly collected specimens from off the coast of Algeria and Museum specimens from off France. Specimens from the type-host, Trachurus trachurus, were found to be similar, for both molecular sequences and morphology, to those found on Boops boops. We can therefore confirm, for the first time with molecular evidence, that B. boops is a host of this parasite. We consider that Pseudaxinoides was erected on the basis of an erroneous interpretation of structures which are not vaginae and, consequently, propose the transfer of most of its species to Pseudaxine, as P. australis (Lebedev, 1968) n. comb., P. bychowskyi (Lebedev, 1977) n. comb., P. caballeroi (Lebedev, 1977) n. comb., P. cariacoensis (Nasir & Fuentes-Zambrano, 1983) n. comb., and P. vietnamensis (Lebedev, Parukhin & Roitman, 1970) n. comb. We also propose Allogastrocotyle dillonhargisorum nom. nov. for Pseudaxine bivaginalis Dillon & Hargis, 1965 to avoid a secondary homonymy.
Résumé
La présence/absence et le nombre de vagins est une caractéristique majeure de la systématique des Monogènes. Trois genres de Gastrocotylidae partagent une morphologie et une anatomie similaires mais se distinguent par ce caractère : Pseudaxine Parona & Perugia, 1890 n’a pas de vagin, Allogastrocotyle Nasir & Fuentes Zambrano, 1983 a deux vagins et Pseudaxinoides Lebedev, 1968 a plusieurs vagins. Au cours d’une étude de Pseudaxine trachuri Parona & Perugia 1890, nous avons trouvé des spécimens avec des structures ressemblant à des « vagins multiples ». Nous les avons comparés, pour la morphologie et les molécules (code à barres COI), à des spécimens sans vagin et avons constaté qu’ils appartenaient à la même espèce. Nous avons également examiné l’organe copulateur mâle (OCM) de cette espèce, dont l’exactitude de la description originale est connue pour être un sujet de débat. Nous avons constaté que l’atrium génital est armé de 12 crochets disposés en un seul cercle et d’un stylet central creux qui est probablement impliqué dans l’insémination traumatique. Nous avons redécrit Pseudaxine trachuri à partir de spécimens nouvellement collectés des côtes algériennes et de spécimens de Musée des côtes de France. Les spécimens de l’hôte type, Trachurus trachurus, se sont avérés similaires, pour les molécules et la morphologie, à ceux trouvés sur Boops boops. Nous pouvons donc confirmer, pour la première fois avec des preuves moléculaires, que B. boops est un hôte de ce parasite. Nous considérons que Pseudaxinoides a été érigé sur la base d’une interprétation erronée de structures qui ne sont pas des vagins et, par conséquent, proposons le transfert de la plupart de ses espèces à Pseudaxine, comme P. australis (Lebedev, 1968) n. comb., P. bychowskyi (Lebedev, 1977) n. comb., P. caballeroi (Lebedev, 1977) n. comb., P. cariacoensis (Nasir et Fuentes-Zambrano, 1983) n. comb. et P. vietnamensis (Lebedev, Parukhin & Roitman, 1970) n. comb. Nous proposons également Allogastrocotyle dillonhargisorum nom. nov. pour Pseudaxine bivaginalis Dillon & Hargis, 1965, pour éviter une homonymie secondaire.
Key words: Monogenea / Gastrocotylidae / Pseudaxine / Pseudaxinoides / Allogastrocotyle / Vagina / Mediterranean Sea / Molecular barcoding
© C. Bouguerche et al., published by EDP Sciences, 2020
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Pseudaxine Parona & Perugia, 1890, Allogastrocotyle Nasir & Fuentes Zambrano, 1983 and Pseudaxinoides Lebedev, 1968 are three genera that belong to the family Gastrocotylidae Price, 1943. Species of these three genera share a general boot-shaped appearance, because of their oblique haptor, and differ mainly in the presence and number of vaginae. The organisation of internal organs is similar, especially the testes and the male copulatory organ (MCO); however, members of Pseudaxine have no vagina, the single member of Allogastrocotyle has two vaginae, and species of Pseudaxinoides are said to have multiple ventro-lateral vaginae [49, 60, 63, 66].
Pseudaxine was established with Pseudaxine trachuri Parona & Perugia, 1890, from the gills of Trachurus trachurus off Genoa, Italy, as the type species [63]. This species was redescribed several times [53, 64, 67, 70, 76, 77]. One confusing aspect of this parasite was the armature of the genital atrium, which was described as “consisting of two crowns of hooks” [63], whereas a single crown was observed in subsequent redescriptions.
During an ongoing investigation on monogeneans infesting the gills of teleosts off the Algerian coast, we collected gastrocotylids consistent with the diagnosis of P. trachuri from gills of the type-host, the carangid T. trachurus, and also from a sparid, Boops boops. Specimens from both hosts were submitted to molecular analysis. Based on this material, we provide a detailed study of the genital atrium of P. trachuri and describe for the first time in this species a central stylet.
Unexpectedly, we found several specimens with multiple ventro-lateral papillae, without ducts, which mimicked vaginae. According to the diagnostic criteria of gastrocotylid genera [53], these specimens of Pseudaxine trachuri should have been attributed to Pseudaxinoides. We compared measurements and COI sequences of these specimens with “multiple vaginae” with typical specimens of Pseudaxine trachuri without vaginae, and found that they belonged to the same species. This clearly questioned the alleged differences between Pseudaxine and Pseudaxinoides, and we finally propose a series of nomenclatural decisions, including the demise of Pseudaxinoides.
This work is part of a general survey of the monogenean fauna of fishes of the southern shores of the Mediterranean Sea [5, 7–10, 15–18, 22, 44].
Materials and methods
Fishes
During the years 2016 and 2019, 2004 specimens of B. boops and 309 T. trachurus were collected from off Zemmouri El Bahri (36°48′4.582033″ N, 3°34′7.01″ E), Bouharoun (36°37′24″ N, 2°39′17″ E), Cherchell (36°36′31″ N, 2°11′50″ E), Dellys (36°54′48″ N, 3°54′51″ E), and Cap Djinet (36°52′37″ N, 3°43′23″ E) off the Algerian coast. Fish specimens were purchased dead from fishermen, transferred to the laboratory shortly after capture, and identified using keys [26]. Gills were removed from each fish and observed under a stereo microscope for the presence of monogeneans.
Monogeneans
Morphological methods
Monogeneans were removed from gills using fine dissection needles, killed in hot seawater, then preserved in 70% ethanol, stained with acetic carmine, dehydrated in ethanol series (70, 96 and 100%), cleared in clove oil, and finally mounted in Canada balsam. Drawings were made with the help of an Olympus BH-2 microscope equipped with Differential Interference Contrast (DIC) and a drawing tube. Micrographs were taken with a Leitz microscope. Drawings were scanned and redrawn on a computer with Adobe Illustrator (CS5). Measurements are in micrometres, and indicated as the means, ± the standard deviation if n > 30, and in parentheses, the range and number of measurements.
Molecular methods
To ensure that hosts and parasites were labelled with respect to their host-parasites relationships, we followed recent works dealing with description of new species of Monogenea by using barcoding [5, 8–10, 43]. For each host fish from which monogeneans were collected, a tissue sample from the gill was taken. Monogeneans were cut into three parts using a scalpel blade. The anterior part (which includes the genital atrium) and posterior part (which includes the haptor) of each specimen were mounted as vouchers (both parts on a single slide) for drawing and deposition in a Museum; their middle part was preserved in absolute ethanol then subjected to molecular analyses. For some specimens, we did not cut them in three part but rather took a lateral part in the middle of the body for the molecular analysis and mounted the rest of the specimen in one part as voucher. Nine specimens were analysed: 2 from the type-host T. trachurus (already published in our study of Allogastrocotyle [10]; 3 typical specimens from the sparid host B. boops, and 4 specimens with “multiple ventro-lateral vaginae” from B. boops.
