| Issue |
Parasite
Volume 20, 2013
|
|
|---|---|---|
| Article Number | 32 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/parasite/2013031 | |
| Published online | 13 September 2013 | |
Research Article
Haemoproteus syrnii in Strix aluco from France: morphology, stages of sporogony in a hippoboscid fly, molecular characterization and discussion on the identification of Haemoproteus species
Haemoproteus syrnii chez Strix aluco en France : morphologie, stades de la sporogonie chez un diptère Hippoboscidae, caractérisation moléculaire et discussion sur l’identification des espèces d’Haemoproteus
1
UMR 7245 MCAM MNHN CNRS, Muséum National d’Histoire Naturelle, 61 rue Buffon, CP 52, 75231 Paris Cedex 05, France
2
Hôpital de la faune sauvage des Garrigues et Cévennes – Clinique vétérinaire, 19 avenue du Vigan, 34190 Ganges, France
3
Malaria Reference Centre – National Public Health Laboratory, Ministry of Health, 9 Hospital Drive, Block C, #04-01, Sing Health Research Facilities, 169612 Singapore
4
Inserm, UMR-S 945, 91 Bd de l’Hôpital, 75013 Paris, France
5
Université Pierre et Marie Curie-Paris 6, Faculté de Médecine Pitié-Salpêtrière, CHU Pitié-Salpêtrière, 91 Bd de l’Hôpital, 75013 Paris, France
* Corresponding author: e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
27
June
2013
Accepted:
30
August
2013
Abstract
In France, Haemoproteus syrnii is frequently found in the Tawny Owl, Strix aluco. Additional and complementary features of this species, and in particular the characteristics of volutin, are presented. The authors consider the volutin granules as constant in a given species, and discuss their taxonomic value. These cytoplasmic inclusions appear early during the first stages of development of the gametocytes as an initial granule which multiplies as the parasite develops. They were reported in some species of Haemoproteus but are seldom considered as a specific character and described with precision. Sporogony from ookinete to apparently mature sporozoites appears to take place in a pupiparous hippoboscid (Ornithomyia sp.). One specimen was crushed between two slides and stained with Giemsa. Gametocytes of H. syrnii, many ookinetes, an immature oocyst and mature sporozoites were observed spread all over the smear. This would allow classifying this species in the Haemoproteus subgenus. We provide associated molecular data derived from the cyt b and cox 1 gene from this parasite. We discuss the problems of multiple infections and the difficulties in identifying Haemoproteus species and in deriving conclusions from sequences deposited in databases.
Résumé
En France, Haemoproteus syrnii est un parasite fréquent des chouettes hulottes, Strix aluco. Le parasite est redécrit et les auteurs insistent particulièrement sur les caractères de la volutine, qu’ils considèrent comme constants dans une espèce donnée et dont ils discutent la valeur taxonomique. Ces inclusions cytoplasmiques apparaissent dès les premiers stades du développement du gamétocyte sous forme d’un granule initial qui se multiplie au cours de l’évolution du parasite. Elles ont été signalées chez certaines espèces d’Haemoproteus mais sont rarement considérées comme un caractère spécifique et décrites avec précision. La sporogonie semble se développer du stade ookinète au stade sporozoïte apparemment mûr chez une Hippoboscidae pupipare (Ornithomyia sp.). Chez un spécimen écrasé entre deux lames et coloré au Giemsa ont été observés des gamétocytes d’H. syrnii, de nombreux ookinètes, un oocyste immature et des sporozoïtes matures dispersés sur la lame. Ceci permettrait de classer l’espèce dans le sous-genre Haemoproteus. Des données moléculaires concernant le parasite sont présentées (cyt b et cox 1). Le polyparasitisme et les difficultés à identifier les espèces d’Haemoproteus ainsi qu’à interpréter les séquences déposées dans les banques de données sont discutées.
Key words: Haemoproteus syrnii / Strix aluco / Volutin granules / Pupiparous fly / Haemoproteus subgenera / Molecular identification
© G. Karadjian et al., published by EDP Sciences, 2013
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Haemoproteus syrnii (Mayer, 1910) [22] was since its original description recorded in 30 species of Strigiformes [31]. Bishop and Bennett [2], in a review of parasites of Strigiformes, redescribed H. syrnii in Strix varia from Oklahoma (USA) and synonymized many other species based on published studies and from blood smears deposited at the International Reference Center for Avian Haematozoa (IRCAH). Martinsen et al. [18] using blood samples provided by Prof. I. Paperna who collected them from Strix seloputo and Ninox scutulata sampled in Singapore, in which H. syrnii was identified, provided gene sequences from these parasites. The corresponding material from Ilan Paperna’s collection was later deposited at the Muséum National d’Histoire Naturelle in Paris (MNHN), where after study we were able to redescribe the species and distinguish it from H. syrnii (Karadjian et al., unpublished).