Molecular barcoding of fish
Total genomic DNA was isolated using a QIAamp DNA Mini Kit (Qiagen, Courtaboeuf, France), as per the manufacturer’s instructions. The 5′ region of the mitochondrial cytochrome c oxidase subunit I (COI) gene was amplified with the primers FishF1 (5′ – TCAACCAACCACAAAGACATTGGCAC – 3′) and FishR1 (5′ – TAGACTTCTGGGTGGCCAAAGAATCA – 3′) [75]. PCR reactions were performed in 20 μL, containing 1 ng of DNA, 1× CoralLoad PCR buffer, 3 mM MgCl2, 66 μM of each dNTP, 0.15 μM of each primer, and 0.5 units of Taq DNA polymerase (Qiagen). The amplification protocol was 4 min at 94 °C, followed by 40 cycles at 94 °C for 30 s, 48 °C for 40 s, and 72 °C for 50 s, with a final extension at 72 °C for 7 min. PCR products were purified (Ampure XP Kit, Beckman Coulter) and sequenced in both directions on a 3730 × l DNA Analyzer 96-capillary sequencer (Applied Biosystems, Foster City, CA, USA). We used CodonCode Aligner version 3.7.1 software (Codon Code Corporation, Dedham, MA, USA) to edit sequences, compared them to the GenBank database content with BLAST, and deposited them in GenBank under accession numbers MT666082–MT666086. Species identification was confirmed with the BOLD identification engine [68].
COI sequences of monogeneans
Total genomic DNA was isolated using a QIAmp DNA Micro Kit (Qiagen). The specific primers JB3 (=COIASmit1) (forward 5′ – TTTTTTGGGCATCCTGAGGTTTAT – 3′) and JB4.5 (=COI-ASmit2) (reverse 5′ – TAAAGAAAGAACATAATGAAAATG – 3′) were used to amplify a fragment of 402 bp of the COI gene [11, 55]. PCR reaction was performed in 20 μL of a mixture containing 1 ng of DNA, 1× CoralLoad PCR buffer, 3 mM MgCl2, 0.25 mM dNTP, 0.15 μM of each primer, and 0.5 units of Taq DNA polymerase (Qiagen). Thermocycles consisted of an initial denaturation step at 94 °C for 2 min, followed by 37 cycles of denaturation at 94 °C for 30 s, annealing at 48 °C for 40 s, and extension at 72 °C for 50 s. The final extension was conducted at 72 °C for 5 min. PCR products were purified (Ampure XP Kit, Beckman Coulter) and sequenced in both directions on a 3730 × l DNA Analyzer 96-capillary sequencer (Applied Biosystems, Foster City, CA, USA). We used CodonCode Aligner version 3.7.1 software (Codon Code Corporation, Dedham, MA, USA) to edit sequences, compared them to the GenBank database content with BLAST, and deposited them in GenBank under accession numbers MT666075–MT666081 (Table 1).
Fish, monogeneans and their COI sequences. To insure full traceability and ensure accurate host-parasite relationships, one or several monogeneans were collected from one fish and all individuals were sequenced. All monogeneans in this Table were identified as Pseudaxine trachuri.
Trees and distances
Most gastrocotylid sequences available in GenBank were included in a preliminary phylogenetic analysis, with 9 sequences of Pseudaxine trachuri, and sequences of two members of the Gotocotylidae Yamaguti, 1963 used as the outgroup. The trees were inferred by the Neighbour-Joining method and the Maximum Likelihood method using MEGA7 [48]. After examining trees and sequences, we refined our analysis and finally used only 12 sequences, as detailed in the results. For the final analysis, we employed the Neighbour-Joining method and the Maximum Likelihood method using MEGA7 [48]; after selecting the best model with MEGA7, the ML was performed with HKY + G [34]. Genetic distances (p-distance and Kimura-2 parameter distance [45]) were estimated with MEGA7 and all codon positions were used.
Results
Molecular identification of fish
The provisional identification of the fish species using morphological characteristics was confirmed by the DNA barcoding approach. BLAST analysis of the COI sequences of the fish species of the present study with the NCBI and BOLD databases showed sequence similarity values of 100% for B. boops and 99.85% for T. trachurus.
Molecular characterization of monogeneans
A preliminary dataset involved 20 COI sequences, including 9 sequences of P. trachuri and sequences from GenBank. After alignment and trimming, the dataset was 391 bp in length with 211 informative sites. The main result for this preliminary analysis was that our sequences of P. trachuri from off the Algerian coast formed a robust monophylum with the single sequence of P. trachuri available from the type-host T. trachurus off Sète, France (AY009168) [40]; this sequence is therefore confirmed as P. trachuri. However, we noted that sequence AY009167, also from T. trachurus off Sète, France, grouped with P. trachuri, although it was identified as Gastrocotyle trachuri Van Beneden & Hesse, 1863 and labelled as such in GenBank by its authors, Jovelin & Justine [40]; we interpret this as a misidentification, and sequence AY009167 should be considered P. trachuri.
We noticed that several GenBank sequences were unconfirmed and/or unpublished, and there was no certainty in the systematic assignment for some of them (in addition to the obvious error mentioned above); also, some had apparent insertions-deletions, or ambiguous bases, and the alignment included gaps. Therefore, from the preliminary dataset, we selected only the sequences that had the same span as our sequences and no ambiguous bases. The restricted dataset included only 12 sequences: 9 sequences of P. trachuri (including 7 new), 2 sequences of Allogastrocotyle bivaginalis, and the COI part of the complete mitogenome (NC016950) of the chauhaneid Pseudochauhanea macrorchis Lin, Liu & Zhang in Zhang, Yang & Liu, 2001 [81]. We obtained perfect alignment, 389 bp in length, without any gap or missing data. The tree (Fig. 1) showed a robust monophylum that includes all sequences of P. trachuri. The P. trachuri clade contained sequences from the type-host and from the sparid host B. boops, confirming the occurrence of P. trachuri on a sparid. Moreover, all specimens of P. trachuri with apparent “multiple ventro-lateral vaginae” and specimens without vaginae clustered in the same clade, without having a distinct branch.
 |
Figure 1 Molecular phylogenetic analysis of monogenean COI sequences using the Maximum Likelihood method. Bootstraps percentages (1000 replicates) are indicated next to the branches when higher than 70. There were a total of 389 positions in the final dataset. The Neighbour-Joining tree had a similar topology and is not represented. All specimens of Pseudaxine trachuri, whether with no vagina or with “multiple vaginae”, and whether from the type-host Trachurus trachurus or from Boops boops, were grouped into a single monophylum and showed little variation (<2%). |
Distances were computed using p-distance and the Kimura-2 parameter. All sequences of P. trachuri, whatever host or apparent number of vaginae, were very similar, with less than 2% variation. This suggests that P. trachuri also occurs on B. boops and that specimens with apparent multiple vaginae are conspecific with typical P. trachuri.
Morphology of Pseudaxine trachuri Parona & Perugia, 1890 (Figs. 2–4)
Type-host: Trachurus trachurus (Linnaeus, 1758) (Carangidae), Atlantic horse mackerel.
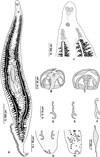 |
Figure 2 Pseudaxine trachuri Parona & Perugia, 1890. (A) Entire worm, typical specimen without a vagina, MNHN HEL1278; (B) clamp, ventral view; (C) clamp, dorsal view, MNHN HEL1278; (D–F) anchors from terminal lappet, MNHN HEL1279, HEL1280; (G and H), terminal lappet, MNHN HEL1280, HEL1281; (I) egg, MNHN HEL1282. (J) anterior extremity of a specimen from Boops boops, showing apparent “multiple lateral vaginae” (MNHN HEL1283). |
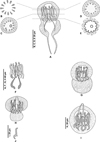 |
Figure 3 Pseudaxine trachuri, genital atrium shown laterally and in different optical sections and the variable position of the stylet. (A) Genital atrium, lateral view, MNHN HEL1284; (B–E), optical sections of a genital atrium in perfect apical view; the level of these sections are shown on the lateral view, MNHN HEL1285. (F) Lateral view of the atrium showing only the sclerotised parts, MNHN 306HG, Box 43, slide 51; (G–I) various views at different orientations, MNHN HEL1286, MNHN HEL1287, MNHN HEL1288. (J) One spine, MNHN HEL1287. |
 |
Figure 4 Clamp, with detailed legends, MNHN HEL1289. (A) Ventral jaw. (B) Dorsal jaw. (C) Whole clamp. |
Type-locality: Off Genoa (Genova), Italy.
Additional host: Boops boops (Linnaeus, 1758) (Sparidae), bogue.
Site on host: Gills.
Additional localities: off Zemmouri el Bahri (36°48′4.58″ N, 3°34′7.01″ E), Bouharoun (36°37′24″ N, 2°39′17″ E), Cherchell (36°36′31″ N, 2°11′50″ E), Dellys (36°54′48″ N, 3°54′51″ E) and Cap Djinet (36°52′37″ N, 3°43′23″ E), off the Algerian coast (present study).
Prevalence and intensity: in T. trachurus off Algeria: 28% (89/309), up to 3 monogeneans/fish; in B. boops off Algeria: 10% (201/2004), up to 2 monogeneans/fish.
Specimens from Algerian waters studied and deposited: 14 vouchers from the type-host, T. trachurus, including 2 with molecular information (MNHN HEL1093, GenBank MN192393, MNHN HEL1094, GenBank MN192394); 23 specimens from B. boops (HEL1270–HEL1292), including 7 with molecular information (HEL1270–HEL1276, GenBank MT666075–MT666081, Table 1). Vouchers with molecular information consist of the anterior and posterior parts of specimens mounted together on a slide; while the middle part was used for molecular analysis.
Comparative material studied: 6 specimens of P. trachuri from its type-host T. trachurus off Sète, French Mediterranean Coast; part of the collection of Prof. Louis Euzet deposited in the Muséum National d’Histoire Naturelle (MNHN 306HG, Box 43, slides 19, 51, 54, 55–63, 80, 81).
Description of specimens from Boops boops from off Algeria
Based on 30 specimens. Measurements in Table 2. Body elongate, typically gastrocotylid. Haptor, oblique, asymmetrical, with single row of clamps. Clamps typically gastrocotylid; ventral arm of median spring Y-shaped; proximal end of ventral arm of median spring narrow; its distal part Y-shaped; dorsal arm of median spring short, has 3–4 pairs of apertures arranged in 2 longitudinal, symmetrical, parallel rows; distal end of dorsal arm with accessory skeletal piece represented by V-shaped process at its distal end (Fig. 4). Dorsal arm of ventral jaw short and curved inwards. Dorsal jaw shorter than ventral. Oblique sclerites with inner ends touching each other in median line. Base of clamp with cuticularised hinge ligament on each side, connecting median spring with base of jaw sclerites (Fig. 4).
Measurements of Pseudaxine trachuri from different hosts and localities.
Terminal lappet present, armed with 2 pairs of anchors, dissimilar in shape and size (Figs. 2D–2F); anteriormost anchor pair large, situated at base of haptoral lappet; posteriormost anchor pair small, at tip of haptoral lappet (Figs. 2G–2H). No additional pair observed.
Pair of prohaptoral suckers sub-oval, muscular and septate. Pharynx pyriform. Intestinal caeca extend to posterior end of body, not confluent (Fig. 2A).
Testes postovarian, large and oval, arranged irregularly in intercaecal field in posterior third of body. Vas deferens passes forward along body midline and enlarges to form ejaculatory bulb at level just anterior to genital atrium (Figs. 3A, 3G and 3H).
Ovary pretesticular, in form of inverted “U”. Ovary begins at level of anteriormost testes, continues anteriorly in midline, reflexes and passes posteriorly toward haptor to terminate as oviduct. Oviduct arises from distal end of ovary and opens into oötype with Mehlis’ gland cells. Uterus, arises from oötype and passes anteriorly. Genito-intestinal canal short, ventral to ovary. Vitellarium follicular, co-extensive with intestinal caeca, extending from level of genital atrium to haptor. Transverse vitelloducts fuse in midline, ventral to ovary, to form Y-shaped vitelline reservoir. Vagina absent (Fig. 2J).
Genital atrium
Genital atrium ventral, with a muscular rim, located in the anterior constriction of the body. The genital atrium includes 2 sclerotised structures: a crown of 12 hooks and a central stylet. Gastrocotylid-like atrial hooks (Fig. 3J) are arranged as a perfect circle; this was seen in some specimens in which the hooks are aligned with the optical axis of the microscope, i.e. perpendicular to the longitudinal body axis. In these polar views, observation with a DIC microscope, which has a very low field depth, enables various optical sections, from distal to proximal: the distal (external) extremity of the hooks, with their points directed toward the centre; the middle part, with cross-sections of cylindrical hooks in circles; and basal region, with the blades of hooks forming a continuous circle. Observation of genital atria with a longitudinal orientation of the hooks confirms this morphology, with the pointed ends directed toward the centre and blade-like bases. The central stylet is a slender, conical structure, which appears in cross-sections as a hollow circle (Figs. 3C and 3E) and in longitudinal sections as a conical, pointed structure (Figs. 3A and 3F–3I). The position of the stylet is variable in respect to the crown of hooks and can protrude distally from it (Figs. 3H and 3I) or not (Figs. 3A, 3F and 3G). The stylet is connected basally with an unsclerotised bulb, which is itself connected at its base to the vas deferens (Fig. 3A). In flattened whole-mounts, the stylet is occasionally tilted such that it appears as a pair of slender spines (Fig. 3I).
Vagina and multiple pseudo-vaginae
In most specimens, a vagina was absent; the DIC microscope enables one to examine with precision the surface of the tegument and we did not detect any opening. In a series of specimens from B. boops, we found, however, multiple ventro-lateral papillae which could be interpreted as two rows of lateral vaginae. We tried to find a duct leading from these openings to the posterior parts, but did not detect it; instead, we noticed that flame cells were often close to these apertures. We consider, therefore, that these openings are pores of the excretory system, which mimic lateral vaginae.
Figure 5 shows micrographs of the region of the genital atrium and vaginae in P. trachuri (both specimens without a vagina and with alleged multiple vaginae) and in Allogastrocotyle bivaginalis.
 |
Figure 5 Micrographs of specimens showing the region of the genital atrium and vaginae (arrows). Left, Pseudaxine trachuri, no vagina visible; Middle, Pseudaxine trachuri, alleged multiple vaginae, two different focuses on the same specimen; Right, Allogastrocotyle bivaginalis, two real vaginae. Unscaled, same scale for all micrographs. |
Discussion
Boops boops as a host for Pseudaxine trachuri
Pseudaxine trachuri is a cosmopolitan monogenean, reported from many carangid hosts, mainly Trachurus spp., and from a single sparid, Boops boops (Table 3). In the present study, measurements and counts of P. trachuri from B. boops were within range of previous descriptions (Table 2), and COI sequences of P. trachuri from B. boops differed from those from the type-host by only 1–2%. Differences higher than 4% in COI sequences are generally interpreted as an indication that species are different [4, 33, 74, 75]; in polyopisthocotylean monogeneans, differences in COI sequences reported as intraspecific ranged 0.2–5.6% [8]. The 1–2% difference reported here is well within the intraspecific range. We thus confirm, for the first time with molecular evidence, that P. trachuri uses B. boops as a host. We noted that, in our specimens, the prevalence in B. boops was lower than in T. trachurus (10% vs. 28%), which could be an indication of a lower adaptation to the sparid host, or a recent host-switch.