The genus Haemoproteus has been subdivided by Bennett et al. (1965) in two subgenera [1]: Haemoproteus and Parahaemoproteus, mainly on the basis of different vectors: hippoboscid flies for the first one and Culicoides for the second, and on morphological differences of the sporogonic stages.
In his book on avian Haemosporidia, Valkiūnas [31] considered that species of the subgenus Haemoproteus must be restricted to parasites of Columbiformes. Later on, he realized, as did other authors [15], that the breadth of hosts of this subgenus could be much larger than previously thought. Levin et al. [15] redescribed Haemoproteus iwa Work & Rameyer (1996) [36] in Fregata minor from the Galapagos, using morphological and molecular tools (cyt b). They provided evidence that H. iwa is phylogenetically closely related to Haemoproteus columbae (Kruse 1890) [14], and was very probably transmitted by hippoboscid flies; consequently, this parasite should be classified in the Haemoproteus subgenus.
In this article we present morphological observations that complement the original description of H. syrnii in Strix aluco and propose to consider the volutin grains as important morphological and biological characters for Haemoproteus spp. gametocytes. We also provide associated sequence data of cytochrome b (cyt b) and cytochrome c oxidase I (cox 1) from this parasite and we discuss the difficulties in attempting to associate a particular sequence to a taxon, and to establish meaningful comparisons with data deposited in GenBank. Finally, we discuss the parasite’s taxonomic status and the host range of subgenera Haemoproteus and Parahaemoproteus.
Materials and methods
Materials
The biological material from S. aluco, included in this study, originated from Émile Brumpt’s collection and two location sites in France (see Table 1):
-
Émile Brumpt’s collection: Seven blood smears collected in 1934 at Richelieu (Indre et Loire).
-
Hôpital de la Faune Sauvage des Garrigues et Cévennes (Hérault): 39 blood smears from 7 adults and 32 juvenile S. aluco. For molecular characterization, two blood samples (one EDTA tube and one blood spot) were withdrawn from the brachial vein of birds (163BF and 154ZI) that harboured single infections with H. syrnii. Samples were collected from June 2011 to January 2013.
-
Centre Régional de Soins pour la Faune Sauvage, Pas-de-Calais: 6 blood smears from 5 adults and 1 juvenile bird. Samples were collected from October to December 2011.
Bird specimens (Strix aluco) studied, details about sampling and collection, and level of infection with Haemoproteus syrnii (−: uninfected; + to +++, level of infection).
Slides
All the blood smears on which the redescription was based were deposited in the collection of the Muséum National d’Histoire Naturelle, Paris, France. The record numbers and the dates at which the smears were made can be found in Table 1. All the smears were fixed by absolute methanol prior to Giemsa staining (10% in phosphate-buffered solution pH = 7.4) during one hour. They were then covered by a coverslip that was mounted with Eukitt® resin before examination under oil immersion. Pigment granules were examined and photographed in unstained methanol fixed blood smears.
Slides from Strix seloputo included in Paperna’s collection are deposited in the MNHN under number 176BF, PIX58-60.
Molecular methods
Blood samples were extracted using DNA Qiagen Micro Kit following the manufacturer’s instruction handbook for whole blood and blood spot extraction.
Partial mitochondrial gene cyt b (750 bp) and cox 1 (1293 bp) amplifications of the samples 163BF and 154 ZI were done using specific primers and protocols from Duval et al. (2007) [5]. The PCR products were sequenced using PLAS3 and PLAS4 primers by Cogenics. The cyt b partial gene sequences obtained included the cyt b gene region proposed as a standard for DNA bar-coding system for avian Haemoproteus species [9].
Avian Haemosporidia cyt b sequences were retrieved from GenBank (http://www.ncbi.nlm.nih.gov) for phylogenetic reconstruction. Molecular phylogeny was performed with 346 bp of mitochondrial cyt b gene by using Maximum-Likelihood methods with GTR model and nodal robustness evaluated by non-parametric bootstrapping (100 replicates) [8] (Figure 36). The phylogenetic tree was rooted using avian Leucocytozoon parasites.
Sequences of cyt b and cox 1 were deposited in GenBank as KF279523 and KF279522, respectively. The genetic distance between the Martinsen et al. (2006) [18] sequences and the newly generated sequences from S. aluco was estimated by p-distance method based on cyt b and cox 1.