General structure of the genital atrium of Pseudaxine trachuri
The original description of P. trachuri is apparently very detailed and beautifully illustrated, especially for a paper of the 19th Century [63]. It has been supplemented by redescriptions and more morphometric data by Yamaguti [76, 77], Sproston [70], Parukhin [64], Lebedev [53] and Radujkovic & Euzet [67]. However, the illustrations were sometimes poor or redrawn from the original 1890 account of Parona & Perugia [63].
Parona and Perugia indicated that the genital atrium was armed with two crowns of hooks [63]: “L’apertura cloacale […] coronata da un cerchio di 24 uncini minutissimi (Fig. 11), con base allargata e punta arcuata rivolta verso il lume del canale. Inferiormente e nell’interno della cloaca si osserva altro cerchio di aculei (Fig. 12) molto più lunghi dei superiori (0,017) che soprastanno all’apertura del deferente e che si può rigardare quale armatura maschile” which translates as “The cloacal opening […] is crowned by a circle of 24 very fine hooks (Fig. 11), with an enlarged base and the point turned towards the lumen of the canal. Beneath and inside the canal, another circle of spines is observed (Fig. 12), which are much longer than those above (0.017) and are present distal to the aperture of the vas deferens; these can be considered the male armature”.
Yamaguti redescribed the species twice, on the basis of two (1938) and one (1942) specimens, from the gills of the type-host off Japan [76, 77]. In both descriptions, Yamaguti clearly indicated that the genital atrium was surrounded by a single crown of hooks, but did not comment on the differences in the genital atrium.
Sproston (1946) redescribed P. trachuri from its type-host, based on a single specimen collected from off Plymouth, England, and indicated the presence of 16 atrial hooks. However, she reproduced in her paper one of the drawings by Parona & Perugia and commented on the original description: “these authors give a curious figure of the genital corona […] and describe it as having an outer ring of 24 small hooks and 17 larger hooks below at the end of the vas deferens”; she stated “neither the Japanese nor Plymouth specimens from the same host reveal anything like this structure”. In this sentence, Sproston mentioned 17 larger hooks, but Parona & Perugia did not provide the number of the larger hooks; a possible explanation is that Sproston confused the length (17 μm) and the number of the larger hooks in the original description in Italian, although the length was provided as “0.017”.
Unnithan [73], probably using Sproston’s text, wrote in his description of Pseudaxine kurra Unnithan, 1968 “no structure identical to the genital corona having an outer rim of 24 small hooks and 17 larger hooks below it at the end of the vas deferens is observed in the new species”.
Lebedev commented “The discussion of Sproston [70] and Unnithan [73] about the allegedly incorrect description of these structures by Parona and Perugia [63] seems unreasonable to us: if we accept that the outer ring of spines in the picture of the copulatory apparatus given in the first description is the apical parts of the hooks, and the inner ring is a stylet, then everything falls into place [53].
Radujkovic & Euzet, on the basis of specimens from off Montenegro, described briefly the genital atrium as having a crown of 12 spines [67].
Our careful examination of new specimens of Pseudaxine trachuri from different hosts and localities showed the presence of a single crown (corona) of 12 hooks. Clearly, the two circles described by Parona & Perugia [63] have never been mentioned in all subsequent descriptions of this species (Table 4). Although Lebedev [53] suggested that this could be considered a minor misinterpretation of the structure, we believe that it is important to clarify this point.
Descriptions of the genital atrium of Pseudaxine trachuri.
The number of atrial hooks in the material examined in this study and other accounts (Table 2) varies according to locality. Yamaguti suggested that it was due to intraspecific variability [76, 77]. However, the number of hooks given in this study is the same as for the specimens from the Mediterranean described by Radujkovic and Euzet [67]. It may be that specimens from seas other than the Mediterranean correspond to different species; this needs to be investigated using molecular methods. We provisionally consider that all records worldwide correspond to one and the same species.
In none of the previous description was a stylet mentioned. Although Lebedev used this term to explain the drawing by Parona & Perugia, he did not explicitly describe a stylet in his specimens.
The central stylet of the genital atrium
We describe in our specimens of P. trachuri the presence of a conical structure in the centre of the crown of spines of the genital atrium; this is the first time that such as structure has been described in this species. Llewellyn (1983) provided a detailed description of the genital atrium of Gastrocotyle trachuri; he named the hooks forming the crown “ribs” and the central stylet a “penis tube” [56]; he also proposed a hypothesis for sperm transfer in which the genital atrium would work as a sucker and the penis tube would penetrate the tegument to allow the deposition of sperm in the subtegumental tissue of the partner. What could be a stylet-like structure was also described by George (1960) in another gastrocotylid, Engraulicola forcipopenis George, 1960, as a “forceps-like spine at the end of the penis” [31]. In the diclidophorid Diclidophora merlangi (Kuhn, 1829) Krøyer, 1838, which has no vagina, MacDonald & Caley showed that sperm were injected into the tissue of the partner [57]; however, a stylet was not described in this species.
Our observations of the structure of the stylet show that it is hollow and thus could allow sperm to be injected into the tissue of the partner, and the absence of a vagina (see below) suggests that P. trachuri uses traumatic insemination, a process which is not uncommon in polyopisthocotylean monogeneans [41].
Vaginae in specimens of the genus Pseudaxine and allied genera
Parona & Perugia, in their original description of P. trachuri, which is interspersed with comparison with another species, clearly stated that there was no vagina “Infatti non travammo traccia di canale e di apertura vaginale”, i.e. “In fact we did not find a trace of a canal or of a vaginal opening” [63].
Yamaguti redescribed the species twice [76, 77]. In neither description did he mention the presence or absence of a vagina. However, in his “Systema Helminthum” he wrote for the diagnosis of Pseudaxine “vagina opening middorsally” [78].
Dillon & Hargis described Pseudaxine bivaginalis Dillon & Hargis, 1965, and emended Yamaguti’s (1963) diagnosis of the genus to include the presence of “a single vagina opening middorsally or paired vaginae opening near lateral margins at or near level of genital atrium” [23].
Unnithan described Pseudaxine kurra Unnithan, 1968, with “vaginal pore unarmed, dorsal, with few gland cells and situated in front of the ovarian loop, slightly shifted to the right side”; apparently unaware of Dillon & Hargis’s work (1965), he emended the diagnosis of the genus as “vagina single median dorsal and unarmed” [73]. Lebedev commented on the position of the vagina in Pseudaxine kurra: “Most likely a mistake was made, since the vagina cannot be in this place. In our specimen (not in very good condition) no vagina was found”; and he questioned the validity of the species [53].
Radujkovic & Euzet, for P. trachuri from off Montenegro, wrote “the vagina seems absent” [67].
Lebedev recorded Pseudaxine trachuri from Trachurus novaezelandiae (which is the type-host of Pseudaxine bivaginalis Dillon & Hargis, 1965) off Australia in 1968, and mentioned the presence of two lateral vaginal openings [49]. Since two vaginae have never been observed in P. trachuri (Table 5), we believe it likely that the gastrocotylids recorded by Lebedev on T. novaezelandiae were in fact P. bivaginalis (see the taxonomic decision made regarding this taxon below).
Vaginal opening and its position in some species attributed to Pseudaxine.
Yamaguti described Pseudaxine decapteri Yamaguti, 1968 with a “vagina on each side divided into longitudinal row of 5–13 transversely elongated cuticular pits, beginning just behind level of genital pore” [80]. He also claimed “The peculiar structure of the vagina must have been overlooked by Parona and Perugia and subsequent authors because it is recognizable in well preserved specimens alone”. Subsequently, Lebedev erected the genus Pseudaxinoides Lebedev, 1968 for monogeneans resembling Pseudaxine spp. but differing from it by the presence of multiple ventro-lateral vaginal papillae [49]. Lebedev (1977) studied descriptions and figures of Pseudaxine decapteri and assigned this species to Pseudaxinoides [51].