Results
In all the materials we have examined, we could only observe gametocytes identifiable as H. syrnii, though a Plasmodium, a Trypanosoma and a few gametocytes from a Leucocytozoon, were also observed in some specimens.
Morphological redescription of Haemoproteus syrnii in Strix aluco
The rings and very young gametocytes lie in an apical or subapical position and are rarely in contact with the red blood cell (RBC) nucleus or its membrane.
The parasite is initially rounded or oval, and it comprises a cap-like nucleus, a large white vacuole and a first large granule of volutin deep garnet in colour with sometimes a less dark centre. Thereafter, the gametocyte elongates and becomes situated along the RBC nucleus. It has one end that is more peaked, even quite sharp, than the other. That first grain of volutin is found at this sharp end, or sometimes close to the nucleus. It is sometimes annular, a very characteristic feature (Figures 1–4, 17–24) that was previously observed by Mayer, in 1911, particularly in Figures 1 and 4 [21]. We have also observed cases where one or two granules appear to detach from the initial granule (Figure 5). At the next developmental stage, the gametocyte elongates further and the extremities become blunted (Figures 5–6, 22–25), the nucleus is median and the large vacuole from the younger stages has disappeared and small vacuoles are scattered in the cytoplasm (Figures 6, 23–25). Thereafter, it becomes loaded with volutin granules that progressively occupy the parasite’s ends (Figures 7, 26), and are particularly predominant in microgametocytes (Figures 11–14, 27, 28). In macrogametocytes, the nucleus is also median but the granules are distributed throughout the cytoplasm where many small vacuoles can be found (Figures 8–10, 29, 30). Finally, the parasite elongates yet further and its extremities curve around the RBC nucleus, though without encompassing it completely (Figures 9–13, 28–30). Generally one notes a thin space between the nucleus and the gametocyte. The opposing lateral border reaches the RBC membrane, but the curving ends leave a free space. Pigment granules are obscured by the volutin in stained slides and are described in unstained material (Figure 31): they are dispersed, black, elongated and of medium size (0.5–1.0 μm).
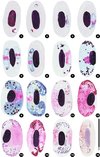 |
Figures 1–16. Drawings of erythrocytic stages of Haemoproteus syrnii in the blood of Strix aluco. 1–4: young trophozoites with an initial volutin granule; 5: two young gametocytes with 1 or 2 volutin granules attached to the initial granule; 6–7 young gametocytes; 8–10 mature macrogametocytes; 11, 13: mature microgametocytes; 12: old microgametocyte; 14: two microgametocytes within the same RBC; 15: a microgametocyte spread beneath the RBC nucleus; 16: microgametocyte with faded staining and where the volutin granules are still visible. |
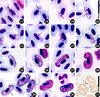 |
Figures 17–31. Microphotographs of Haemoproteus syrnii gametocytes in the blood of Strix aluco. 17–24: young gametocytes with the initial volutin granule (arrow); 25–26: immature gametocytes; 27–28: microgametocytes; 29: two macrogametocytes and a microgametocyte (arrow); 30: macrogametocyte and microgametocyte (arrow). Giemsa staining; 31: unstained smear: pigment of gametocyte. |
Volutin granules are distinctly finer in mature microgametocytes as compared to those in macrogametocytes. For both sexes the granules become smaller and more numerous as the gametocytes age. This ageing is accompanied by hyper-chromophilia; the chromatin is less granular and small very white vacuoles are seen throughout.
The RBC nucleus is central in young forms, whereas it appears more or less displaced in older or mature forms depending on the way the gametocyte is positioned on the slide at the time the blood smear is drawn; in these mature or aged gametocytes the nucleus is generally on a plane above that of the parasites and it might be rounded and condensed.
Notes: Mayer noted the presence of the first volutin granule in the very young stages [22] though he interpreted it as a second nucleus (kern). Our observations, and in particular the appearance of other attached granules, lead us to consider it as the initial volutin granule.
Sporogony in a hippoboscid fly (Ornithomyia sp.)
In the smear obtained by squashing the hippoboscid fly we observed numerous elongated or rounded gametocytes (Figures 32, 33), and ookinetes (Figure 35). The shape and appearance of the gametocytes, including the presence of volutin, correspond to those of H. syrnii. Numerous sporozoites were dotted on the smear (Figures 32, 33). We have measured those that appeared to us to have reached maturity (Figure 34) (an average of 12 μm). They have a central nucleus formed by three grains, and a sharp extremity. At one place on the slide we noted a collection of smaller immature sporozoites radiating from centres (Figure 33), which probably represents a nearly mature oocyst that was burst by the squashing.