Taxonomic decisions
As explained above, three genera of gastrocotylids are very similar in all aspects, except for their (alleged) number of vaginae. Figure 6 shows drawings of, side by side, specimens of Pseudaxine trachuri (no vagina), Pseudaxine trachuri (“multiple vaginae”), and Allogastrocotyle bivaginalis (two vaginae). Figure 5 shows micrographs of the anterior region of similar specimens. Specimens with multiple vaginae would have been attributed to Pseudaxinoides in its traditional concept.
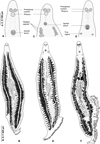 |
Figure 6 Illustrations of worms with the apparent vaginal arrangement agreeing with the diagnoses of three different genera: Pseudaxine, Pseudaxinoides, and Allogastrocotyle. (A and B) Pseudaxine trachuri, typical aspect, no vagina, MNHN HEL1290, HEL1291; (C and D) Pseudaxine trachuri, specimen with apparent “multiple vaginae”, MNHN HEL1283, HEL1292; (E and F), Allogastrocotyle bivaginalis, two real vaginae, MNHN HEL1293 (habitus redrawn from Bouguerche et al. [10]). |
In the present study, we have demonstrated, on the basis of morphology and molecules, that specimens of P. trachuri without a vagina and specimens with apparent multiple lateral vaginae are conspecific. We thus consider that all similar species described with apparent multiple vaginae are based on an erroneous interpretation of the excretory system; this makes the distinction between Pseudaxine and Pseudaxinoides void, and we propose the synonymy of these two genera. Since Pseudaxine has priority over Pseudaxinoides (1890 vs. 1968), we propose to transfer most Pseudaxinoides spp. to Pseudaxine, with the following new combinations: Pseudaxine australis (Lebedev, 1968) n. comb.; Pseudaxine bychowskyi (Lebedev, 1977) n. comb.; Pseudaxine caballeroi (Lebedev, 1977) n. comb.; Pseudaxine cariacoensis (Nasir & Fuentes-Zambrano, 1983) n. comb.; and Pseudaxine vietnamensis (Lebedev, Parukhin & Roitman, 1970) n. comb. [49–52, 54, 60, 80]. Pseudaxine decapteri Yamaguti, 1968 is kept within Pseudaxine. The status of P. kurra is pending [53]. We noticed that Pseudaxinoides pusanowi Lebedev, 1984 has two of its testes that are paraovarian [52], in contrast to species of Pseudaxine in which they are all post-ovarian; Lebedev noted later [53] that is was close to species of Allopseudaxinoides Yamaguti, 1968; a reappraisal of this species is probably necessary and thus we provisionally refrain from transferring it to Pseudaxine.
A comparison of Pseudaxine bivaginalis Dillon & Hargis, 1965 with the single species of Allogastrocotyle, A. bivaginalis Nasir & Fuentes Zambrano, 1984 [10, 60], suggests that the former species should be transferred to Allogastrocotyle, as indicated by the presence of two ventrolateral vaginal openings surrounded by muscular tissue near the genital atrium [23, 53]. Since the name Allogastrocotyle bivaginalis is already occupied, we propose here the new name Allogastrocotyle dillonhargisorum nom. nov. to avoid homonymy. The new name has been registered in ZooBank as: urn:lsid:zoobank.org:act:1ECC6CF2-4BC4-45E8-9AE7-049C00FC7735.
Comments on other genera
We noticed that “multiple lateral vaginae” have been described in other polyopisthocotylean monogeneans. Here are a few examples:
-
The description of Sibitrema poonui Yamaguti, 1966, type-species of Sibitrema Yamaguti, 1966, reads as: “vaginae symmetrical, about 0.5 mm long, divided into a series of several (8–14) areolae, situated laterally about halfway between genital pore and anterior end of vitellaria” [79].
-
The description of Allopseudaxine yaito Yamaguti, 1968 reads as “vaginae opening ventrosubmarginally at a distance of 0.9 mm from head end in the type, each divided into a longitudinal row of about ten transverse slit-like areolae with heavily cuticularized margins” [80].
-
The figure of Gastrocotyle indica Subhapradha, 1951 in Yamaguti (1963; his Figure 398) shows multiple lateral vaginae, which were commented upon by Lebedev (1986) [53, 72, 78].
Considering that in a single species, P. trachuri, we found specimens with and without what look like “multiple lateral vaginae”, we believe that the taxa mentioned above, and probably others, should be re-examined, preferably using both morphology and molecular information. More generally, we stress the importance of integrative taxonomy [21] for the taxonomy of parasites, including correct identification of hosts using both morphology and molecular analysis as in the present work and recent studies [5, 8–10, 43] and, whenever possible, deposition of hosts in curated collections [12, 36, 39, 42].
Conflict of interest
The Editor-in-Chief of Parasite is one of the authors of this manuscript. COPE (Committee on Publication Ethics, http://publicationethics.org), to which Parasite adheres, advises special treatment in these cases. In this case, the peer-review process was handled by an Invited Editor, Jérôme Depaquit.
Acknowledgments
This research was supported by Direction Générale de la Recherche Scientifique et du Développement Technologique (DGRSDT, Algiers, Algeria) and Institut de Systématique, Évolution, Biodiversité (ISYEB, MNHN, Paris, France). We thank Drs. Gerardo Pérez-Ponce de León, David Gibson, Delane Kritsky, Ken MacKenzie and Mario Santoro who kindly provided literature. We are grateful to Drs. Rodney Bray and José Amet Rivaz Hérnandez for valuable discussions concerning nomenclature. Special thanks go to Nawel Mekarma and Doria Bouabache for their help in collecting biological material.
References
- Akmirza A. 2013. Monogeneans of fish near Gökçeada, Turkey. Turkish Journal of Zoology, 37(4), 441–448. [Google Scholar]
- Alves DR, da Silva Gonçalves PH. 2012. Community ecology of the metazoan parasites of rough scad, Trachurus lathami Nichols, 1920 (Osteichthyes: Carangidae) from the coastal zone State of Rio de Janeiro, Brazil. Cadernos UniFOA, 7(20), 105–113. [Google Scholar]
- Ângelo ÂCFP. 2011. Contribution to the parasitic study of wild fish from the Atlantic coast: the case of mackerel and horse mackerel. Portugal: The School of Agriculture of Coimbra. [Google Scholar]
- Aquaro G, Salogni C, Galli P, Gibelli L, Gelmetti D. 2012. Sciadicleithrum variabilum (Dactylogyridae: Monogenea) infection in Symphysodon discus: a case report. Fish Pathology, 47(1), 23–26. [Google Scholar]
- Ayadi ZEM, Gey D, Justine J-L, Tazerouti F. 2017. A new species of Microcotyle (Monogenea: Microcotylidae) from Scorpaena notata (Teleostei: Scorpaenidae) in the Mediterranean Sea. Parasitology International, 66(2), 37–42. [CrossRef] [PubMed] [Google Scholar]
- Benhamou F, Marzoug D, Boutiba Z, Kostadinova A, Pérez-Del-Olmo A. 2017. Parasite communities in two sparid fishes from the western Mediterranean: a comparative analysis based on samples from three localities off the Algerian coast. Helminthologia, 54(1), 26–35. [Google Scholar]
- Boudaya L, Neifar L. 2016. Triloculotrema euzeti n. sp. (Monogenea, Monocotylidae) from the nasal tissues of the blackspotted smooth-hound Mustelus punctulatus (Carcharhiniformes, Triakidae) from off Tunisia. Parasite, 23, 62. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Bouguerche C, Gey D, Justine J-L, Tazerouti F. 2019. Microcotyle visa n. sp. (Monogenea: Microcotylidae), a gill parasite of Pagrus caeruleostictus (Valenciennes) (Teleostei: Sparidae) off the Algerian coast, Western Mediterranean. Systematic Parasitology, 96(2), 131–147. [CrossRef] [PubMed] [Google Scholar]
- Bouguerche C, Gey D, Justine J-L, Tazerouti F. 2019. Towards the resolution of the Microcotyle erythrini species complex: description of Microcotyle isyebi n. sp. (Monogenea, Microcotylidae) from Boops boops (Teleostei, Sparidae) off the Algerian coast. Parasitology Research, 118(5), 1417–1428. [CrossRef] [PubMed] [Google Scholar]
- Bouguerche C, Tazerouti F, Gey D, Justine J-L. 2019. Redescription and molecular characterisation of Allogastrocotyle bivaginalis Nasir & Fuentes Zambrano, 1983 (Monogenea: Gastrocotylidae) from Trachurus picturatus (Bowdich, 1825) (Carangidae) off the Algerian coast, Mediterranean Sea. Systematic Parasitology, 96, 681–694. [CrossRef] [PubMed] [Google Scholar]
- Bowles J, Blair D, McManus DP. 1995. A molecular phylogeny of the human schistosomes. Molecular Phylogenetics and Evolution, 4(2), 103–109. [CrossRef] [PubMed] [Google Scholar]
- Bradley RD, Bradley LC, Honeycutt RL, MacDonald KA, Amarilla-Stevens HN, Stevens RD. 2020. Nomenclatural, curatorial, and archival best practices for symbiotypes and other type materials in natural history collections. Occasional Papers of the Museum of Texas Tech University, 366, 1–20. [Google Scholar]
- Braicovich PE, Luque JL, Timi JT. 2012. Geographical patterns of parasite infracommunities in the rough scad, Trachurus lathami Nichols, in the Southwestern Atlantic Ocean. Journal of Parasitology, 98(4), 768–777. [CrossRef] [Google Scholar]
- Campbell N. 2008. Population studies of horse mackerel (Trachurus trachurus (L.)) and herring (Clupea harengus L.) using parasites as biological tags, Thesis. Aberdeen, Scotland: University of Aberdeen. p. 1–146. [Google Scholar]
- Chaabane A, Neifar L, Justine J-L. 2015. Pseudorhabdosynochus regius n. sp. (Monogenea, Diplectanidae) from the mottled grouper Mycteroperca rubra (Teleostei) in the Mediterranean Sea and Eastern Atlantic. Parasite, 22, 9. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Chaabane A, Justine J-L, Gey D, Bakenhaster MD, Neifar L. 2016. Pseudorhabdosynochus sulamericanus (Monogenea, Diplectanidae), a parasite of deep-sea groupers (Serranidae) occurs transatlantically on three congeneric hosts (Hyporthodus spp.), one from the Mediterranean Sea and two from the western Atlantic. PeerJ, 4, e2233. [Google Scholar]
- Chaabane A, Neifar L, Gey D, Justine J-L. 2016. Species of Pseudorhabdosynochus (Monogenea, Diplectanidae) from groupers (Mycteroperca spp., Epinephelidae) in the Mediterranean and Eastern Atlantic Ocean, with special reference to the “beverleyburtonae group” and description of two new species. PLoS One, 11(8), e0159886. [Google Scholar]
- Chaabane A, Neifar L, Justine J-L. 2017. Diplectanids from Mycteroperca spp. (Epinephelidae) in the Mediterranean Sea: Redescriptions of six species from material collected off Tunisia and Libya, proposal for the “Pseudorhabdosynochus riouxi group”, and a taxonomic key. PLoS One, 12(2), e0171392. [Google Scholar]
- Çinar ME. 2014. Checklist of the phyla Platyhelminthes, Xenacoelomorpha, Nematoda, Acanthocephala, Myxozoa, Tardigrada, Cephalorhyncha, Nemertea, Echiura, Brachiopoda, Phoronida, Chaetognatha, and Chordata (Tunicata, Cephalochordata, and Hemichordata) from the coasts of Turkey. Turkish Journal of Zoology, 38(6), 698–722. [CrossRef] [Google Scholar]
- Costa G, Melo-Moreira E, de Carvalho MP. 2012. Helminth parasites of the oceanic horse mackerel Trachurus picturatus Bowdich 1825 (Pisces: Carangidae) from Madeira Island, Atlantic Ocean, Portugal. Journal of Helminthology, 86(3), 368–372. [CrossRef] [PubMed] [Google Scholar]
- Dayrat B. 2005. Towards integrative taxonomy. Biological Journal of the Linnean Society, 85(3), 407–415. [Google Scholar]
- Derouiche I, Neifar L, Gey D, Justine J-L, Tazerouti F. 2019. Holocephalocotyle monstrosae n. gen., n. sp. (Monogenea, Monocotylidae) from the olfactory rosette of the rabbit fish, Chimaera monstrosa (Holocephali, Chimaeridae) in deep water off Algeria. Parasite, 26, 59. [CrossRef] [EDP Sciences] [PubMed] [Google Scholar]
- Dillon WA, Hargis WJ. 1965. Monogenetic trematodes from the Southern Pacific Ocean. 2. Polyopisthocotyleids from New Zealand fishes: the families Discocotylidae, Microcotylidae, Axinidae and Gastrocotylidae. Biology of the Antarctic Seas II, Antarctic Research Series, 5, 251–280. [Google Scholar]
- Feki M, Châari M, Neifar L, Boudaya L. 2016. Spatial variability of helminth parasites to recognize the discrimination of juvenile and young adult areas of horse mackerel, Trachurus trachurus (Linnaeus, 1758) off the coast of Tunisia. Fisheries Research, 183, 318–325. [Google Scholar]
- Fernandez-Jover D, Faliex E, Sanchez-Jerez P, Sasal P, Bayle-Sempere JT. 2010. Coastal fish farming does not affect the total parasite communities of wild fish in SW Mediterranean. Aquaculture, 300(1–4), 10–16. [Google Scholar]
- Fischer W, Bauchot M-L, Schneider M. 1987. Fiches FAO d’identification des espèces pour les besoins de la pêche. (Révision 1). Méditerranée et mer Noire. Zone de pêche 37, in Volume II. Vertébrés. Publication préparée par la FAO, résultat d’un accord entre la FAO et la Commission des Communautés Européennes (Projet GCP/INT/422/EEC) financée conjointement par ces deux organisations. FAO: Rome, Vol. 2. p. 761–1530. [Google Scholar]
- Gaevskaya AV, Kovaleva AA. 1979. Monogenean parasites of Atlantic horsemackerels of the genus Trachurus. Zoologicheskii Zhurnal, 58(8), 1110–1116. (in Russian). [Google Scholar]
- Gaevskaya AV, Kovaleva AA. 1980. Eco-geographical features of the parasitofauna of the common Atlantic Horse Mackerel. Kaliningrad, AtlantNIRO: Studies of the biological resources of the Atlantic Ocean. p. 18–24. (in Russian). [Google Scholar]
- Gaevskaya AV, Kovaleva AA. 1985. The parasite fauna of the oceanic horse mackerel Trachurus picturatus picturatus and eco-geographical characteristics of its formation. Ekologiya Morya, 20, 80–84. (in Russian). [Google Scholar]
- Gallegos-Navarro Y, Violante-González J, Monks S, García-Ibáñez S, Rojas-Herrera AA, Rosas-Acevedo JL. 2019. Metazoan parasite communities of Selar crumenophthalmus and Decapterus muroadsi (Pisces: Carangidae) from Mexican Pacific coasts. New Zealand Journal of Marine and Freshwater Research, 53(3), 377–396. [CrossRef] [Google Scholar]
- George K. 1960. On a new gastrocotylid trematode, Engraulicola forcipopenis gen. et sp. nov. on white-bait, from southern India. Journal of the Marine Biological Association of India, 2(2), 208–215. [Google Scholar]