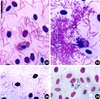 |
Figures 32–35. Microphotographs of developmental stages of Haemoproteus syrnii in the hippoboscid fly. 32: sporozoites issued from a burst oocyst and a gametocyte; 33: burst oocyst with sporozoites still attached to the cytomeres, and a gametocyte; 34: two mature sporozoites; 35: ookinetes. Giemsa staining. |
 |
Figure 36. Maximum-likelihood phylogeny of mitochondrial cytochrome b lineages (346 bp) of avian Plasmodium spp. (8 sequences) and Haemoproteus spp. (21 sequences). Three sequences of Leucocytozoon spp. are used as outgroup. Bootstrap values > 70% are indicated near the nodes. GenBank accessions numbers and hosts (in parentheses) are indicated after the name of the parasite. |
Molecular characterizations and phylogenetic reconstruction
750 bp of cyt b and 1293 bp of cox 1 sequences obtained were associated with H. syrnii morphospecies identified and described in this paper. Alignment of these partial gene sequences with cyt b and cox 1 associated with H. syrnii published by Martinsen et al. [18] showed a molecular divergence of 3% for cyt b and 3.3% for cox 1.
Phylogenetically, H. syrnii is included in the group/clade that contains parasites from both the subgenera Parahaemoproteus and Haemoproteus.
Discussion
Prevalence
The great majority of the infected Tawny Owls were adults 6/10 (60%) while only 1/33 (3%) was a juvenile. However, most of the juveniles (31/33) had been examined in June. Although many had heavy infections with Leucocytozoon only one juvenile examined (07/05/2012) harboured Haemoproteus (Table 1).
RBC nucleus
The morphology of the RBC and the position of the parasite with respect to the RBC nucleus are important characters for each species and they are generally noted in their descriptions. Many parasites surround the RBC nucleus completely or partially. In H. syrnii, the extremities of the parasite curve around the extremities of the RBC nucleus, though without encompassing it completely. In thin red blood films the parasite has a tendency to position itself on an inferior plane to that of the RBC nucleus, and as it reaches maturity to spread on the slide; by varying the focal point of the microscope one can perceive that the RBC nucleus is not on the same plane as the gametocyte. However, sometimes this appearance is misleading as it is interpreted in mature forms as that of a gametocyte that completely surrounds and engulfs the RBC nucleus (Figure 15).
Volutin
Heavy production of volutin is observed in a number of Haemoproteus species that infect raptors, particularly species from two related families: Accipitridae and Strigidae. It was not noted in species infecting the Falconidae.
Bishop & Bennett (1989) [2] noted that the observation of volutin sometimes depends on the staining. Valkiūnas [31] considered that these cytoplasmic inclusion are of little interest to systematics: “The presence and amount of valutin in gametocytes of the majority of haemoproteid species are variable characters. Taken separately, as a rule, these characters cannot be used for the identification of H. buteonis and other species”. On the contrary, we consider it to be a constant characteristic, playing an important metabolic role, and to present distinct variations between the species. Émile Brumpt followed the infection in S. aluco over long periods (many weeks). We observed that volutin was always present in samples from his collection and that its characteristics remained invariant even when the smears were partially discoloured (Figure 16). Therefore, we consider these granules as important indicators of the parasite’s metabolism, and given the morphology and property that are characteristic for each species, as useful for parasite systematics.
The nature of the volutin grains observed in avian Haemoproteus has not been determined but it is probable that they represent acidocalcisomes (personal communication of Prof. Docampo). These are acidic cytoplasmic inclusions that are frequently observed in plants and in different parasites such as Leishmania donovani [28], Toxoplasma gondii [27], Plasmodium berghei [17], Plasmodium falciparum [16] and Trypanosoma cruzi [29, 30]. In the latter, the authors did detect a polyphosphate kinase and exopolyphosphatase activity in the acidocalcisome, and suggested that the organelle has a metabolic role that allows the parasite to adapt to environmental changes [29]. It would be interesting to investigate their role in some of the avian Haemoproteidae where they appear particularly early and with abundance, in contrast to numerous other species where they are not observed following Giemsa staining.