- Hamdi I, Hermida M, Tlig SZ, Benmansour B. 2019. Distribution of two monogenean [sic] of Trachurus picturatus from Tunisia, Mediterranean Sea and Madeira Island, Atlantic Ocean. Rapport du Congrès de la Commission Internationale pour l’Exploration Scientifique de la Mer Méditerranée. p. 23. [Google Scholar]
- Hansen H, Bakke TA, Bachmann L. 2007. DNA taxonomy and barcoding of monogenean parasites: lessons from Gyrodactylus. Trends in Parasitology, 23(8), 363–367. [CrossRef] [PubMed] [Google Scholar]
- Hasegawa M, Kishino H, Yano T-A. 1985. Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. Journal of Molecular Evolution, 22(2), 160–174. [CrossRef] [PubMed] [Google Scholar]
- Hermida M, Pereira A, Correia A, Cruz C, Saraiva A. 2016. Metazoan parasites of blue jack mackerel Trachurus picturatus (Perciformes: Carangidae) from Portuguese mainland waters. Journal of Helminthology, 90(4), 410–416. [CrossRef] [PubMed] [Google Scholar]
- Hoberg EP, Pilitt PA, Galbreath KE. 2009. Why museums matter: a tale of pinworms (Oxyuroidea: Heteroxynematidae) among pikas (Ochotona princeps and O. collaris) in the American West. Journal of Parasitology, 95, 490–501. [CrossRef] [Google Scholar]
- Ichalal K, Chikoune A, Ramdane Z, Iguer-Ouada M, Kacher M. 2017. The parasite fauna of Trachurus trachurus (Linnaeus, 1758) (Teleostei: Carangidae) from the Eastern coast of Algeria. Bulletin de la Société Zoologique de France, 142(1), 29–45. [Google Scholar]
- Ider D, Ramdane Z, Trilles J-P, Amara R. 2018. Metazoan parasites of Boops boops (Linnaeus, 1758) from the Algerian coast. Cahiers de Biologie Marine, 59(3), 225–233. [Google Scholar]
- Jorissen MWP, Huyse T, Pariselle A, Wamuini Lunkayilakio S, Muterezi Bukinga F, Chocha Manda A, Kapepula Kasembele G, Vreven EJ, Snoeks J, Decru E, Artois T, Vanhove MPM. 2020. Historical museum collections help detect parasite species jumps after tilapia introductions in the Congo Basin. Biological Invasions. DOI: 10.1007/s10530-020-02288-4. [PubMed] [Google Scholar]
- Jovelin R, Justine J-L. 2001. Phylogenetic relationships within the polyopisthocotylean monogeneans (Platyhelminthes) inferred from partial 28S rDNA sequences. International Journal for Parasitology, 31, 393–401. [CrossRef] [PubMed] [Google Scholar]
- Justine J-L. 1985. Étude ultrastructurale comparée de la spermiogenèse des Digènes et des Monogènes (Plathelminthes). Université des Sciences et Techniques du Languedoc. Relations entre la morphologie du spermatozoïde, la biologie de la fécondation et la phylogénie. Thèse d’Etat. Montpellier, France: Université des Sciences et Techniques du Languedoc (Montpellier II). [Google Scholar]
- Justine J-L, Briand MJ. 2010. Three new species, Lamellodiscus tubulicornis n. sp., L. magnicornis n. sp. and L. parvicornis n. sp. (Monogenea: Diplectanidae) from Gymnocranius spp. (Lethrinidae: Monotaxinae) off New Caledonia, with proposal of the new morphological group ‘tubulicornis’ within Lamellodiscus Johnston & Tiegs, 1922. Systematic Parasitology, 75, 159–179. [CrossRef] [PubMed] [Google Scholar]
- Justine J-L, Rahmouni C, Gey D, Schoelinck C, Hoberg EP. 2013. The monogenean which lost its clamps. PLoS One, 8(11), e79155. [Google Scholar]
- Kheddam H, Justine J-L, Tazerouti F. 2016. Hexabothriid monogeneans from the gills of deep-sea sharks off Algeria, with the description of Squalonchocotyle euzeti n. sp. (Hexabothriidae) from the kitefin shark Dalatias licha (Euselachii, Dalatiidae). Helminthologia, 53(4), 354–362. [Google Scholar]
- Kimura M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. Journal of Molecular Evolution, 16(2), 111–120. [CrossRef] [PubMed] [Google Scholar]
- Korotaeva VD. 1975. Contribution to the fauna of helminths and parasitic crustaceans of marine commercial fishes of the Australian and New Zealand waters. Trudy Biologo-Pochvennogo Instituta, Novaya Seriya, 26(129), 46–60. [Google Scholar]
- Ktari M-H. 1971. Recherches sur la reproduction et le développement de quelques Monogènes (Polyopisthocotylea) parasites de poissons marins. Thèse d’État. Montpellier, France: University of Montpellier. [Google Scholar]
- Kumar S, Stecher G, Tamura K. 2016. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33, 1870–1874. [CrossRef] [PubMed] [Google Scholar]
- Lebedev BI. 1968. Monogeneans of fish of the New Zealand-Australian shelf in the South China Sea (Monogenoidea: Gastrocotylidae, Gastrocotylinae), in Helminths of animals of the Pacific Ocean, Skrjabin KI, Mamaev YL, Editors. Nauka: Moscow. [Google Scholar]
- Lebedev BI. 1970. Helminths of epipelagic fishes of the South China Sea, in Helminths of animals of Southeast Asia, Oshmarin PG, Mamaev YL, Lebedev BI, Editors. Nauka: Moscow. [Google Scholar]
- Lebedev BI. 1977. Two new species of monogeneans from fishes of Indo-Pacific waters wilh notes concerning the genera Pseudaxine and Pseudaxinoides. Excerta parasitologica en memoria del Dr.Eduardo Caballero y Cabellero, 4, 69–78. [Google Scholar]
- Lebedev BI. 1984. New taxa of gastrocotylid monogeneans, in Parasites of animals and plants, Mamaev YL, Dvoryadkin VA, Eroshenko AS, Editors. Far-Eastern Scientific Center of the USSR Academy of Sciences: Vladivostok. [Google Scholar]
- Lebedev BI. 1986. Monogenea: suborder Gastrocotylinea. Leningrad: Nauka. p. 200. [Google Scholar]
- Lebedev BI, Parukhin AM, Roitman VA. 1970. Monogenetic trematodes, Oligonchoinea (Monogenoidea), parasites of horse mackerel fishes of North Vietnam Gulf. Biologiya Morya, 20, 167–187. (in Russian). [Google Scholar]
- Littlewood DTJ, Rohde K, Clough KA. 1997. Parasite speciation within or between host species? – phylogenetic evidence from site-specific polystome monogeneans. International Journal for Parasitology, 27, 1289–1297. [CrossRef] [PubMed] [Google Scholar]
- Llewellyn J. 1983. Sperm transfer in the monogenean gill parasite Gastrocotyle trachuri. Proceedings of the Royal Society of London, B Biological Sciences, 219, 439–446. [CrossRef] [Google Scholar]
- MacDonald S, Caley J. 1975. Sexual reproduction in the monogenean Diclidophora merlangi: tissue penetration by sperms. Zeitschrift für Parasitenkunde, 45, 323–334. [CrossRef] [Google Scholar]
- Marzoug D, Boutiba Z, Kostadinova A, Pérez-del-Olmo A. 2012. Effects of fishing on parasitism in a sparid fish: Contrasts between two areas of the Western Mediterranean. Parasitology International, 61(3), 414–420. [CrossRef] [PubMed] [Google Scholar]
- Mollaret I, Jamieson BGM, Justine J-L. 2000. Phylogeny of the Monopisthocotylea and Polyopisthocotylea (Platyhelminthes) inferred from 28S rDNA sequences. International Journal for Parasitology, 30, 171–185. [CrossRef] [PubMed] [Google Scholar]
- Nasir P, Fuentes Zambrano J. 1983. Algunos tremátodos monogenéticos venezolanos. Rivista di Parassitologia, 44(3), 335–380. [Google Scholar]
- Öktener A. 2014. An updated checklist of parasitic helminths of marine fish from Turkey. Transylvanian Review of Systematical and Ecological Research, 16(2), 55–96. [CrossRef] [Google Scholar]
- Orecchia P, Paggi L. 1978. Aspetti di sistematica e di ecologia degli elminti parassiti di pesci marini studiati presso l’Istituto di Parassitologia dell’Universitá di Roma. Parassitologia, 20(1, 2, 3), 73. [PubMed] [Google Scholar]
- Parona C, Perugia A. 1890. Intorno ad alcune Polystomeae e considerazioni sulla sistematica di questa famiglia. Atti della Società Ligustica di Scienze Naturali e Geografiche, 1(3), 225–242 + Plates. [Google Scholar]
- Parukhin AM. 1976. Parasitic worms of food fishes of the southern seas. Kiev: Naukova Dumka. p. 183. (in Russian). [Google Scholar]
- Pérez-del Olmo A, Fernandez M, Gibson DI, Raga JA, Kostadinova A. 2007. Descriptions of some unusual digeneans from Boops boops L. (Sparidae) and a complete checklist of its metazoan parasites. Systematic Parasitology, 66, 137–157. [CrossRef] [PubMed] [Google Scholar]
- Price EW. 1943. North American monogenetic trematodes: VI. The family Diclidophoridae (Diclidophoroidea). Journal of the Washington Academy of Sciences, 33(2), 44–54. [Google Scholar]
- Radujkovic BM, Euzet L. 1989. Parasites des poissons marins du Monténégro: Monogènes. In: Radujkovic, BM & Raibaut, A (Eds.), Faune des parasites de poissons marins des côtes du Monténégro (Adriatique Sud). Acta Adriatica, 30(1/2), 51–135. [Google Scholar]
- Ratnasingham S, Hebert PDN. 2007. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Molecular Ecology Notes, 7(3), 355–364. [CrossRef] [PubMed] [Google Scholar]
- Rezaiguia W. 2008. Les monogènes parasites : inventaire et distribution chez 9 espèces de poissons téléostéens pêchés dans le golfe d’Annaba. Thesis (Magistère). Annaba, Algeria: Université Badji-Mokhtar. [Google Scholar]
- Sproston N. 1946. A synopsis of the monogenetic trematodes. Transactions of the Zoological Society of London, 25, 185–600. [Google Scholar]
- Strona G, Stefani F, Galli P. 2010. Monogenoidean parasites of Italian marine fish: an updated checklist. Italian Journal of Zoology, 77(4), 419–437. [CrossRef] [Google Scholar]
- Subhapradha CK. 1951. Vallisiopsis contorta n. g. and n. sp. and Gastrocotyle indica n. sp., monogenetic trematodes from marine fishes of the Madras Coast. Parasitology, 41(3–4), 162–165. [PubMed] [Google Scholar]
- Unnithan RV. 1968. On six species of monogenetic trematodes, parasitic on the gills of marine fishes from the Indian seas. Treubia, 27(2/3), 141–164. [Google Scholar]
- Vanhove MP, Tessens B, Schoelinck C, Jondelius U, Littlewood DT, Artois T, Huyse T. 2013. Problematic barcoding in flatworms: a case-study on monogeneans and rhabdocoels (Platyhelminthes). Zookeys, 365, 355–379. [Google Scholar]
- Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PD. 2005. DNA barcoding Australia’s fish species. Philosophical Transactions of the Royal Society of London B Biological Sciences, 360(1462), 1847–1857. [Google Scholar]
- Yamaguti S. 1938. Studies on the helminth fauna of Japan. Part 24. Trematodes of fishes, V. Japanese Journal of Zoology, 8, 15–74. [Google Scholar]
- Yamaguti S. 1942. Studies on the helminth fauna of Japan. Part 37. Trematodes of fishes, VIII. Japanese Journal of Medical Sciences. VI. Bacteriology and Parasitology, 2(3), 105–129. [Google Scholar]
- Yamaguti S. 1963. Systema Helminthum Volume IV Monogenea and Aspidocotylea. New York: John Wiley & Sons. [Google Scholar]
- Yamaguti S. 1966. New monogenetic trematodes from Hawaiian fishes, II. Pacific Science, 20, 419–434. [Google Scholar]
- Yamaguti S. 1968. Monogenetic trematodes of Hawaiian fishes. Honolulu: University of Hawaii Press. p. 287. [Google Scholar]
- Zhang J, Wu X, Xie M, Li A. 2012. The complete mitochondrial genome of Pseudochauhanea macrorchis (Monogenea: Chauhaneidae) revealed a highly repetitive region and a gene rearrangement hot spot in Polyopisthocotylea. Molecular Biology Reports, 39(8), 8115–8125. [CrossRef] [PubMed] [Google Scholar]
Cite this article as: Bouguerche C, Tazerouti F, Gey D & Justine J–L. 2020. No vagina, one vagina, or multiple vaginae? An integrative study of Pseudaxine trachuri (Monogenea, Gastrocotylidae) leads to a better understanding of the systematics of Pseudaxine and related genera. Parasite 27, 50.
All Tables
Fish, monogeneans and their COI sequences. To insure full traceability and ensure accurate host-parasite relationships, one or several monogeneans were collected from one fish and all individuals were sequenced. All monogeneans in this Table were identified as Pseudaxine trachuri.
All Figures
 |
Figure 1 Molecular phylogenetic analysis of monogenean COI sequences using the Maximum Likelihood method. Bootstraps percentages (1000 replicates) are indicated next to the branches when higher than 70. There were a total of 389 positions in the final dataset. The Neighbour-Joining tree had a similar topology and is not represented. All specimens of Pseudaxine trachuri, whether with no vagina or with “multiple vaginae”, and whether from the type-host Trachurus trachurus or from Boops boops, were grouped into a single monophylum and showed little variation (<2%). |
| In the text | |
 |
Figure 2 Pseudaxine trachuri Parona & Perugia, 1890. (A) Entire worm, typical specimen without a vagina, MNHN HEL1278; (B) clamp, ventral view; (C) clamp, dorsal view, MNHN HEL1278; (D–F) anchors from terminal lappet, MNHN HEL1279, HEL1280; (G and H), terminal lappet, MNHN HEL1280, HEL1281; (I) egg, MNHN HEL1282. (J) anterior extremity of a specimen from Boops boops, showing apparent “multiple lateral vaginae” (MNHN HEL1283). |
| In the text | |
 |
Figure 3 Pseudaxine trachuri, genital atrium shown laterally and in different optical sections and the variable position of the stylet. (A) Genital atrium, lateral view, MNHN HEL1284; (B–E), optical sections of a genital atrium in perfect apical view; the level of these sections are shown on the lateral view, MNHN HEL1285. (F) Lateral view of the atrium showing only the sclerotised parts, MNHN 306HG, Box 43, slide 51; (G–I) various views at different orientations, MNHN HEL1286, MNHN HEL1287, MNHN HEL1288. (J) One spine, MNHN HEL1287. |
| In the text | |
 |
Figure 4 Clamp, with detailed legends, MNHN HEL1289. (A) Ventral jaw. (B) Dorsal jaw. (C) Whole clamp. |
| In the text | |
 |
Figure 5 Micrographs of specimens showing the region of the genital atrium and vaginae (arrows). Left, Pseudaxine trachuri, no vagina visible; Middle, Pseudaxine trachuri, alleged multiple vaginae, two different focuses on the same specimen; Right, Allogastrocotyle bivaginalis, two real vaginae. Unscaled, same scale for all micrographs. |
| In the text | |
 |
Figure 6 Illustrations of worms with the apparent vaginal arrangement agreeing with the diagnoses of three different genera: Pseudaxine, Pseudaxinoides, and Allogastrocotyle. (A and B) Pseudaxine trachuri, typical aspect, no vagina, MNHN HEL1290, HEL1291; (C and D) Pseudaxine trachuri, specimen with apparent “multiple vaginae”, MNHN HEL1283, HEL1292; (E and F), Allogastrocotyle bivaginalis, two real vaginae, MNHN HEL1293 (habitus redrawn from Bouguerche et al. [10]). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