Interpretation of the phylogenetic analysis
Phylogenetic analyses were based on partial sequences of the parasites’ gene coding for cytochrome b. Three well-supported groups emerge. The first group comprises Plasmodium species; the second group comprises Haemoproteus (Haemoproteus) species that include H. columbae and H. multipigmentatus Valkiūnas (2010) [35], both parasites of Columbiformes, and H. iwa Work & Rameyer (1996) [36], a parasite of Pelecaniformes, as well as other sequences that originate from Columbiformes and Pelecaniformes parasites that have not been identified at the species level; the third group contains H. turtur Covaleda (1950) [4], sensu Martinsen et al. (2006) [18], H. sacharovi (Novy & MacNeal 1904) [24] and H. syrnii (this study). The development of these last species in pupiparous dipterans justifies their classification in the Haemoproteus subgenus.
Another group, “Strix parasites” contains the H. syrnii (this study) and four other parasite sequences derived from other Strix. The Haemoproteus cyt b sequence identified by Martinsen et al. (2006) [18] diverges by 3% from the one we obtained. Two other Haemoproteus sequences found in parasites from Strix varia are identical to each other but differ from the H. syrnii sequence. Finally, a sequence from a parasite found in S. aluco in Germany [13] is identical to the one we obtained, though the author identified it as H. noctuae (Celli & San Felice 1891) [3], a species that, in contrast to H. syrnii, does not contain volutin. Thus, it is likely that the sequence attributed to H. noctuae is in fact from H. syrnii, a parasite that would have been missed during microscopic examination.
Taxonomic status
The hippoboscid fly reached us squashed between two glass slides, which precluded the possibility to identify the species. The species most frequently observed on the birds of prey in Europe is Ornithomyia avicularia (Linnaeus, 1761). This pupipara is a ubiquitous ectoparasite recorded in a diversity of bird species [6, 11]. Indeed, this species retains well-developed wings that allow it to switch to another host with ease, when the one it is on is in distress or dies [20]. Through this behaviour, this insect could play an important role as a vector of haemoproteids within bird nests, colonies or dormitories. In our study, the hippoboscid fly was collected engorged with the blood of a S. aluco that was infected by H. syrnii and to which it was attached. Some observations show that Haemoproteus may develop until the oocyst stage in an abnormal host however they do not reach the sporozoite stage as seen in our infected Hippoboscid fly [34].
We consider it most likely that the complete development of the parasite that we observed in the insect, i.e. the mature sporozoite stage, belongs to H. syrnii. We thus consider, pending confirmation, that this suggests that this species should be classified within the subgenus Haemoproteus.
Problems of species identification
Many natural infections are a mixture of different species. Ancient authors considered that each host harboured a single species and described morphological differences that were attributed to the variability of the same species. Molecular tools may sometimes detect the presence of more than one taxon. However, often only one of the species present in the blood is revealed and it is not always the predominant one. It could even be one that was overlooked when examining the slides with a microscope.
Valkiūnas et al. pointed out this bias [32], and also recommended great caution when comparing new sequences with those registered in GenBank which were not properly identified. They also considered that it is essential to associate the morphological description to the sequence data.
In several cases there are important discrepancies between the morphology in the original descriptions and the redescriptions by subsequent authors, and the sequences deposited in GenBank which are of uncertain identification.
An example of misidentifications relevant to our work is the following. In 2008, Paperna [25] succinctly described a “Haemoproteus syrnii” in Strix seloputo and Ninox scutulata from Singapore. A specimen of blood was sent to USA [19] for molecular analysis and the blood smears were deposited in the collection of the Muséum National d’Histoire Naturelle in Paris. It appeared that the parasite from Singapore [18, 19] and the parasite redescribed in this study are morphologically and molecularly two different species (Karadjian et al., unpublished data).
The subgenera of Haemoproteus
Three species considered to belong to the subgenus Haemoproteus cluster with the Parahaemoproteus group in the various trees proposed [23, 33]: H. syrnii, H. sacharovi and H. turtur. H. syrnii and H. saccharovi seem to be single infections. We have examined more than 15 blood smears positive for H. syrnii without finding any evidence of a multiple species infection. Blood smears of H. sacharovi in Zenaida macroura, a parasite with a very characteristic morphology, were examined thoroughly by Valkiūnas et al. [33] and Krizanauskiene et al. [12] who concluded that the infection was single. In the case of H. turtur isolated by Prof Paperna from S. senegalensis and sequenced by Martinsen et al. [19] we cannot at present be sure that the infection was pure.
Krizanauskiene et al. [12] on the sole basis of the sequences obtained from the partial cyt b concluded that the Haemoproteus of birds should be classified in two monophyletic groups: subgenus Haemoproteus comprising H. columbae and all species clustering with it and subgenus Parahaemoproteus gathering all the other species [12]. They challenged the experimental conditions of transmission by pupiparans of Haemoproteus saccharovi by Huff [10] and of H. turtur by Rashdan [26], both species clustering with Parahaemoproteus.
We think that it is premature to establish a systematic classification of the haemoproteid species based only on the cyt b partial sequences and to draw conclusions from these data on the classification of the parasites which is based by definition on their the life cycle, i.e. vector, type of sporogony and type of tissue schizogony [1, 7].
Acknowledgments
We thank all the volunteers at the l’Hôpital de la Faune Sauvage des Garrigues et Cévennes (C. Audic, M.T. Pallarès, and F. Ledru, to name but a few) as well as N. Labaeye and Dr. J. Bonvoisin who are in charge of the Centre Régional de Soins pour la Faune Sauvage du Nord Pas-de-Calais, and who constantly strive to save the birds of prey and the wild fauna of France. We are very grateful to M. and R. Killick-Kendrick for having suggested this study and for their unwavering help. We are equally very grateful to Profs Docampo R. and Moreno B. for providing us with information on volutin and for their help. Finally, many thanks to H. Ginsburg who never fails to help, to inform us or direct us to an expert who can answer our queries. The slides from Ilan Paperna’s collection were deposited in the collections of the Muséum National d’Histoire Naturelle de Paris, through the courtesy of Professor Prof. Jaap van Rijn, Director of the Department of Animal Sciences, The Robert H. Smith Faculty of Agriculture, Food and Environment, Rehovot, Israel. LD was supported by a postdoctoral fellowship from the Labex BCDiv (Biological and Cultural Diversities), Muséum National d’Histoire Naturelle de Paris.
References
- Bennett GF, Garnham PCC, Fallis AM. 1965. On the status of the genera Leucocytozoon Ziemann, 1893 and Haemoproteus Kruse, 1890 (Haemosporidiida: Leucocytozoidae and Haemoproteidae). Canadian Journal of Zoology, 43(6), 927–932. [CrossRef] [PubMed] [Google Scholar]
- Bishop MA, Bennett GF. 1989. The haemoproteids of the avian order Strigiformes. Canadian Journal of Zoology, 67, 2676–2684. [CrossRef] [Google Scholar]
- Celli A, San Felice F. 1891. Ueber die Parasiten des rothen Blutkörperchens im Menschen und in Thieren. Fortschritte der Medizin, 9, 499–511, 541–552, 581–586. [Google Scholar]
- Covaleda Ortega J, Gallego Berenguer J. 1950. Haemoproteus aviares. Revista Iberica de Parasitologia, 10, 141–185. [Google Scholar]
- Duval L, Robert V, Csorba G, Hassanin A, Randrianarivelojosia M, Walston J, Nhim T, Goodman SM, Ariey F. 2007. Multiple host-switching of Haemosporidia parasites in bats. Malaria Journal, 6, 157. [CrossRef] [PubMed] [Google Scholar]
- Facoz L. 1926. Diptères pupipares – Faune de France. Fédération Française des Sociétés de Sciences Naturelles – Office Centrale de faunistique. Lachevalier P. (Ed.), Paris, 64 pp. [Google Scholar]
- Garnham PCC. 1968. Malaria parasites and other Haemosporidia. Blackwell Scientific Publications, 1114 pp. [Google Scholar]
- Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O. 2010. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Systems Biology, 59(3), 307–321. [CrossRef] [PubMed] [Google Scholar]
- Hellgren O, Waldenstrom J, Perez-Tris J, Szoll E, Si O, Hasselquist D, Krizanauskiene A, Ottosson U, Bensch S. 2007. Detecting shifts of transmission areas in avian blood parasites: a phylogenetic approach. Molecular Ecology, 16(6), 1281–1290. [CrossRef] [PubMed] [Google Scholar]
- Huff C. 1932. Studies on Haemoproteus of mourning doves. American Journal of Epidemiology, 16, 618–623. [Google Scholar]
- Hutson AM. 1984. Keds, flat flies, and bat flies. Diptera, Hippoboscidae and Nycteribiidae. Handbook for the identification of British Insects, Vol. 10, Part 7. Royal Entomological Society of London, 40 pp. [Google Scholar]
- Krizanauskiene A, Iezhova TA, Sehgal RNM, Carlson JS, Palinauskas V, Bensch S, Valkiūnas G. 2013. Molecular characterization of Haemoproteus sacharovi (Haemosporida, Haemoproteidae), a common parasite of columbiform birds, with remarks on classification of haemoproteids of doves and pigeons. Zootaxa, 3613(1), 85–94. [Google Scholar]
- Krone O, Waldenstrom J, Valkiūnas G, Lessow O, Muller K, Iezhova TA, Fickel J, Bensch S. 2008. Haemosporidian blood parasites in European birds of prey and owls. Journal of Parasitology, 94(3), 709–715. [CrossRef] [Google Scholar]
- Kruse W. 1890. Ueber Blutparasiten. Archiv für pathologische Anatomie und Physiologie und für klinische Medizin, 121, 359–372, 453–485. [CrossRef] [Google Scholar]
- Levin II, Valkiūnas G, Santiago-Alarcon D, Cruz LL, Iezhova TA, O’Brien SL, Hailer F, Dearborn D, Schreiber EA, Fleischer RC, Ricklefs RE, Parker PG. 2011. Hippoboscid-transmitted Haemoproteus parasites (Haemosporida) infect Galapagos Pelecaniform birds: evidence from molecular and morphological studies, with a description of Haemoproteus iwa. International Journal for Parasitology, 41(10), 1019–1027. [CrossRef] [PubMed] [Google Scholar]
- Luo S, Marchesini N, Moreno SN, Docampo R. 1999. A plant-like vacuolar H(+)-pyrophosphatase in Plasmodium falciparum. FEBS Letter, 460(2), 217–220. [CrossRef] [Google Scholar]
- Marchesini N, Luo S, Rodrigues CO, Moreno SN, Docampo R. 2000. Acidocalcisomes and a vacuolar H+-pyrophosphatase in malaria parasites. Biochemical Journal, 347 Pt 1, 243–253. [CrossRef] [Google Scholar]
- Martinsen ES, Paperna I, Schall JJ. 2006. Morphological versus molecular identification of avian Haemosporidia: an exploration of three species concepts. Parasitology, 133(Pt 3), 279–288. [CrossRef] [PubMed] [Google Scholar]
- Martinsen ES, Perkins SL, Schall JJ. 2008. A three-genome phylogeny of malaria parasites (Plasmodium and closely related genera): evolution of life-history traits and host switches. Molecular Phylogenetics and Evolution, 47(1), 261–273. [CrossRef] [PubMed] [Google Scholar]
- Massonat E. 1909. Contribution à l’étude des pupipares. Annales de l’Université de Lyon. Nouvelle Série – I. Sciences, Médecine, Fascicule 24, 406 pp. [Google Scholar]
- Mayer M. 1911. Über ein Halteridium und Leucocytozoon des Waldkauzes une deren Weiterentwicklung in Stechmücken. Archiv für Protistenkunde, 21, 232–254. [Google Scholar]
- Mayer M. 1910. Über sein Entwicklung von Halteridium. Archiv für Schiffs und Tropenhygiene, 14, 197–202. [Google Scholar]
- Merino S, Hennicke J, Martinez J, Ludynia K, Torres R, Work TM, Stroud S, Masello JF, Quillfeldt P. 2012. Infection by Haemoproteus parasites in four species of frigatebirds and the description of a new species of Haemoproteus (Haemosporida: Haemoproteidae). Journal of Parasitology, 98(2), 388–397. [CrossRef] [Google Scholar]
- Novy F, MacNeal W. 1904. Trypanosomes and bird malaria. American Medicine, 8, 932–934. [Google Scholar]
- Paperna I, Keong MSC, May CYA. 2008. Haemosporozoan parasites found in birds in Peninsular Malaysia, Singapore, Sarawak and Java. Raffles Bulletin of Zoology, 56(2), 211–243. [Google Scholar]
- Rashdan NA. 1998. Role of Pseudolynchia canariensis in the transmission of Haemoproteus turtur from the migrant Streptopelia turtur to new bird hosts in Egypt. Journal of Egyptian Society for Parasitology, 28, 221–228. [Google Scholar]
- Rodrigues CO, Scott DA, Bailey BN, De Souza W, Benchimol M, Moreno B, Urbina JA, Oldfield E, Moreno SN. 2000. Vacuolar proton pyrophosphatase activity and pyrophosphate (PPi) in Toxoplasma gondii as possible chemotherapeutic targets. Biochemical Journal, 349(Pt 3), 737–745. [Google Scholar]
- Rodrigues CO, Scott DA, Docampo R. 1999. Presence of a vacuolar H+-pyrophosphatase in promastigotes of Leishmania donovani and its localization to a different compartment from the vacuolar H+-ATPase. Biochemical Journal, 340(Pt 3), 759–766. [CrossRef] [Google Scholar]
- Ruiz FA, Rodrigues CO, Docampo R. 2001. Rapid changes in polyphosphate content within acidocalcisomes in response to cell growth, differentiation, and environmental stress in Trypanosoma cruzi. Journal of Biological Chemistry, 276(28), 26114–26121. [CrossRef] [Google Scholar]
- Scott DA, Docampo R. 1998. Two types of H+-ATPase are involved in the acidification of internal compartments in Trypanosoma cruzi. Biochemical Journal, 331(Pt 2), 583–589. [Google Scholar]
- Valkiūnas G. 2005. Avian malaria parasites and other Haemosporidia. CRC Press, Boca Raton, Florida, 946 pp. [Google Scholar]
- Valkiūnas G, Atkinson CT, Bensch S, Sehgal RN, Ricklefs RE. 2008. Parasite misidentifications in GenBank: How to minimize their number? Trends in Parasitology, 24(6), 247–248. [CrossRef] [PubMed] [Google Scholar]
- Valkiūnas G, Iezhova TA, Evans E, Carlson JS, Martinez-Gomez JE, Sehgal RN. 2013. Two new Haemoproteus species (Haemosporida: Haemoproteidae) from columbiform birds. Journal of Parasitology, 99(3), 513–521. [CrossRef] [Google Scholar]
- Valkiūnas G, Kazlauskiene R, Bernotiene R, Palinauskas V, Iezhova TA. 2013. Abortive long-lasting sporogony of two Haemoproteus species (Haemosporida, Haemoproteidae) in the mosquito Ochlerotatus cantans, with perspectives on haemosporidian vector research. Parasitology Research, 112(6), 2159–2169. [CrossRef] [PubMed] [Google Scholar]
- Valkiūnas G, Santiago-Alarcon D, Levin II, Lezhova TA, Parker PG. 2010. A new Haemoproteus species (Haemosporida: Haemoproteidae) from the endemic Galapagos dove Zenaida galapagoensis, with remarks on the parasite distribution, vectors, and molecular diagnostics. Journal of Parasitology, 96(4), 783–792. [CrossRef] [Google Scholar]
- Work TM, Rameyer RA. 1996. Haemoproteus iwa n. sp. in great frigatebirds (Fregata minor) from Hawaii: parasite morphology and prevalence. Journal of Parasitology, 82(3), 489–491. [CrossRef] [Google Scholar]
Cite this article as: Karadjian G, Puech MP, Duval L, Chavatte JM, Snounou G & Landau I: Haemoproteus syrnii in Strix aluco from France: morphology, stages of sporogony in a hippoboscid fly, molecular characterization and discussion on the identification of Haemoproteus species. Parasite, 2013, 20, 32.
All Tables
Bird specimens (Strix aluco) studied, details about sampling and collection, and level of infection with Haemoproteus syrnii (−: uninfected; + to +++, level of infection).
All Figures
 |
Figures 1–16. Drawings of erythrocytic stages of Haemoproteus syrnii in the blood of Strix aluco. 1–4: young trophozoites with an initial volutin granule; 5: two young gametocytes with 1 or 2 volutin granules attached to the initial granule; 6–7 young gametocytes; 8–10 mature macrogametocytes; 11, 13: mature microgametocytes; 12: old microgametocyte; 14: two microgametocytes within the same RBC; 15: a microgametocyte spread beneath the RBC nucleus; 16: microgametocyte with faded staining and where the volutin granules are still visible. |
| In the text | |
 |
Figures 17–31. Microphotographs of Haemoproteus syrnii gametocytes in the blood of Strix aluco. 17–24: young gametocytes with the initial volutin granule (arrow); 25–26: immature gametocytes; 27–28: microgametocytes; 29: two macrogametocytes and a microgametocyte (arrow); 30: macrogametocyte and microgametocyte (arrow). Giemsa staining; 31: unstained smear: pigment of gametocyte. |
| In the text | |
 |
Figures 32–35. Microphotographs of developmental stages of Haemoproteus syrnii in the hippoboscid fly. 32: sporozoites issued from a burst oocyst and a gametocyte; 33: burst oocyst with sporozoites still attached to the cytomeres, and a gametocyte; 34: two mature sporozoites; 35: ookinetes. Giemsa staining. |
| In the text | |
 |
Figure 36. Maximum-likelihood phylogeny of mitochondrial cytochrome b lineages (346 bp) of avian Plasmodium spp. (8 sequences) and Haemoproteus spp. (21 sequences). Three sequences of Leucocytozoon spp. are used as outgroup. Bootstrap values > 70% are indicated near the nodes. GenBank accessions numbers and hosts (in parentheses) are indicated after the name of the parasite. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


